
As filed with the Securities and Exchange Commission on February 4, 2022
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
(Exact name of registrant as specified in its charter) |
2834 | ||||
(State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) |
(
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
Gerald T. Proehl
President and Chief Executive Officer
3525 Del Mar Heights Rd., #322
San Diego, CA 92130
(858) 800-2543
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Copies to:
Michael J. Lerner Steven M. Skolnick Lowenstein Sandler LLP 1251 Avenue of the Americas New York, New York 10020 (212) 262-6700 | Barry Grossman Sarah E. Williams Matthew Bernstein Ellenoff Grossman & Schole LLP 1345 Avenue of the Americas New York, New York 10105 (212) 370-1300 |
Approximate date of commencement of proposed sale to public:
As soon as practicable after this Registration Statement is declared effective.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box. ☒
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer | ☐ | Accelerated filer | ☐ |
☒ | Smaller reporting company | ||
Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided to Section 7(a)(2)(B) of the Securities Act. ☒
CALCULATION OF REGISTRATION FEE
Title of Each Class of Securities to Be Registered |
| Proposed Maximum Offering Price(1) |
|
| Amount of |
| ||
Units consisting of: |
|
|
|
|
|
| ||
(i) Common Stock, $0.0001 par value per share (3) |
| $ | 11,500,000 |
|
| $ | 1,117.80 |
|
(ii) Warrants to purchase Common Stock (4) |
|
| - |
|
|
| - |
|
Common Stock issuable upon exercise of the Warrants |
| $ | 11,500,000 |
|
| $ | 1,117.80 |
|
Pre-Funded Warrants to purchase shares of Common Stock and Common Stock issuable upon exercise thereof (5) |
| - |
|
| - | |||
Total |
| $ | 23,000,000 |
|
| $ | 2,235.60 |
|
____________
(1) | Estimated solely for the purpose of calculating the registration fee pursuant to Rule 457(o) under the Securities Act of 1933, as amended (the “Securities Act”). |
(2) | Calculated pursuant to Rule 457(o) based on an estimate of the proposed maximum aggregate offering price. |
(3) | Includes shares of Common Stock and/or warrants which may be issued on exercise of a 45-day option granted to the underwriter to cover over-allotments, if any. |
(4) | No separate registration fee required pursuant to Rule 457(g) under the Securities Act. |
(5) | The proposed maximum aggregate offering price of the Common Stock proposed to be sold in the offering will be reduced on a dollar-for-dollar basis based on the aggregate offering price of the Pre-Funded Warrants offered and sold in the offering (plus the aggregate exercise price of the Common Stock issuable upon exercise of the Pre-Funded Warrants), and as such the proposed aggregate maximum offering price of the Common Stock and Pre-Funded Warrants (including the Common Stock issuable upon exercise of the Pre-Funded Warrants), if any, is $11,500,000 (including the underwriter’s option to purchase additional shares of Common Stock). |
The Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until the Registration Statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
ii |
The information contained in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any state where the offer or sale is not permitted.
PRELIMINARY PROSPECTUS |
| SUBJECT TO COMPLETION |
| DATED FEBRUARY 4, 2022 |
Units consisting of
4,444,444 Shares of Common Stock or Pre-Funded Warrant to Purchase
4,444,444 Shares of Common Stock
and
Warrants to purchase up to 4,444,444 Shares of Common Stock

Dermata Therapeutics, Inc.
Dermata Therapeutics, Inc. is offering units, or Units, each consisting of one share of our common stock, $0.0001 par value per share (or pre-funded warrant in lieu thereof, each a Pre-Funded Warrant), and one warrant to purchase one share of our common stock, or each, a Warrant. The Units have no stand-alone rights and will not be certificated or issued as stand-alone securities. The common stock and Warrants are immediately separable and will be issued separately in this offering. Each Warrant offered hereby is immediately exercisable on the date of issuance at an exercise price per share of common stock equal to 100% of the offering price of the Unit in this offering, and will expire five years from the date of issuance.
We are also offering to certain purchasers whose purchase of Units in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if such purchasers so choose, in lieu of Units including shares of common stock, Units including Pre-Funded Warrants in lieu of shares of common stock that would otherwise result in any such purchaser’s beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. The purchase price of each Unit including a Pre-Funded Warrant will be equal to the price at which a Unit is sold to the public in this offering, minus $0.0001, and the exercise price of each Pre-Funded Warrant will be $0.0001 per share. For each Unit including a Pre-Funded Warrant we sell, the number of Units including a share of our common stock will be decreased on a one-for-one basis.
Pursuant to this prospectus, we are also offering the shares of common stock issuable upon exercise of the Warrants or the Pre-Funded Warrants.
Each Pre-Funded Warrant will be exercisable for one share of our common stock and will be exercisable at any time after its original issuance until exercised in full, provided that the purchaser will be prohibited from exercising any Pre-Funded Warrant for shares of our common stock if, as a result of such exercise, the purchaser, together with its affiliates and certain related parties, would own more than 4.99% of the total number of shares of our common stock then issued and outstanding. However, any holder may increase such percentage to any other percentage not in excess of 9.99%, provided that any increase in such percentage shall not be effective until 61 days after such notice to us.
The shares of common stock and Pre-Funded Warrants, if any, can each be purchased in this offering only with the accompanying Warrants as part of a Unit (other than pursuant to the underwriter’s option to purchase additional shares of common stock and/or Warrants), but the components of the Units will be immediately separable and will be issued separately in this offering.
Our common stock and certain of our outstanding warrants are currently listed on The Nasdaq Capital Market, or Nasdaq, under the symbols “DRMA” and “DRMAW,” respectively. The last reported sale price of our common stock on Nasdaq on January 31, 2022 was $2.25 per share. There is no established public trading market for the Warrants or the Pre-Funded Warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply to list the Warrants or the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system, including Nasdaq. Without an active trading market, the liquidity of the Warrants and the Pre-Funded Warrants will be limited.
The final public offering price per Unit will be determined through negotiation between us and the lead underwriter in the offering and the recent market price used throughout this prospectus may not be indicative of the final offering price.
We are an “emerging growth company” under the Jumpstart our Business Startups Act of 2012, or JOBS Act, and, as such, have elected to comply with certain reduced public company reporting requirements for this prospectus and future filings. See “Summary - Implications of Being an Emerging Growth Company.”
iii |
Investing in our securities is highly speculative and involves a high degree of risk. See “Risk Factors” beginning on page 12 of this prospectus for a discussion of information that should be considered in connection with an investment in our securities.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus. Any representation to the contrary is a criminal offense.
|
| Per Unit |
|
| Total |
| ||
Public offering price (1) |
| $ |
|
| $ |
| ||
Underwriting discounts and commissions (2) |
| $ |
|
| $ |
| ||
Proceeds to us, before expenses |
| $ |
|
| $ |
| ||
(1) | Units sold with Pre-Funded Warrants will be priced at the offering price minus $0.0001. |
(2) | Does not include the reimbursement of certain expenses of the underwriters. We refer you to “Underwriting” beginning on page 134 for additional information regarding the underwriter’s compensation. |
We have granted the representative of the underwriter an option, exercisable within 45 days from the date of this prospectus, to purchase from us, up to an additional 666,666 shares of common stock at the public offering price and/or up to an additional 666,666 Warrants to purchase up to 666,666 shares of common stock at a price per Warrant equal to the public offering price, less, in each case, the underwriting discounts and commissions, to cover over-allotments, if any. If the underwriter exercises the option in full, the total underwriting discounts and commissions payable will be $ , and the total proceeds to us, before expenses, will be $ .
The underwriter expects delivery of common stock (or Pre-Funded Warrant in lieu thereof) and Warrants comprising the Units will be made on or about , 2022.
Sole Book Running Manager Maxim Group LLC |
The date of this prospectus is , 2022
iv |
TABLE OF CONTENTS
|
| Page |
|
| 1 |
| |
| 7 |
| |
| 9 |
| |
| 10 |
| |
| 56 |
| |
| 57 |
| |
| 58 |
| |
| 59 |
| |
| 60 |
| |
| 61 |
| |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
| 62 |
|
| 73 |
| |
| 105 |
| |
| 112 |
| |
| 122 |
| |
| 124 |
| |
| 126 |
| |
|
|
|
|
| 132 |
| |
| 138 |
| |
| 144 |
| |
| 145 |
| |
| 146 |
| |
| F-1 |
|
We have not authorized anyone to provide you with different information, and we take no responsibility for any other information others may give you. If anyone provides you with different or inconsistent information, you should not rely on it. We are not, and the underwriters are not, making an offer to sell these securities in any jurisdiction where the offer or sale is not permitted. You should not assume that the information contained in this prospectus is accurate as of any date other than the date on the front of this prospectus.
No action is being taken in any jurisdiction outside the United States to permit a public offering of our common stock or possession or distribution of this prospectus in that jurisdiction. Persons who come into possession of this prospectus in jurisdictions outside the United States are required to inform themselves about and to observe any restrictions as to this offering and the distribution of this prospectus applicable to that jurisdiction.
We and the underwriters are offering to sell, and seeking offers to buy, our common stock only in jurisdictions where offers and sales are permitted. Neither we nor the underwriters have done anything that would permit this offering or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. Persons outside of the United States who come into possession of this prospectus must inform themselves about, and observe any restrictions relating to, the offering of our common stock and the distribution of this prospectus outside of the United States.
v |
| Table of Contents |
|
|
|
| PROSPECTUS SUMMARY
This summary highlights certain information contained elsewhere in this prospectus and does not contain all of the information that you should consider before making your investment decision. Before investing in our securities, you should carefully read this entire prospectus, including our financial statements and the related notes thereto and the information set forth in the sections titled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” If any of the risks materialize, our business, financial condition, operating results and prospects could be materially and adversely affected. In that event, the price of our common stock or Warrants could decline, and you could lose part or all of your investment. Unless the context otherwise requires, we use the terms “Dermata,” “company,” “we,” “us” and “our” in this prospectus to refer to Dermata Therapeutics, Inc. Except where the historical context specifically requires otherwise, disclosures in this prospectus reflect our conversion from a limited liability company to a corporation on March 24, 2021.
Our Company
We are a clinical-stage biotechnology company focusing on the treatment of medical and aesthetic skin conditions. Our lead product candidate, DMT310, is the first product candidate being developed from our Spongilla technology platform. DMT310 is a once-weekly topical product derived from a naturally sourced freshwater sponge with multiple components, including mechanical and chemical mechanisms of action. Based on DMT310’s multiple mechanisms of action, it is currently under clinical development for the treatment of acne vulgaris (or acne), psoriasis vulgaris (or psoriasis), and papulopustular rosacea (or rosacea). To date, DMT310 has generated efficacy data in a Phase 2b trial of acne, and an efficacy signal in a Phase 1b trial of psoriasis, and we are currently collecting efficacy data in a Phase 2 trial in rosacea. DMT410 is our second product candidate, which is a combination treatment regimen that uses the unique mechanical features of our Spongilla technology to facilitate the intradermal delivery of botulinum toxin by topical application rather than intradermal injections with a needle. The treatment is intended to consist of an initial topical application of our proprietary Spongilla powder to the treatment area where Spongilla’s spicules penetrate the stratum corneum, creating microchannels into the dermis allowing for the topical application and penetration of the botulinum toxin. We are investigating DMT410 as a novel method for topical intradermal delivery of botulinum toxin for the treatment of multiple aesthetic and medical skin conditions. To date, we have completed a Phase 1b proof-of-concept, or POC, trial for the treatment of axillary hyperhidrosis and a Phase 1b POC trial of multiple aesthetic skin conditions. In all clinical trials with DMT310 and DMT410, we believe the data collected supports our belief about the product candidates’ safety and tolerability by patients.
Our Spongilla technology platform is based on the use of a naturally growing freshwater sponge, Spongilla lacustris, or Spongilla. Spongilla is a freshwater sponge that grows in rivers and lakes in select regions of the world, typically in the northern hemisphere. However, there are only a limited number of places in which it grows in the quantity, and of the quality, necessary to be used as raw material in a commercial pharmaceutical product. One such location is the Volga River in central Russia. We have signed an exclusive supply agreement with one of the larger known harvesters of Spongilla raw material in the Volga River area for utilization in DMT310 and DMT410. Our exclusive supplier has been harvesting Spongilla for over eighteen (18) years and has the capacity to collect and process large quantities of Spongilla raw material per year. Traditionally, locals would harvest the sponge to be used as a folk medicine to treat various inflammatory ailments, but research has been conducted to evaluate the use of Spongilla for the possible treatment of various skin diseases and aesthetic conditions.
The Spongilla technology used in our product candidates, such as DMT310 and DMT410, has unique chemical and mechanical components, which are produced by Spongilla for its natural defense mechanisms. These specialized mechanisms consist of unique, naturally occurring microscopic siliceous spicules that are uniquely sized to penetrate a patient’s skin, creating microchannels which allow for the naturally occurring chemical components of the Spongilla, or other macromolecules such as botulinum toxin, to penetrate into the dermis for a clinical result. In addition to the creation of microchannels, the mechanical spicules are also believed to open closed comedones (also known as blackheads) and promote collagen production which may accelerate the skin’s rejuvenation period, thus accelerating the skins natural healing properties. These mechanical components are coupled with chemical components that have been shown in-vitro to directly kill C. acnes, the bacteria that cause inflammatory lesions in acne patients. During in-vitro studies for DMT310, our first product candidate utilizing our Spongilla technology, we observed the inhibition of lipogenesis of sebocytes, which may translate to a reduction in sebum (an oily and waxy substance produced by the human body’s sebaceous glands) production and oiliness of the skin in patients. Additionally, we have shown DMT310 in-vitro has a dose dependent down regulation of two inflammatory cytokines, IL-17A and IL-17F, which are believed to play a role in causing inflammation in psoriasis and rosacea. We believe the dual mode of action of our Spongilla technology may allow our product candidates to treat many different skin diseases and aesthetic conditions, unlike other current products that treat only one aspect of a disease. We believe these unique properties may also only require DMT310 to be applied once-weekly, which, if approved for psoriasis, acne, or rosacea would make DMT310 the first once-weekly topical prescription product for diseases of the skin on the market. |
|
|
|
|
| 1 |
| Table of Contents |
|
|
|
| We received results from our Phase 2b clinical trial of DMT310 for the once-weekly treatment of moderate-to-severe acne in June 2020. This trial produced statistically significant results for all efficacy endpoints, including absolute reduction in inflammatory lesions, non-inflammatory lesions and the Investigator Global Assessment (or IGA), of acne, as early as week four (4) and continued until the end of the trial at week twelve (12). The clinical endpoints used in the Phase 2b trial are identical to those endpoints we intend to use in our two Phase 3 acne clinical trials, which trials we believe will form the basis of our new drug application, or NDA, for DMT310.
While moderate-to-severe acne is the first indication for which we are developing DMT310, based on the data obtained from non-clinical trials of DMT310, which suggests DMT310’s ability to inhibit various cytokines that trigger inflammatory responses in humans. In March 2021, we initiated a Phase 1b POC trial of DMT310 for once-weekly topical treatment of mild-to-moderate psoriasis. This signal detection trial produced safety, tolerability, and efficacy results at multiple timepoints that we believe warrant further development in psoriasis by showing an improvement in the Physician’s Global Assessment, or PGA, and a decrease in pruritis (itch). Prior to initiating a Phase 2a trial in psoriasis in the first half of 2022, we intend to conduct some additional work to better inform the clinical design of the Phase 2 trial. In addition, we recently enrolled our first patient in a Phase 2 trial of DMT310 for the once-weekly treatment of rosacea, another inflammatory skin disease similar to acne, which we believe has an unmet need in the market. We anticipate results from this Phase 2 rosacea trial in the second half of 2022.
We believe there is a growing demand within the dermatology market, which consists of both medical and aesthetic skin conditions including acne vulgaris, psoriasis vulgaris, papulopustular rosacea, hyperhidrosis, reduction of fine lines and pore size and improved skin quality, for innovative, convenient, and naturally derived treatment options. We believe our Spongilla technology provides us the opportunity to develop product candidates that satisfy this growing demand, as there are no other naturally derived prescription products currently on the market for the treatment of these skin diseases and conditions which impact millions of people worldwide annually.
Our Clinical Development Pipeline
|
|
|
|
|
|
|
|
| 2 |
| Table of Contents |
|
|
| ||
| Our Strategy
We plan on developing, in-licensing, and commercializing differentiated medical and aesthetic dermatology product candidates for the treatment of various skin diseases and conditions, which we believe have significant unmet needs in the market. The key components of this strategy are as follows: |
| ||
|
|
|
|
|
|
| • | Complete development and regulatory approval of DMT310 for acne. We have completed a Phase 2b clinical trial of DMT310 for the treatment of moderate-to-severe acne. We plan to hold an end of Phase 2 meeting with the U.S. Food and Drug Administration, or FDA, in the second half of 2022, and subject to the end of this Phase 2 meeting, we plan to initiate two pivotal Phase 3 clinical trials for DMT310 in the second half of 2022, with top-line results expected in the first half of 2024. Assuming we receive positive clinical results from the two pivotal Phase 3 clinical trials in acne, we would expect to submit an NDA for DMT310 for the treatment of moderate-to-severe acne in the second half of 2024. |
|
|
|
|
|
|
|
| • | Initiate a Phase 2 trial of DMT310 for the treatment of psoriasis. In October 2021, we announced top-line results of our Phase 1b POC trial in patients with mild-to-moderate psoriasis. We believe the data seen in this POC trial warrant the continued development of DMT310 for the treatment of psoriasis. We plan to initiate a Phase 2 trial for DMT310 in psoriasis in the first half of 2022. If successfully developed and commercialized, we believe DMT310 would be the first once-weekly topical product available to treat psoriasis. |
|
|
|
|
|
|
|
| • | Complete a Phase 2 trial of DMT310 in rosacea. Based on the data obtained from our Phase 2 trials in acne and the anti-inflammatory results seen in-vitro, we initiated a Phase 2 trial of DMT310 for the treatment of rosacea in November 2021, with top-line results expected in the second half of 2022. We believe this will help bolster our pipeline and could establish the broader use of DMT310 in multiple inflammatory skin diseases and address unmet needs for rosacea patients. |
|
|
|
|
|
|
|
| • | Explore mutually beneficial partnership opportunities for our DMT410 program in hyperhidrosis and aesthetic skin conditions. In November 2021, we announced top-line results of our Phase 1b POC trial of DMT410 for the treatment of multiple aesthetic skin conditions using our Spongilla technology for the topical application of OnabotulinumtoxinA (brand name BOTOX®). We believe this trial provides further data of the ability for DMT410 to topically deliver botulinum toxin into the dermis for skin conditions and diseases. Based on the results of DMT410 in hyperhidrosis and aesthetics, we are currently exploring partnership opportunities to further develop DMT410 for the topical treatment of skin diseases and aesthetic skin conditions. |
|
|
|
|
|
|
|
| • | Acquire or in-license additional dermatology product candidates to our portfolio that complement our current product candidates. We continuously evaluate potential partnering opportunities that will bolster our current product candidate portfolio and provide substantial value to our organization. We intend to focus on early to mid-stage development product candidates to generate clinical data and potentially move to later stages of development and ultimately on to commercialization. |
|
|
|
|
|
|
|
| • | Maximize the value of our portfolio by commercializing our product candidates in territories where we can do so effectively and partner for other territories to help us reach new markets. If we receive FDA approval for our product candidates, we plan to maximize the territories where they could be sold by partnering with established companies in new territories outside of the U.S. market, if possible. |
|
|
|
|
|
|
|
| • | Further strengthen our intellectual property portfolio, path to new chemical entity, or NCE, exclusivity, raw material supply and advance our regulatory filings. We plan to continue to strengthen our IP portfolio, seek NCE exclusivity for DMT310, maintain our exclusive supply agreement for our raw material requirements, and continue to protect our proprietary information. We believe these activities will be our primary competitive advantages if our product candidates receive regulatory approval. |
|
|
|
|
|
|
| The dates reflected in the foregoing are estimates only, and there can be no assurances that the events included will be completed on the anticipated timeline presented, or at all. Further, there can be no assurances that we will be successful in the development of DMT310 or DMT410, or any other product candidate we may develop in the future, or that DMT310 or DMT410, or any other product candidate we may develop in the future, will receive FDA approval for any indication. |
| ||
|
|
|
|
|
| 3 |
| Table of Contents |
|
|
|
| Our Team
Our founders and management team have many years of experience in the biotechnology space holding senior management and board positions at leading life science companies, including Hoechst Marion Roussel, Johnson & Johnson, Santarus, Inc., SkinMedica Inc., Allergan Inc., Receptos Inc., Dura Pharmaceuticals Inc., and Micromet Inc., among others, and possess substantial experience and expertise across the spectrum of drug development and commercialization of pharmaceutical products.
Gerald T. Proehl, our Co-Founder, Chairman, President and Chief Executive Officer, was previously President and Chief Executive Officer of Santarus, Inc. until its acquisition by Salix Pharmaceuticals Inc. in 2014 for $2.6 billion and has also served in senior leadership roles at Hoechst Marion Roussel. David Hale, our Co-Founder and Lead Director, is Chairman and Chief Executive Officer of Hale BioPharma Ventures, LLC and has over 30 years of senior leadership experience at both large and small biopharmaceutical companies, including as Chairman of Santaurus, Inc. and SkinMedica Inc. Kyri Van Hoose, C.P.A and M.B.A, our Senior Vice President, Chief Financial Officer, has served as Chief Financial Officer, the head of finance, and other senior leadership finance and accounting roles with life sciences companies for over 15 years. Christopher J. Nardo, M.P.H., Ph.D., our Senior Vice President, Development, previously served as Senior Director, Clinical Development at Allergan Inc. and Vice President, Clinical Operations, at Spectrum Pharmaceuticals Inc. Maria Munera, Ph.D., M.B.A., our Senior Vice President, Regulatory Affairs and Quality Assurance, previously served in similar roles at Receptos Inc. and Santarus, Inc. The development, operational, and commercial expertise of our executive team will be essential as we execute on our strategy of building a successful dermatology biopharmaceutical company to deliver significant value to both patients and stockholders.
Recent Developments
Initial Public Offering
On August 17, 2021, we closed our initial public offering, or IPO, of 2,571,428 shares of our common stock and accompanying warrants to purchase up to 2,571,428 shares of common stock. Each share of common stock was sold together with one warrant to purchase one share of common stock with an exercise price of $7.00 per share at a combined offering price of $7.00, for gross proceeds of approximately $18.0 million, before deducting underwriting discounts and offering expenses. In addition, we granted the underwriters a 45-day option to purchase up to an additional 385,714 shares of common stock and/or warrants to purchase 385,714 shares of common stock to cover over-allotments at the initial public offering price, less the underwriting discount, of which the underwriter exercised its option to purchase an additional 385,714 warrants.
DMT310 Top-line Psoriasis Results
On October 18, 2021, we announced top-line results from a Phase 1b POC clinical trial of DMT310 for the treatment of mild-to-moderate psoriasis. DMT310 demonstrated improvements in Physician Global Assessment (or PGA), Psoriasis Area and Severity Index (or PASI), and pruritus scores for the target lesion. DMT310 achieved a PGA score of 0 or 1 for the target lesion in 29.6% of patients, a total PASI score of 0 or 1 for the target lesion in 25.9% of patients, and a 19.6% reduction from baseline in pruritus at week 8. DMT310 also appeared to be well tolerated with most tolerability issues being mild in severity.
Enrolled First Patient in DMT310 Phase 2 Rosacea Trial
On November 17, 2021, we announced that we had enrolled the first patient in a Phase 2 trial of the once-weekly application of DMT310 for the treatment of rosacea. This study is a 12-week, multi-center, randomized, double-blinded, placebo-controlled trial designed to evaluate the tolerability, safety, and efficacy of once-weekly dosing of DMT310 in 180 moderate-to-severe rosacea patients. The primary endpoints include the mean change in inflammatory lesion counts and IGAs using a 5-point scale, with IGA treatment success being defined as an IGA score of “clear” or “almost clear” and a 2-point improvement from baseline at week 12. We expect to have top-line results in the second half of 2022.
DMT410 Top-line Aesthetic Results
On November 19, 2021, we announced top-line results from a Phase 1b POC trial of DMT410 for the treatment of multiple aesthetic skin conditions using a topical application of botulinum toxin. DMT410 demonstrated an improvement in multiple aesthetic skin quality endpoints including a decrease in pore size, decrease in sebum production, improvement in the Global Aesthetic Improvement score, improvement in luminosity, and an improvement in brightness all peaking at 2-3 months post treatment. This trial also saw no serious adverse events and no potential distant spread of botulinum toxin leading us to believe the topically applied botulinum toxin remained in the dermis and did not migrate to the muscle which could have led to unwanted side effects. |
|
|
|
|
| 4 |
| Table of Contents |
|
|
|
|
|
Summary of Risks Associated with Our Business | ||||
|
|
|
|
|
| Our ability to implement and execute our business strategy is subject to numerous risks and uncertainties that you should be aware of prior to making any investment decisions. The risks described in the section titled “Risk Factors” immediately following this summary may cause us to not realize the full benefits of our strengths or to be unable to successfully execute all or part of our strategy. These risks include, but are not limited to, the following: |
| ||
|
|
|
|
|
|
| • | We are a pre-revenue, clinical-stage biopharmaceutical company with limited operating history. |
|
|
|
|
|
|
|
| • | We have incurred significant losses in the past and expect to incur additional significant losses and may never become profitable or be able to sustain profitability. |
|
|
|
|
|
|
|
| • | The net proceeds of this offering will only fund our operations into February 2023 and we will need to raise additional capital to support our product development and commercialization efforts. |
|
|
|
|
|
|
|
| • | Our business success is dependent on our ability to successfully complete the development of our main product candidates, DMT310 and DMT410, and other product candidates. The development process is long and expensive and success is uncertain. |
|
|
|
|
|
|
|
| • | Even if we successfully complete the development and clinical testing of our product candidates, we still need to obtain regulatory approval from the FDA in order to market our products in the U.S., which may be difficult to obtain. |
|
|
|
|
|
|
|
| • | We may not receive regulatory approval for any of our product candidates, and even if we receive such regulatory approval, we may not be able to successfully commercialize any products and the revenue that we generate from sales, if any, may be limited. |
|
|
|
|
|
|
|
| • | The commercial markets we are targeting are highly competitive and we may be unsuccessful commercializing due to, among other things, third-party coverage and reimbursement policies, and unfavorable pricing models. |
|
|
|
|
|
|
|
| • | Although we attempt to keep apprised of risks related to competition in commercialization of our product candidates, it is difficult to predict the effects of third-party activities on our development activities and future markets for our product candidates. Also, we cannot predict the effect potential competing patent claims may have on our patent portfolio and our business. |
|
|
|
|
|
|
|
| • | It is difficult and costly to protect our intellectual property rights. Accordingly, we cannot predict the breadth of patent claims that may be allowable, or whether any claims will be allowed in our pending patent applications or, the enforceability of our existing and future patents. DMT310, DMT410 and our other product candidates may infringe the intellectual property rights of others, all of which could increase our costs and delay or prevent our development and commercialization efforts. |
|
|
|
|
|
|
|
| • | Clinical drug development involves a lengthy and expensive process with an uncertain outcome, and results of earlier studies and trials may not be predictive of future trial results. |
|
|
|
|
|
|
|
| • | If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to limit product development and commercialization of our product candidates. |
|
|
|
|
|
|
|
| • | The market for DMT310 and DMT410 may not be as large as we expect. |
|
|
|
|
|
|
|
| • | The raw material for our product candidates, DMT310 and DMT410, is derived from naturally occurring ingredients that grow only in limited areas that need to be harvested annually. Due to unforeseen environmental conditions or circumstances, our supplier may not be able to harvest as much raw material as we require, or any at all, which may negatively impact our ability to conduct preclinical studies, clinical trials, and ultimately commercialize our product candidates. |
|
|
|
|
|
|
|
| • | We currently rely on a third party for the raw materials needed for DMT310 and DMT410, and if we encounter any difficulties in accessing or procuring alternative sources on acceptable terms, or at all, our business may suffer. |
|
|
|
|
|
|
| 5 |
| Table of Contents |
|
|
|
|
|
|
| • | Our current issued patents covering DMT310 expire in 2022 and 2023, which is prior to our anticipated date for any market launch. Our current pending patents covering DMT310 and DMT410 have not been issued yet and there is no guarantee they will get issued. We may not be able to obtain additional patent coverage, which could limit our market opportunity due to competition from other products. |
|
|
| • | We will need to add personnel, which will increase the size and complexity of our organization and we may experience difficulties executing growth and corporate strategies. |
|
|
| • | The continued spread of COVID-19 and the resulting global pandemic could adversely impact our clinical trial operations, including our ability to recruit and retain patients and principal investigators and site staff who, as healthcare providers, may have heightened exposure to COVID-19 if an outbreak occurs in their geography. |
|
|
| • | After this offering, our directors, executive officers, and certain stockholders (certain of which are affiliates of members of our board of directors) will continue to own a significant percentage of our common stock and, if they choose to act together, will be able to exert significant control over matters subject to stockholder approval. |
|
|
|
| ||
| Corporate Information
We were formed as a Delaware limited liability company under the name Dermata Therapeutics, LLC in December 2014. On March 24, 2021, we converted into a Delaware corporation and changed our name to Dermata Therapeutics, Inc. Our website address is www.dermatarx.com and our telephone number is 858-800-2543. Information contained in, or accessible through, our website does not constitute part of this prospectus or registration statement and inclusions of our website address in this prospectus or registration statement are inactive textual references only.
“Dermata” and our other common law trademarks, service marks or trade names appearing herein are the property of Dermata Therapeutics, Inc. We do not intend the use or display of other companies’ trademarks and trade names to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
Implications of Being an Emerging Growth Company
As a company with less than $1.0 billion in revenue during our most recently completed fiscal year, we qualify as an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. An emerging growth company may take advantage of relief from certain reporting requirements and other burdens that are otherwise applicable generally to public companies. These provisions include: |
| ||
|
|
|
|
|
|
| • | reduced obligations with respect to financial data, including presenting only two years of audited financial statements and only two years of selected financial data in this prospectus; |
|
|
|
|
|
|
|
| • | an exception from compliance with the auditor attestation requirement of Section 404 of the Sarbanes-Oxley Act of 2002, as amended, or the Sarbanes-Oxley Act; |
|
|
|
|
|
|
|
| • | reduced disclosure about our executive compensation arrangements in our periodic reports, proxy statements, and registration statements; and |
|
|
|
|
|
|
|
| • | exemptions from the requirements of holding non-binding advisory votes on executive compensation or golden parachute arrangements. |
|
|
|
|
|
|
| We may take advantage of exemptions for up to five years or such earlier time that we are no longer an emerging growth company. Accordingly, the information contained herein may be different than the information you receive from other public companies in which you hold stock. We would cease to be an emerging growth company upon the earliest to occur of: (1) the last day of the fiscal year in which we have more than $1.07 billion in annual gross revenue or (2) following the fifth anniversary of the closing of this offering, (3) we are deemed to be a “large accelerated filer” under the rules of the U.S. Securities and Exchange Commission, or SEC, which means the market value of our securities that is held by non-affiliate exceeds $700 million as of the last day of the second fiscal quarter, or June 30, of any calendar year, and (4) the issuance of, in any three year period, by us of more than $1.0 billion in non-convertible debt securities.
The JOBS Act also permits us, as an emerging growth company, to take advantage of an extended transition period to comply with the new or revised accounting standards applicable to public companies and thereby allow us to delay the adoption of those standards until those standards would apply to private companies. We have irrevocably elected not to avail ourselves of this exemption and therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies. |
| ||
|
|
|
|
|
| 6 |
| Table of Contents |
|
|
|
|
|
THE OFFERING | ||||
|
|
|
|
|
| Units being offered |
| 4,444,444 Units. Each Unit will consist of one share of common stock (or Pre-Funded Warrant to purchase one share of our common stock in lieu thereof) and one Warrant to purchase one share of common stock. The Units have no stand-alone rights and will not be certificated or issued as stand-alone securities. The shares of common stock and Pre-Funded Warrants, if any, can each be purchased in this offering only with the accompanying Warrants as part of Units (other than pursuant to the underwriter’s option to purchase additional shares of common stock and/or Warrants), but the components of the Units will be immediately separable and will be issued separately in this offering. |
|
|
|
|
|
|
| Number of shares of common stock outstanding immediately before this offering |
| 8,328,629 shares |
|
|
|
|
|
|
| Number of shares of common stock to be outstanding after this offering (1) |
| 12,773,073 shares (or 13,439,739 shares if the underwriter exercises the option to purchase additional shares in full, and assuming in each case, no exercise of the Warrants and no sale of Units including a Pre-Funded Warrant). |
|
|
|
|
|
|
| Description of the Warrants |
| The Warrants will have an exercise price per share of common stock equal to 100% of the offering price of the Unit in this offering, will be immediately exercisable and will expire five years from the date of issuance. Each Warrant is exercisable for one share of common stock, subject to adjustment in the event of stock dividends, stock splits, stock combinations, reclassifications, reorganizations or similar events affecting our common stock. A holder may not exercise any portion of a Warrant to the extent that the holder, together with its affiliates and any other person or entity acting as a group, would own more than 4.99% of our outstanding shares of common stock after exercise, as such ownership percentage is determined in accordance with the terms of the Warrants, except that upon notice from the holder to us, the holder may waive such limitation up to a percentage, not in excess of 9.99%.
This prospectus also relates to the offering of the common stock issuable upon exercise of the Warrants. To better understand the terms of the Warrants, you should carefully read the “Description of Capital Stock” section of this prospectus. You should also read the form of Warrant, which is filed as an exhibit to the registration statement that includes this prospectus.
|
|
| Description of Pre-Funded Warrants |
| We are also offering to certain purchasers whose purchase of Units in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if such purchasers so choose, in lieu of Units including shares of common stock, Units including Pre-Funded Warrants in lieu of shares of common stock that would otherwise result in any such purchaser’s beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock. The purchase price of each Unit including a Pre-Funded Warrant will be equal to the price at which a Unit is sold to the public in this offering, minus $0.0001, and the exercise price of each Pre-Funded Warrant will be $0.0001 per share.
Each Pre-Funded Warrant will be exercisable for one share of our common stock and will be exercisable at any time after its original issuance until exercised in full, provided that the purchaser will be prohibited from exercising Pre-Funded Warrants for shares of our common stock if, as a result of such exercise, the purchaser, together with its affiliates and certain related parties, would own more than 4.99% of the total number of shares of our common stock then issued and outstanding. However, any holder may increase such percentage to any other percentage not in excess of 9.99%, provided that any increase in such percentage shall not be effective until 61 days after such notice to us. |
|
|
|
|
|
|
| Use of proceeds |
| We estimate that we will receive net proceeds from this offering of approximately $9.0 million, or approximately $10.4 million if the underwriter exercise its overallotment option in full, based upon an assumed public offering price of $2.25 per share (the last reported sale price of our common stock on the Nasdaq Capital Market on January 31, 2022) and after deducting the underwriting discounts and commissions and estimating offering expenses payable by us and excluding the proceeds, if any, from the exercise of the Warrants issued as part of the Units and assuming no sale of Units including Pre-Funded Warrants. |
|
|
|
|
|
|
|
|
| We intend to use substantially all of the net proceeds from this offering to complete FDA required studies to hold an End of Phase 2 meeting for the DMT310 acne program, initiate a DMT310 Phase 2 psoriasis trial, complete our DMT310 Phase 2 rosacea trial, advance the clinical development of DMT410 for the treatment of various aesthetic indications, manufacturing drug product for our DMT310 Phase 3 acne program, and for working capital and other general corporate purposes. |
|
|
|
|
|
|
| 7 |
| Table of Contents |
|
|
|
|
| ||||
| Lock-up |
| In connection with this offering, we, our directors and executive officers have agreed not to offer, issue, sell, contract to sell, encumber, grant any option for the sale of or otherwise dispose of any of our securities for a period of ninety (90) days following the closing of this offering. See “Underwriting” for more information. |
| ||||
|
|
|
|
| ||||
| Underwriter’s over-allotment option |
| We have granted the underwriters an option, exercisable within 45 days after the closing of this offering, to acquire up to an additional 15% of the total number of shares of common stock and/or Warrants to be offered by us pursuant to this offering, solely for the purpose of covering over-allotments. |
| ||||
|
|
|
|
| ||||
| Stock exchange symbol |
| Our common stock and certain of our warrants trade on the Nasdaq Capital Market under the symbols “DRMA” and “DRMAW,” respectively. There is no established public trading market for the Warrants or the Pre-Funded Warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply to list the Warrants or the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system, including Nasdaq. Without an active trading market, the liquidity of the Warrants and the Pre-Funded Warrants will be limited. |
| ||||
|
|
|
|
| ||||
| Risk factors |
| Investing in our Units involves a high degree of risk. See “Risk Factors” beginning on page 10, and the other information included in this prospectus for a discussion of factors you should consider carefully before deciding to invest in our Units. |
| ||||
|
|
|
|
| ||||
| (1) The number of shares of our common stock to be outstanding immediately after this offering is based on 8,328,629 shares of our common stock outstanding as of January 31, 2022, and excludes: | |||||||
|
|
|
| |||||
|
| • | 1,000,326 shares of common stock issuable upon exercise of stock options, at a weighted-average exercise price of $3.96 per share; | |||||
|
|
|
|
| ||||
|
| • | 2,957,142 shares of common stock issuable upon exercise of warrants issued in our initial public offering, at an exercise price of $7.00 per share; |
| ||||
|
|
|
|
| ||||
| • | 134,515 shares of common stock issuable upon exercise of other warrants outstanding, at a weighted-average exercise price of $13.33 per share; |
| |||||
|
|
|
|
| ||||
|
| • | 731,173 shares of our common stock that are available for future issuance under our 2021 Omnibus Equity Incentive Plan (or, the 2021 Plan) or shares that will become available under our 2021 Plan; and |
| ||||
|
|
|
|
| ||||
|
| • | all shares of our common stock underlying the Pre-Funded Warrants and Warrants to be issued in this offering. |
| ||||
|
|
|
|
| ||||
| Unless otherwise indicated, this prospectus reflects and assumes the following: |
| ||||||
|
|
|
|
| ||||
|
| • | no exercise of outstanding options or warrants described above; |
| ||||
|
|
|
|
| ||||
|
| • | no exercise of the Warrants, and no sale of Units including a Pre-Funded Warrant; and |
| ||||
|
|
|
|
| ||||
|
| • | no exercise by the underwriter of its over-allotment option. |
| ||||
|
|
|
|
| ||||
| 8 |
| Table of Contents |
SUMMARY SELECTED FINANCIAL DATA
You should read the following summary selected financial data together with our financial statements and the related notes appearing at the end of this prospectus and the “Management’s Discussion and Analysis of Financial Condition and Results of Operations” section of this prospectus. We have derived the statements of operations for the nine months ended September 30, 2020 and September 30, 2021, and the balance sheet data as of September 30, 2021 from our unaudited interim financial statements appearing at the end of this prospectus. These unaudited financial statements have been prepared on a basis consistent with our audited financial statements and, in our opinion, contain all adjustments, consisting only of normal and recurring adjustments, necessary for a fair presentation of such financial data. We have derived the statement of operations data for the years ended December 31, 2019 and December 31, 2020 from our audited financial statements appearing at the end of this prospectus. Our historical results are not necessarily indicative of results that should be expected in any future period.
|
| Year Ended |
|
| For the Nine Months Ended |
| ||||||||||
|
| December 31, |
|
| September 30, |
| ||||||||||
Statements of Operations Data: |
| 2019 |
|
| 2020 |
|
| 2020 |
|
| 2021 |
| ||||
|
|
|
|
|
| (unaudited) |
|
| (unaudited) |
| ||||||
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
| ||||
Research and development |
| $ | 2,623,280 |
|
| $ | 1,607,819 |
|
| $ | 1,493,520 |
|
| $ | 2,347,564 |
|
General and administrative |
|
| 1,847,328 |
|
|
| 1,565,034 |
|
|
| 1,187,906 |
|
|
| 2,956,444 |
|
Total operating expenses |
|
| 4,470,608 |
|
|
| 3,172,853 |
|
|
| 2,681,426 |
|
|
| 5,304,008 |
|
Loss from operations |
|
| (4,470,608 | ) |
|
| (3,172,853 | ) |
|
| (2,681,426 | ) |
|
| (5,304,008 | ) |
Other income and expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Forgiveness of Paycheck Protection Plan loan |
|
| - |
|
|
| (133,592 | ) |
|
| - |
|
|
| - |
|
Interest expense, net |
|
| 250,748 |
|
|
| 197,269 |
|
|
| 158,791 |
|
|
| 45,613 |
|
Net loss |
| $ | (4,721,356 | ) |
| $ | (3,236,530 | ) |
| $ | (2,840,217 | ) |
| $ | (5,349,621 | ) |
Deemed dividend upon the redemption of 5,221,156 shares of Series 1c preferred stock (see Notes to Financial Statements) |
| $ | - |
|
| $ | - |
|
| $ | - |
|
| $ | 269,038 |
|
Deemed dividend upon the amendment of terms of the Series 1d convertible preferred stock (see Notes to Financial Statements) |
| $ | - |
|
| $ | - |
|
| $ | - |
|
| $ | 2,293,199 |
|
Net loss attributable to common stockholders |
| $ | (4,721,356 | ) |
| $ | (3,236,530 | ) |
| $ | (2,840,217 | ) |
| $ | (7,911,858 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per share of common stock, basic and diluted |
| $ | (2.47 | ) |
| $ | (1.69 | ) |
| $ | (1.49 | ) |
| $ | (2.69 | ) |
Weighted-average basic and diluted common units/shares |
|
| 1,911,009 |
|
|
| 1,911,009 |
|
|
| 1,911,009 |
|
|
| 2,945,351 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these financial statements. | ||||||||||||||||
|
| December 31, |
|
| September 30, |
| ||||||
Balance Sheet Data: |
| 2019 |
|
| 2020 |
|
| 2021 |
| |||
|
|
|
|
|
| (unaudited) |
| |||||
Cash |
| $ | 1,991,802 |
|
| $ | 530,400 |
|
| $ | 12,603,341 |
|
Working capital (deficit) |
|
| 343,032 |
|
|
| (3,177,939 | ) |
|
| 12,490,555 |
|
Total assets |
|
| 2,065,803 |
|
|
| 605,453 |
|
|
| 13,685,787 |
|
Accounts payable and accrued liabilities |
|
| 992,224 |
|
|
| 237,753 |
|
|
| 1,195,232 |
|
Convertible subordinated promissory notes, net of discount |
|
| - |
|
|
| 1,848,495 |
|
|
| - |
|
Related party convertible subordinated promissory notes, net of discount |
|
| - |
|
|
| 1,140,984 |
|
|
| - |
|
Total liabilities |
|
| 2,275,962 |
|
|
| 3,783,392 |
|
|
| 1,195,232 |
|
Convertible preferred units |
|
| 21,556,395 |
|
|
| 21,825,145 |
|
|
| - |
|
Preferred warrant units |
|
| 723,431 |
|
|
| 723,431 |
|
|
|
|
|
Common units |
|
| 2,353,283 |
|
|
| 2,353,283 |
|
|
| - |
|
Common stock, par value |
|
| - |
|
|
| - |
|
|
| 833 |
|
Additional paid-in capital |
|
| - |
|
|
| - |
|
|
| 45,919,140 |
|
Accumulated deficit |
| $ | (24,843,268 | ) |
| $ | (28,079,798 | ) |
| $ | (33,429,418 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these financial statements. | ||||||||||||
| 9 |
| Table of Contents |
RISK FACTORS
Investing in our securities involves a high degree of risk. You should consider carefully the risks and uncertainties described below, together with all of the other information in this prospectus, including the consolidated financial statements, the notes thereto and the section entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included elsewhere in this prospectus before deciding whether to invest in our securities. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties that we are unaware of or that we deem immaterial may also become important factors that adversely affect our business. If any of the following risks actually occur, our business, financial condition, results of operations and future prospects could be materially and adversely affected. In that event, the market price of our common stock and/or Warrants could decline, and you could lose part or all of your investment.
Risks Related to Our Financial Position and Need for Capital
We are a clinical stage pharmaceutical company with a limited operating history.
We are a clinical-stage biopharmaceutical company with a limited operating history upon which you can evaluate our business and prospects. We must complete clinical studies and receive regulatory approval before commercial sales of a product can commence. The likelihood of success of our business plan must be considered in light of the problems, substantial expenses, difficulties, complications and delays frequently encountered in connection with developing and expanding early-stage businesses and the regulatory and competitive environment in which we operate. Pharmaceutical product development is a highly speculative undertaking, involves a substantial degree of risk and is a capital-intensive business.
Accordingly, you should consider our prospects in light of the costs, uncertainties, delays and difficulties frequently encountered by companies in the early stages of development, especially early stage clinical pharmaceutical companies such as ours. Potential investors should carefully consider the risks and uncertainties that a company with a limited operating history will face. In particular, potential investors should consider that we cannot assure you that we will be able to, among other things:
| • |
| successfully implement or execute our current business plan, and we cannot assure you that our business plan is sound; |
| • |
| successfully complete the clinical trials, non-clinical testing, and other requirements necessary to obtain regulatory approval for the marketing of our drug candidates, including DMT310 and DMT410; |
| • |
| successfully manufacture our clinical products and establish commercial drug supply; |
| • |
| secure, maintain and, as necessary, defend our intellectual property rights; |
| • |
| secure market exclusivity and/or adequate intellectual property protection for our drug candidates; |
| • |
| attract and retain an experienced management and advisory team; |
| • |
| secure acceptance of our drug candidates in the medical community and with third-party payors and consumers; |
| • |
| launch commercial sales of our drug candidates, whether alone or in collaboration with others; |
| • |
| comply with post-marketing regulatory requirements; |
| • |
| raise sufficient funds in the capital markets or otherwise to effectuate our business plan; and |
| • |
| utilize the funds that we do have and/or raise in this offering or in the future to efficiently execute our business strategy. |
If we cannot successfully execute any one of the foregoing, our business may fail and your investment will be adversely affected.
| 10 |
| Table of Contents |
We have incurred losses since inception and anticipate that we will continue to incur losses for the foreseeable future. We are not currently profitable, and we may never achieve or sustain profitability.
We have never generated revenue from operations, are unlikely to generate revenues for several years, and are currently operating at a loss and expect our operating costs will increase significantly as we incur costs related to preclinical development, the clinical trials for our drug candidates, and to operate as a public company. We expect to incur substantial expenses without corresponding revenues unless and until we are able to obtain regulatory approval and successfully commercialize any of our drug candidates. We may never be able to obtain regulatory approval for the marketing of our drug candidates in any indication in the United States or internationally. Even if we are able to commercialize our drug candidates, there can be no assurance that we will generate significant revenues or ever achieve profitability. We have incurred losses in each year since we commenced operations in December 2014. We incurred net losses of approximately $3.2 million and approximately $4.7 million for the years ended December 31, 2020 and 2019, respectively, and incurred net losses of approximately $5.3 million and $2.8 million for the nine months ended September 30, 2021 and 2020, respectively. As of December 31, 2020, we had an accumulated deficit of approximately $28.1 million, and as of September 30, 2021, we had an accumulated deficit of approximately $33.4 million. The size of our future net losses will depend, in part, on our future expenses and our ability to generate revenue, if any. Revenue from our current and potential future collaborations is uncertain because milestones or other contingent payments under our agreements may not be achieved or received.
As of December 31, 2021, we had capital resources consisting of cash of $10.8 million. We will continue to expend substantial cash resources for the foreseeable future for the clinical development of our product candidates and development of any other indications and product candidates we may choose to pursue. These expenditures will include costs associated with research and development, conducting preclinical studies and clinical trials, manufacturing and supply, as well as marketing and selling any products approved for sale. In particular, our Phase 3 clinical studies for our product candidates will require substantial funds to complete. Because the conduct and results of any clinical trial are highly uncertain, we cannot reasonably estimate the actual amounts necessary to successfully complete the development and commercialization of our current and any future product candidates.
We are uncertain when or if we will be able to achieve or sustain profitability. If we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods. Failure to become and remain profitable would impair our ability to sustain operations and adversely affect the price of our common stock or warrants and our ability to raise capital.
Even if this offering is successful, we will require additional capital to fund our operations, and if we fail to obtain necessary financing, we may not be able to complete the development and commercialization of our drugs.
We believe that the net proceeds from this offering and our existing cash, together with interest thereon, will be sufficient to fund our operations into February 2023. We have based these estimates, however, on assumptions that may prove to be wrong, and we could spend our available capital resources much faster than we currently expect or require more capital to fund our operations than we currently expect. Our currently anticipated expenditures for the development of our product candidates, DMT310 and DMT410, exceed the net proceeds from this offering and our existing cash. We will need to raise additional capital following this offering to fund our operations and continue to support our planned development and commercialization activities.
The amount and timing of our future funding requirements will depend on many factors, including:
| • |
| the timing, rate of progress and cost of any preclinical and clinical trials and other product development activities for our current and any future product candidates that we develop, in-license or acquire; |
| • |
| the results of the clinical trials for our product candidates in the United States and any foreign countries; |
| • |
| the timing of, and the costs involved in, FDA approval and any foreign regulatory approval of our product candidates, if at all; |
| • |
| the number and characteristics of any additional future product candidates we develop or acquire; |
| • |
| our ability to establish and maintain strategic collaborations, licensing, co-promotion or other arrangements and the terms and timing of such arrangements; |
| 11 |
| Table of Contents |
| • |
| the cost of commercialization activities if our current or any future product candidates are approved for sale, including manufacturing, marketing, sales and distribution costs; |
| • |
| the degree and rate of market acceptance of any approved products; |
| • |
| costs under our third-party manufacturing and supply arrangements for our current and any future product candidates and any products we commercialize; |
| • |
| costs and timing of completion of any additional outsourced commercial manufacturing or supply arrangements that we may establish; |
| • |
| costs of preparing, filing, prosecuting, maintaining, defending, and enforcing any patent claims and other intellectual property rights associated with our product candidates; |
| • |
| costs associated with prosecuting or defending any litigation that we are or may become involved in and any damages payable by us that result from such litigation; |
| • |
| costs associated with any product recall that could occur; |
| • |
| costs of operating as a public company; |
| • |
| the emergence, approval, availability, perceived advantages, relative cost, relative safety and relative efficacy of alternative and competing products or treatments; |
| • |
| costs associated with any acquisition or in-license of products and product candidates, technologies, or businesses; and |
| • |
| personnel, facilities, and equipment requirements. |
We cannot be certain that additional funding will be available on acceptable terms, or at all. In addition, future debt financing into which we enter may impose upon us covenants that restrict our operations, including limitations on our ability to incur liens or additional debt, pay dividends, redeem our stock, make certain investments and engage in certain merger, consolidation or asset sale transactions.
If we are unable to raise additional capital when required or on acceptable terms, we may be required to significantly delay, scale back or discontinue the development or commercialization of one or more of our product candidates, restrict our operations or obtain funds by entering into agreements on unattractive terms, which would likely have a material adverse effect on our business, stock price and our relationships with third parties with whom we have business relationships, at least until additional funding is obtained. If we do not have sufficient funds to continue operations, we could be required to seek bankruptcy protection or other alternatives that would likely result in our stockholders losing some or all of their investment in us. In addition, our ability to achieve profitability or to respond to competitive pressures would be significantly limited.
Raising additional capital may cause dilution to our stockholders, including purchasers of Units in this offering, restrict our operations or require us to relinquish rights to our technologies or product candidates.
Until such time, if ever, as we can generate substantial revenue, we may finance our cash needs through a combination of equity offerings, debt financings, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements or other sources. We do not currently have any committed external source of funds. In addition, we may seek additional capital due to favorable market conditions or strategic considerations, even if we believe that we have sufficient funds for our current or future operating plans.
To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect your rights as a common stockholder. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures, or declaring dividends. If we raise additional funds through collaborations, strategic alliances or marketing, distribution, or licensing arrangements with third parties, we may be required to relinquish valuable rights to our technologies, intellectual property, future revenue streams or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate product candidate development or future commercialization efforts.
| 12 |
| Table of Contents |
The reports of our independent registered public accounting firm for the fiscal years ended December 31, 2020 and 2019 contain an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern.
Due to the uncertainty of our ability to meet our current operating and capital expenses, in its report on our audited annual financial statements as of and for the years ended December 31, 2020 and December 31, 2019, our independent auditors included an explanatory paragraph regarding concerns about our ability to continue as a going concern. Substantial doubt about our ability to continue as a going concern may materially and adversely affect the price per share of our common stock and warrants and we may have a more difficult time obtaining financing. Further, the perception that we may be unable to continue as a going concern may impede our ability to raise additional funds or operate our business due to concerns regarding our ability to discharge our contractual obligations.
Changes in tax laws may materially adversely affect our business financial condition, results of operations and cash flows.
We are subject to tax laws, regulations and policies of the jurisdictions in which we do business, which may include U.S. federal, state, and local governments and taxing authorities in foreign jurisdictions. Changes in tax laws, as well as other factors, could cause us to experience fluctuations in our tax obligations and otherwise adversely affect our tax positions and/or our tax liabilities. The income tax rules in the jurisdictions in which we operate are constantly under review by taxing authorities and other governmental bodies. Changes to tax laws (which changes may have retroactive application) could adversely affect us or our stockholders. We are unable to predict what tax proposals may be proposed or enacted in the future or what effect such changes would have on our business, but such changes, to the extent they are brought into tax legislation, regulations, policies or practices, could affect our financial position and overall effective tax rates in the future in jurisdictions where we have operations, and increase the complexity, burden and cost of tax compliance.
Risks Related to Development, Regulatory Approval and Commercialization
A pandemic, epidemic, or outbreak of an infectious disease, such as COVID-19 could cause a disruption to the development of our product candidates.
Public health crises such as pandemics or similar outbreaks could adversely impact our business. In December 2019, a novel strain of a virus named SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2), or coronavirus, which causes COVID-19, surfaced in Wuhan, China and has since spread worldwide. The coronavirus pandemic is evolving, and to date has led to the implementation of various responses, including government-imposed quarantines, travel restrictions and other public health safety measures. The extent to which the coronavirus impacts our operations or those of our third-party partners, including our preclinical studies or clinical trial operations, will also depend on future developments, which are highly uncertain and cannot be predicted with confidence, including the duration of the outbreak, new information that will emerge concerning the severity of the coronavirus and the actions to contain the coronavirus or treat its impact, among others. The continued spread of COVID-19 globally could adversely impact our preclinical or clinical trial operations in the U.S. and abroad, including our ability to recruit and retain patients and principal investigators and site staff who, as healthcare providers, may have heightened exposure to COVID-19. For example, similar to other biopharmaceutical companies, we may experience delays in enrolling our current and/or planned clinical trials. COVID-19 may also affect employees of third-party CROs located in affected geographies that we rely upon to carry out our clinical trials. In addition, the patient populations that our lead and other core product candidates target may be particularly susceptible to COVID-19, which may make it more difficult for us to identify patients able to enroll in our future clinical trials and may impact the ability of enrolled patients to complete any such trials. Any negative impact COVID-19 has to patient enrollment or treatment or the execution of our product candidates could cause costly delays to clinical trial activities, which could adversely affect our ability to obtain regulatory approval for and to commercialize our product candidates, increase our operating expenses, and have a material adverse effect on our financial results.
| 13 |
| Table of Contents |
Additionally, timely enrollment in planned clinical trials is dependent upon clinical trial sites which could be adversely affected by global health matters, such as pandemics. We plan to conduct clinical trials for our product candidates in geographies which are currently being affected by the coronavirus. Some factors from the coronavirus outbreak that will delay or otherwise adversely affect enrollment in the clinical trials of our product candidates, as well as our business generally, include:
| • |
| the potential diversion of healthcare resources away from the conduct of clinical trials to focus on pandemic concerns, including the attention of physicians serving as our clinical trial investigators, hospitals serving as our clinical trial sites and hospital staff supporting the conduct of our prospective clinical trials; |
| • |
| limitations on travel that could interrupt key trial and business activities, such as clinical trial site initiations and monitoring, domestic and international travel by employees, contractors or patients to clinical trial sites, including any government-imposed travel restrictions or quarantines that will impact the ability or willingness of patients, employees or contractors to travel to our clinical trial sites or secure visas or entry permissions, a loss of face-to-face meetings and other interactions with potential partners, any of which could delay or adversely impact the conduct or progress of our prospective clinical trials; |
| • |
| the potential negative effect on the operations of our third-party manufacturers. |
| • |
| interruption in global shipping, affecting the transport of raw materials for our products, clinical trial materials, such as patient samples, investigational drug product and conditioning drugs and other supplies used in our prospective clinical trials; and |
| • |
| business disruptions caused by potential workplace, laboratory and office closures and an increased reliance on employees working from home, disruptions to or delays in ongoing laboratory experiments and operations, staffing shortages, travel limitations or mass transit disruptions, any of which could adversely impact our business operations or delay necessary interactions with local regulators, ethics committees and other important agencies and contractors. |
We have taken temporary precautionary measures intended to help minimize the risk of the virus to our employees, including having all of our employees to work remotely, suspending all non-essential travel worldwide for our employees and discouraging employee attendance at industry events and in-person work-related meetings, which could negatively affect our business. We cannot presently predict the scope and severity of the planned and potential shutdowns or disruptions of businesses and government agencies, such as the Securities and Exchange Commission, or the SEC, or FDA.
Our business is dependent on the successful development, regulatory approval and commercialization of our product candidates, in particular DMT310.
Our portfolio of product candidates includes one mid-stage product candidate, DMT310, a once-weekly topical, naturally-derived product candidate for the treatment of acne, rosacea and psoriasis, and an early-stage candidate, DMT410, a combination treatment regimen to aid in the topical delivery of botulinum toxin for the treatment of hyperhidrosis and aesthetic skin conditions. The success of our business, including our ability to finance our company and generate any revenue in the future, will primarily depend on the successful development, regulatory approval and commercialization or partnering of our product candidates. In the future, we may also become dependent on just one of our product candidates or any future product candidates that we may in-license, acquire or develop. The clinical and commercial success of our product candidates will depend on a number of factors, including the following:
| • |
| the ability to raise additional capital on acceptable terms, or at all; |
| • |
| timely completion of our clinical trials, which may be significantly slower or cost more than we currently anticipate and will depend substantially upon the performance of third-party contractors; |
| • |
| whether we are required by the FDA, or similar foreign regulatory agencies to conduct additional clinical trials beyond those planned to support the approval and commercialization of our product candidates or any future product candidates; |
| 14 |
| Table of Contents |
| • |
| acceptance of our proposed indications and primary endpoint assessments relating to the proposed indications of our product candidates by the FDA and similar foreign regulatory authorities; |
| • |
| our ability to demonstrate to the satisfaction of the FDA and similar foreign regulatory authorities, the safety and efficacy of our product candidates or any future product candidates; |
| • |
| our ability to develop a suitable drug product release assay; |
| • |
| our ability to identify an active compound within the drug product that can be detected in a pharmacokinetics study; |
| • |
| the prevalence, duration and severity of potential side effects experienced in connection with our product candidates or future approved products, if any; |
| • |
| the timely receipt of necessary marketing approvals from the FDA and similar foreign regulatory authorities; |
| • |
| achieving and maintaining, and, where applicable, ensuring that our third-party contractors achieve and maintain, compliance with our contractual obligations and with all regulatory requirements applicable to our product candidates or any future product candidates or approved products, if any; |
| • |
| the ability of third parties with whom we contract to manufacture clinical trial and commercial supplies of our product candidates or any future product candidates, remain in good standing with regulatory agencies and develop, validate and maintain commercially viable manufacturing processes that are compliant with current good manufacturing practices, or cGMP, or good agricultural and collection practices, or GACP; |
| • |
| a continued acceptable safety profile during clinical development and following approval of our product candidates or any future product candidates; |
| • |
| our ability to successfully commercialize our product candidates or any future product candidates in the United States and internationally, if approved for marketing, sale and distribution in such countries and territories, whether alone or in collaboration with others; |
| • |
| acceptance by physicians, patients and payors of the benefits, safety and efficacy of our product candidates or any future product candidates, if approved, including relative to alternative and competing treatments; |
| • |
| our ability to comply with numerous post-approval regulatory requirements; |
| • |
| our and our partners’ ability to establish and enforce intellectual property rights in and to our product candidates or any future product candidates; |
| • |
| our and our partners’ ability to avoid third-party patent interference or intellectual property infringement claims; and |
| • |
| our ability to in-license or acquire additional product candidates or commercial-stage products that we believe we can successfully develop and commercialize. |
If we are unable to achieve one or more of the above factors, many of which are beyond our control, in a timely manner or at all, we could experience significant delays and increased costs or an inability to obtain regulatory approvals or commercialize our product candidates. Even if regulatory approvals are obtained, we may never be able to successfully commercialize any of our product candidates. Accordingly, we cannot assure you that we will be able to generate sufficient revenue through the sale of our product candidates or any future product candidates to continue operations.
| 15 |
| Table of Contents |
According to the FDA Guidelines, we may be required to complete a 90-day dermal minipig study and a standard dermal pharmacokinetic study prior to holding an End of Phase 2 meeting with the agency. The minipig and pharmacokinetic studies can be complicated, time consuming, and expensive and we may be unsuccessful in completing the required studies, which could prevent or delay the Phase 3 program and regulatory approval and commercialization.
Part of required drug development for topical products is completion of a 90-day dermal minipig study and a standard dermal pharmacokinetic development plan prior to holding an End of Phase 2 meeting with the FDA. This plan can be difficult to design and conduct and its outcome is uncertain. As part of this dermal pharmacokinetic development plan, we need to develop and validate an analytical method to detect an analyte in rat, minipig and human plasma, which will be difficult, time consuming and expensive. While we have identified an analyte that is present in DMT310, we have yet to develop a method with sufficient sensitivity to detect the analyte at the required levels. If we are delayed or unable to develop such method, we may be required to identify a new analyte of DMT310 to use in the pharmacokinetic development plan. Even if we are able to successfully develop a bioanalytical method to detect the identified analyte, we still must validate the bioanalytical method under good laboratory practice, or GLP, conditions. There is no guarantee that we will be able to do so successfully. If we are unable to validate such a bioanalytical method then we may be required to develop a new bioanalytical method or identify a new analyte for the pharmacokinetic development plan, which could prevent or delay regulatory approval and commercialization.
Prior to holding an End of Phase 2 meeting with the FDA and after validating the bioanalytical method under GLP conditions, we may be required to conduct a 90-day dermal minipig study and a maximum use pharmacokinetic study in humans, the outcomes of which are unknown and difficult to predict. Due to delays and constraints in the global supply chain for the minipigs to be used in the 90-day dermal minipig study, we may be forced to delay the start of such study. While we believe we may have to complete this study at some time during our clinical development, because DMT310 has been used in completed clinical trials of over 170 human patients in which it exhibited an acceptable safety and tolerability profile, we have recently submitted requests to the FDA to waive conduct of a 90-day dermal minipig study and standard dermal pharmacokinetic study prior to holding an End of Phase 2 meeting with the FDA. Although we have submitted these requests to the FDA, the FDA may not grant one or both waiver requests. This decision by the FDA could result in additional development costs and a delay in our development timeline, in which case our business could be materially harmed. Additionally, if the outcomes of these studies are not satisfactory to the FDA, we may be required to conduct the entire pharmacokinetic development plan again, which could result in additional development costs and a delay in our development timeline, in which case our business could be materially harmed.
Clinical drug development for our product candidates is very expensive, time-consuming and uncertain. Our clinical trials may fail to adequately demonstrate the safety and efficacy of our product candidates, which could prevent or delay regulatory approval and commercialization.
Clinical drug development for our product candidates is very expensive, time-consuming, difficult to design and implement and its outcome is inherently uncertain. Before obtaining regulatory approval for the commercial sale of a product candidate, we must demonstrate through clinical trials that a product candidate is both safe and effective for use in the target indication, which is impossible to predict. Most product candidates that commence clinical trials are never approved by regulatory authorities for commercialization. Our product candidates are in various stages of development and a failure of one more clinical trials can occur at any stage of testing or at any time during the trial process. We expect that clinical trials for these product candidates will continue for several years, but may take significantly longer than expected to complete.
We have not completed all clinical trials for the approval of any of our product candidates. We have yet to hold an End of Phase 2 meeting with the FDA and do not know what feedback they will provide us. In previous communications with the FDA they had asked us to show that hydrogen peroxide was not an active ingredient in our DMT310 product. If we do not supply sufficient evidence and data to convince the FDA that hydrogen peroxide is not an active ingredient and merely a fluidizing agent (hydrogen peroxide), then we may have to alter our clinical plans or reformulate our product based on FDA feedback. If we chose to reformulate our lead product, DMT310, then we may decide to redo our Phase 2 studies, which would be time consuming and expensive and there is no certainty of success.
| 16 |
| Table of Contents |
We may experience delays in ongoing and future clinical trials for our product candidates and do not know if future clinical trials, if any, will begin on time, need to be redesigned, enroll adequate number of patients on time or be completed on schedule, if at all. In addition, we, any partner with which we currently or may in the future collaborate, the FDA, an IRB or other regulatory authorities, including state and local agencies and counterpart agencies in foreign countries, may suspend, delay, require modifications to or terminate our clinical trials at any time, for various reasons, including:
| • |
| discovery of safety or tolerability concerns, such as serious or unexpected toxicities or side effects or exposure to otherwise unacceptable health risks, experienced by study participants or other safety issues; |
| • |
| lack of effectiveness of any product candidate during clinical trials or the failure of our product candidates to meet specified endpoints; |
| • |
| slower than expected rates of subject recruitment and enrollment rates or inability to enroll a sufficient number of patients in clinical trials resulting from numerous factors, including the prevalence of other companies’ clinical trials for their product candidates for the same indication, or clinical trials for indications for which patients do not as commonly seek treatment; |
| • |
| delays or difficulties in our clinical trials due to quarantines or other restrictions resulting from the COVID-19 pandemic; |
| • |
| difficulty in retaining subjects who have initiated a clinical trial but may withdraw at any time due to adverse side effects from the therapy, insufficient efficacy, fatigue with the clinical trial process or for any other reason; |
| • |
| difficulty in obtaining IRB approval for studies to be conducted at each clinical trial site; |
| • |
| delays in manufacturing or obtaining, or inability to manufacture or obtain, sufficient quantities of materials for use in clinical trials; |
| • |
| difficulty or inability to find a partner that will allow us to test their product for our DMT410 program; |
| • |
| inadequacy of or changes in our manufacturing process or the product formulation or method of delivery; |
| • |
| changes in applicable laws, regulations and regulatory policies; |
| • |
| delays or failure in reaching agreement on acceptable terms in clinical trial contracts or protocols with prospective CROs, clinical trial sites and other third-party contractors; |
| • |
| inability to add a sufficient number of clinical trial sites; |
| • |
| uncertainty regarding proper formulation and dosing; |
| • |
| failure by us, our employees, our CROs or their employees or other third-party contractors to comply with contractual and applicable regulatory requirements or to perform their services in a timely or acceptable manner; |
| • |
| failure by us, our employees, our CROs or their employees or any partner with which we may collaborate or their employees to comply with applicable FDA or other regulatory requirements relating to the conduct of clinical trials or the handling, storage, security and recordkeeping for drug and biologic products; |
| • |
| scheduling conflicts with participating clinicians and clinical institutions; |
| • |
| failure to design appropriate clinical trial protocols; |
| • |
| insufficient data to support regulatory approval; |
| • |
| inability or unwillingness of medical investigators to follow our clinical protocols; or |
| • |
| difficulty in maintaining contact with subjects during or after treatment, which may result in incomplete data. |
| 17 |
| Table of Contents |
In the case of our topical product candidates, we are seeking to deliver sufficient concentrations of the active pharmaceutical ingredient, or API, through the skin barrier to the targeted dermal tissue to achieve the intended therapeutic effect. As a result, safety and efficacy can be difficult to establish. The topical route of administration may involve new formulations and dosage forms, which can be difficult to develop and manufacture and may raise novel regulatory issues and result in development or review delays. For example, the API for DMT310 is a milled sponge powder, and we are not aware of previous FDA approvals of sponges as a prescription drug.
We or any partner with which we may collaborate may suffer significant setbacks in our clinical trials similar to the experience of a number of other companies in the pharmaceutical and biotechnology industries, even after receiving promising results in earlier trials. In the event that we or our potential partners abandon or are delayed in the clinical development efforts related to our product candidates, we may not be able to execute on our business plan effectively and our business, financial condition, operating results and prospects would be harmed.
Changes in methods of product candidate manufacturing or formulation may result in additional costs or delay.
As product candidates proceed through preclinical studies to late-stage clinical trials towards potential approval and commercialization, it is common that various aspects of the development program, such as manufacturing methods and formulation, are altered along the way in an effort to optimize processes and results. Such changes carry the risk that they will not achieve these intended objectives. Any of these changes could cause our product candidates to perform differently and affect the results of planned clinical trials or other future clinical trials conducted with the altered materials. Such changes may also require additional testing, FDA notification or FDA approval. This could delay completion of clinical trials, require the conduct of bridging clinical trials or the repetition of one or more clinical trials.
We may be unable to obtain regulatory approval for DMT310 or DMT410, or our early-stage product candidates under applicable regulatory requirements. The FDA and foreign regulatory bodies have substantial discretion in the approval process, including the ability to delay, limit or deny approval of product candidates. The delay, limitation or denial of any regulatory approval would adversely impact commercialization, our potential to generate revenue, our business and our operating results.
We currently have no products approved for sale, and we may never obtain regulatory approval to commercialize any of our current or future product candidates. The research, testing, manufacturing, safety surveillance, efficacy, quality control, recordkeeping, labeling, packaging, storage, approval, sale, marketing, distribution, import, export, and reporting of safety and other post-market information related to our drug products are subject to extensive regulation by the FDA and other regulatory authorities in the United States and in foreign countries, and such regulations differ from country to country. We are not permitted to market any of our current product candidates in the United States until we receive approval of a new drug application, or NDA, or other applicable regulatory filing from the FDA. We are also not permitted to market any of our current product candidates in any foreign countries until we or our partners receive the requisite approval from the applicable regulatory authorities of such countries. To gain approval to market a new drug such as DMT310 or DMT410, the FDA and/or foreign regulatory authorities must receive, among other things, preclinical and clinical data that adequately demonstrate the safety, purity, potency, efficacy, and compliant manufacturing of the drug product for the intended indication applied for in an NDA, or other applicable regulatory filing. The development and approval of a product derived from a natural source and new drug products involves a long, expensive and uncertain process, and delay or failure can occur at any stage. A number of companies in the pharmaceutical and biopharmaceutical industry have suffered significant setbacks in nonclinical development, clinical trials, including in Phase 3 clinical development, even after promising results in earlier preclinical studies or clinical trials. These setbacks have been caused by, among other things, findings made while clinical trials were underway and safety or efficacy observations made in clinical trials, including previously unreported adverse events. Success in clinical trials does not ensure that later clinical trials will be successful, or that nonclinical studies will be successful. The results of clinical trials by other parties may not be indicative of the results in trials we or our partners may conduct. For example, for DMT310, the results of our Phase 2a and Phase 2b clinical trials may not accurately predict results in Phase 3 clinical trials that will have larger numbers of patients. Nor will the human safety data collected from our Phase 2a and Phase 2b clinical trial predict the outcome of our pharmacokinetic plan.
| 18 |
| Table of Contents |
The FDA and foreign regulatory bodies have substantial discretion in the drug approval process, including the ability to delay, limit or deny approval of product candidates for many reasons. The FDA or the applicable foreign regulatory body may:
| • |
| disagree with the design or implementation of one or more clinical trials; |
| • |
| not deem a product candidate safe and effective for its proposed indication, or may deem a product candidate’s safety or other perceived risks to outweigh its clinical or other benefits; |
| • |
| not find the data from preclinical studies and clinical trials sufficient to support approval, or the results of clinical trials may not meet the level of statistical or clinical significance required by the FDA or the applicable foreign regulatory body for approval; |
| • |
| disagree with our interpretation of data from preclinical studies or clinical trials performed by us or third parties, or with the interpretation of any partner with which we may collaborate; |
| • |
| determine the data collected from clinical trials may not be sufficient to support the submission of an NDA, or other applicable regulatory filing; |
| • |
| require additional preclinical studies or clinical trials; |
| • |
| identify deficiencies in the formulation, quality control, labeling or specifications of our current or future product candidates; |
| • |
| require clinical trials in pediatric patients in order to establish pharmacokinetics or safety for this more drug-sensitive population; |
| • |
| grant approval contingent on the performance of costly additional post-approval clinical trials; |
| • |
| approve our current or any future product candidates for a more limited indication or a narrower patient population than we originally requested or with strong warnings that may affect marketability; |
| • |
| not approve the labeling that we believe is necessary or desirable for the successful commercialization of our product candidates; |
| • |
| not approve of the manufacturing processes, controls or facilities of third-party manufacturers or testing labs with which we contract; |
| • |
| consider our products a device instead of a drug requiring a different approval process and manufacturing needs; |
| • |
| consider one of our products a combination product instead of a singular drug requiring additional clinical trials or increased number of patients per study, or |
| • |
| change its approval policies or adopt new regulations in a manner rendering our clinical data or regulatory filings insufficient for approval. |
There have been only two products approved by the FDA under the botanical guidance and none in the indication for acne vulgaris with both approved products’ active ingredient coming from an extract of a plant. While freshwater sponges, such as Spongilla, are technically animals, because we are using the entirety of the sponge for the DMT310 drug product, the FDA has allowed us to reference the botanical guidance for raw material quality control relating to the manufacturing of the drug product. We do not know how any other regulatory authority will treat DMT310 for their approval process. In addition, the FDA or other regulatory authorities may change their policies, issue additional regulations or revise existing regulations or take other actions, which may prevent or delay approval of our future products under development on a timely basis. Such policy or regulatory changes could impose additional requirements upon us that could delay our ability to obtain approvals, increase the costs of compliance or restrict our ability to maintain any marketing authorizations we may have obtained.
Any delay, limitation or denial in any applicable regulatory approval for any of our product candidates would delay or adversely impact commercialization of our product candidates and would harm our business, financial condition, operating results and prospects.
| 19 |
| Table of Contents |
We are preparing to conduct our first Phase 3 clinical trials and may be unable to successfully complete it or any future clinical trials.
The conduct of a Phase 3 clinical trial is a complicated process. Although members of our management team have conducted Phase 3 clinical trials in the past while employed at other companies, we as a company have not conducted a Phase 3 clinical trial before, and as a result may require more time and incur greater costs than we anticipate. Failure to include the correct treatment regimen, complete, or delays in, our Phase 3 clinical trials, could prevent us from or delay us in commencing future clinical trials for DMT310, obtaining regulatory approval of and commercializing our product candidates, which would adversely impact our financial performance. In addition, some of our competitors are currently conducting clinical trials for product candidates that treat the same indications as DMT310, and patients who are otherwise eligible for our clinical trials may instead enroll in clinical trials of our competitors’ product candidates.
Patient enrollment is affected by other factors including:
| • |
| the severity of the disease under investigation; |
| • |
| the eligibility criteria for the study in question; |
| • |
| the perceived risks and benefits of the product candidate under study; |
| • |
| the efforts to facilitate timely enrollment in clinical trials; |
| • |
| the patient referral practices of physicians; |
| • |
| the ability to monitor patients adequately during and after treatment; |
| • |
| the proximity and availability of clinical trial sites for prospective patients; and |
| • |
| factors we may not be able to control, such as potential pandemics that may limit subjects, principal investigators or staff or clinical site availability (e.g., the outbreak of COVID-19). |
Even if our current product candidates or any future product candidates obtain regulatory approval, they may fail to achieve the broad degree of physician and patient adoption and use necessary for commercial success.
The commercial success of any of our current or future product candidates, if approved, will depend significantly on the broad adoption and use of the resulting product by physicians, patients and payors for approved indications, and may not be commercially successful. The degree and rate of adoption of our current or future product candidates, if approved, will depend on a number of factors, including:
| • |
| the clinical indications for which the product is approved and patient demand for approved products that treat those indications; |
| • |
| the effectiveness of our product as compared to other available therapies; |
| • |
| the availability of coverage and adequate reimbursement from managed care plans and other healthcare payors for any of our product candidates that may be approved; |
| • |
| the cost of treatment with our product candidates in relation to alternative treatments and willingness to pay for the product, if approved, on the part of patients; |
| • |
| acceptance by physicians, major operators of clinics and patients of the product as a safe and effective treatment; |
| • |
| physician and patient willingness to adopt a new therapy, including for DMT310, a sponge product, over other available therapies to treat approved indications; |
| • |
| patients’ perception of a product derived from a freshwater sponge as one for which will provide medical treatment; |
| • |
| overcoming any biases physicians or patients may have toward particular therapies for the treatment of approved indications; |
| 20 |
| Table of Contents |
| • |
| proper training and administration of our product candidates by physicians and medical staff; |
| • |
| patient satisfaction with the results and administration of our product candidates and overall treatment experience; |
| • |
| the willingness of patients to pay for certain of our product candidates relative to other discretionary items, especially during economically challenging times; |
| • |
| the revenue and profitability that our product candidate may offer a physician as compared to alternative therapies; |
| • |
| the prevalence and severity of side effects; |
| • |
| limitations or warnings contained in the FDA-approved labeling for our product candidates; |
| • |
| any FDA requirement to undertake a risk evaluation and mitigation strategy, or REMS; |
| • |
| the effectiveness of our sales, marketing and distribution efforts; |
| • |
| our ability to maintain sufficient quantities of supply to meet demand; |
| • |
| adverse publicity about our product candidates or favorable publicity about competitive products; and |
| • |
| potential product liability claims. |
If any of our current or future product candidates are approved for use but fail to achieve the broad degree of physician and patient adoption necessary for commercial success, our operating results and financial condition will be adversely affected, which may delay, prevent or limit our ability to generate revenue and continue our business.
We intend to seek NCE exclusivity for DMT310 and future product candidates, and we may be unsuccessful in obtaining such exclusivity.
As part of our business strategy, we intend to seek new chemical entity, or NCE, exclusivity for DMT310 or future product candidates. In the United States, a pharmaceutical manufacturer may obtain five years of non-patent exclusivity upon NDA approval of an NCE which is a drug that contains an active moiety that has not been approved by the FDA in any other NDA. An “active moiety” is defined as the molecule or ion responsible for the drug substance’s physiological or pharmacologic action. During the five-year exclusivity period, the FDA cannot accept for filing any ANDA seeking approval of a generic version of that drug or any 505(b)(2) NDA for the same active moiety and that relies on the FDA’s findings regarding that drug, except that FDA may accept an application for filing after four years if the follow-on applicant makes a paragraph IV certification. This exclusivity period may be extended by an additional six months if certain requirements are met to qualify the product for pediatric exclusivity, including the receipt of a written request from the FDA that we conduct certain pediatric studies, the submission of study reports from such studies to the FDA after receipt of the written request and satisfaction of the conditions specified in the written request. We believe that DMT310 constitutes an NCE and should be eligible for NCE exclusivity. However, we may be unable to successfully obtain such exclusivity, and if any of our competitors obtains FDA approval of an NDA for a similar drug product before we do, they, and not us, may be eligible for NCE exclusivity. If we do not obtain NCE exclusivity for DMT310, or if a competitor obtains NCE exclusivity for a similar product before we submit and receive approval of an NDA for DMT310, our ability to commence sales and generate revenue would be adversely affected.
| 21 |
| Table of Contents |
Our product candidates, if approved, will face significant competition and our failure to effectively compete may prevent us from achieving significant market penetration.
The pharmaceutical industry is characterized by rapidly advancing technologies, intense competition, and a strong emphasis on developing proprietary therapeutics. Numerous pharmaceutical companies, generic drug companies, biotechnology companies, cosmetic companies, and academic and research institutions are engaged in the development, patenting, manufacturing, and marketing of health care products competitive with those that we are developing, including VYNE Therapeutics, Cassiopea, Sol-Gel, Arcutis Biotherapeutics, Amgen, AbbVie, Bristol Meyers Squib, Lilly, Nestle, Pfizer, and others. Many of our competitors have greater financial resources, marketing capabilities, sales forces, manufacturing capabilities, research and development capabilities, clinical trial expertise, intellectual property portfolios, experience in obtaining patents and regulatory approvals for product candidates and other resources than us. Some of the companies that offer competing products also have a broad range of other product offerings, large direct sales forces and long-term customer relationships with our target physicians, which could inhibit our market penetration efforts. In addition, certain of our product candidates, if approved, may compete with other dermatological products, including over-the-counter treatments, for a share of some patients’ discretionary budgets and for physicians’ attention within their clinical practices.
We anticipate that, if we obtain regulatory approval of our product candidates, we will face significant competition from other approved therapies. If approved, our product candidates may also compete with unregulated, unapproved, off-label, and over the counter treatments. Certain of our product candidates, if approved, will present novel therapeutic approaches for the approved indications and will have to compete with existing therapies, some of which are widely known and accepted by physicians and patients. To compete successfully in this market, we will have to demonstrate that the relative cost, safety and efficacy of our approved products, if any, provide an attractive alternative to existing and other new therapies. Such competition could lead to reduced market share for our product candidates and contribute to downward pressure on the pricing of our product candidates, which could harm our business, financial condition, operating results and prospects. For more information about the competition we face, see “Business-Competition.”
Due to less stringent regulatory requirements in certain foreign countries, there are many more dermatological products and procedures available for use in those international markets than are approved for use in the United States. In certain international markets, there are also fewer limitations on the claims that our competitors can make about the effectiveness of their products and the manner in which they can market them. As a result, we expect to face more competition in these markets than in the United States.
We expect to face generic or similar type of product competition for our product candidates, which could adversely affect our business, financial condition, operating results and prospects.
Upon the expiration or loss of any patent protection for any of our product candidates that are approved, or upon the “at-risk” launch, despite pending patent infringement litigation against the generic product or its equivalent, by a generic competitor of a generic version of any of our product candidates that are approved, which may be sold at significantly lower prices than our approved product candidates, we could lose a significant portion of sales of that product in a short period of time, which would adversely affect our business, financial condition, operating results and prospects.
It is unknown how the FDA or any regulatory authority will view an attempted generic version of DMT310 because it is a derived from a natural material that refers to principles of the botanical guidance. There are no currently approved generic versions of a natural product on the market and no FDA guidelines on the approval process for a generic version of a natural product. Therefore, it is unknown how difficult it will be for a generic version of a natural product to be approved for commercial sale in the United States. It is unclear whether the FDA will view spongilla lacustris or a similar sponge species that is harvested from a different location than DMT310 raw material is harvested as identical to DMT310 raw material and therefore could follow the generic pathway to approval.
Any product candidates that we commercialize, or that any partner with which we may collaborate commercializes, will be subject to ongoing and continued regulatory review.
Even after we or our partners achieve U.S. regulatory approval for a product candidate, if any, we or our partners will be subject to continued regulatory review and compliance obligations. For example, with respect to our product candidates, the FDA may impose significant restrictions on the approved indicated uses for which the product may be marketed or on the conditions of approval. A product candidate’s approval may contain requirements for potentially costly post-approval studies and surveillance, including Phase 4 clinical trials or a REMS, to monitor the safety and efficacy of the product. We will also be subject to ongoing FDA obligations and continued regulatory review with respect to, among other things, the manufacturing, processing, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and recordkeeping for our product candidates. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance with cGMP requirements, with the FDA’s good clinical practice, or GCP, or good agricultural and collections practices, or GACP, requirements and good laboratory practice, or GLP, requirements, which are regulations and guidelines enforced by the FDA for all of our product candidates in clinical and preclinical development, and for any clinical trials that we conduct post-approval. To the extent that a product candidate is approved for sale in other countries, we may be subject to similar restrictions and requirements imposed by laws and government regulators in those countries.
| 22 |
| Table of Contents |
If we, our partners, our product candidates or the manufacturing facilities for our product candidates fail to comply with applicable regulatory requirements, a regulatory agency may:
| • |
| impose restrictions on the marketing or manufacturing of the product, suspend or withdraw product approvals or revoke necessary licenses; |
| • |
| mandate modifications to promotional materials or require us to provide corrective information to healthcare practitioners; |
| • |
| require us or our partners to enter into a consent decree, which can include imposition of various fines, reimbursements for inspection costs, required due dates for specific actions and penalties for noncompliance; |
| • |
| issue warning letters, show cause notices or untitled letters describing alleged violations, which may be publicly available; |
| • |
| commence criminal investigations and prosecutions; |
| • |
| impose injunctions, suspensions or revocations of necessary approvals or other licenses; |
| • |
| impose other civil or criminal penalties; |
| • |
| suspend any ongoing clinical trials; |
| • |
| delay or refuse to approve pending applications or supplements to approved applications filed by us or our potential partners; |
| • |
| refuse to permit drugs or precursor chemicals to be imported or exported to or from the United States; |
| • |
| suspend or impose restrictions on operations, including costly new manufacturing requirements; or |
| • |
| seize or detain products or require us or our partners to initiate a product recall. |
The regulations, policies or guidance of the FDA and other applicable government agencies may change and new or additional statutes or government regulations may be enacted that could prevent or delay regulatory approval of our product candidates or further restrict or regulate post-approval activities. We cannot predict the likelihood, nature or extent of adverse government regulation that may arise from future legislation or administrative action, either in the United States or abroad. If we are not able to achieve and maintain regulatory compliance, we may not be permitted to market our product candidates, which would adversely affect our ability to generate revenue and achieve or maintain profitability.
We may in the future conduct clinical trials for our product candidates outside the United States and the FDA and applicable foreign regulatory authorities may not accept data from such trials.
We may in the future choose to conduct one or more of our clinical trials outside the United States, including in Canada, Europe and South America. Although the FDA or applicable foreign regulatory authority may accept data from clinical trials conducted outside the United States or the applicable jurisdiction, acceptance of such study data by the FDA or applicable foreign regulatory authority may be subject to certain conditions. Where data from foreign clinical trials are intended to serve as the basis for marketing approval in the United States, the FDA will not approve the application on the basis of foreign data alone unless those data are applicable to the U.S. population and U.S. medical practice; the studies were performed by clinical investigators of recognized competence; and the data are considered valid without the need for an on-site inspection by the FDA or, if the FDA considers such an inspection to be necessary, the FDA is able to validate the data through an on-site inspection or other appropriate means. Many foreign regulatory bodies have similar requirements. In addition, such foreign studies would be subject to the applicable local laws of the foreign jurisdictions where the studies are conducted. There can be no assurance the FDA or applicable foreign regulatory authority will accept data from trials conducted outside of the United States or the applicable jurisdiction. If the FDA or applicable foreign regulatory authority does not accept such data, it would likely result in the need for additional trials, which would be costly and time-consuming and delay aspects of our business plan.
| 23 |
| Table of Contents |
Our product candidates may cause undesirable side effects or have other unexpected properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label or result in post-approval regulatory action.
Unforeseen side effects from any of our product candidates could arise either during clinical development or, if approved, after the approved product has been marketed. Undesirable side effects caused by product candidates could cause us, any partners with which we may collaborate or regulatory authorities to interrupt, modify, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval by the FDA or comparable foreign authorities. Results of clinical trials could reveal a high and unacceptable severity and prevalence of side effects. In such an event, trials could be suspended or terminated and the FDA or comparable foreign regulatory authorities could order us, or our potential partners, to cease further development of or deny approval of product candidates for any or all targeted indications. The drug-related side effects could affect patient recruitment or the ability of enrolled patients to complete the trial or result in product liability claims. Any of these occurrences may harm our business, financial condition, operating results and prospects.
Additionally, if we or others identify undesirable side effects, or other previously unknown problems, caused by our product candidates after obtaining U.S. or foreign regulatory approval or other products with the same or related active ingredients, a number of potentially negative consequences could result, including:
| • |
| regulatory authorities may withdraw their approval of the product; |
| • |
| regulatory authorities may require a recall of the product or we or our potential partners may voluntarily recall a product; |
| • |
| regulatory authorities may require the addition of warnings or contraindications in the product labeling, narrowing of the indication in the product label or field alerts to physicians and pharmacies; |
| • |
| we may be required to create a medication guide outlining the risks of such side effects for distribution to patients or institute a REMS; |
| • |
| we may have limitations on how we promote the product; |
| • |
| we may be required to change the way the product is administered or modify the product in some other way; the FDA or applicable foreign regulatory authority may require additional clinical trials or costly post-marketing testing and surveillance to monitor the safety or efficacy of the product; |
| • |
| the FDA or applicable foreign regulatory authority may require additional clinical trials or costly post-marketing testing and surveillance to monitor the safety or efficacy of the product |
| • |
| sales of the product may decrease significantly; |
| • |
| we could be sued and held liable for harm caused to patients; and |
| • |
| our brand and reputation may suffer. |
Any of the above events resulting from undesirable side effects or other previously unknown problems could prevent us or our potential partners from achieving or maintaining market acceptance of the affected product candidate and could substantially increase the costs of commercializing our product candidates.
| 24 |
| Table of Contents |
We may face product liability exposure, and if successful claims are brought against us, we may incur substantial liability if our insurance coverage for those claims is inadequate.
We face an inherent risk of product liability as a result of the clinical testing of our product candidates and will face an even greater risk if we commercialize any products. This risk exists even if a product is approved for commercial sale by the FDA and manufactured in facilities licensed and regulated by the FDA or an applicable foreign regulatory authority. Our products and product candidates are designed to affect important bodily functions and processes. Any side effects, manufacturing defects, misuse or abuse associated with our product candidates could result in injury to a patient or even death. We cannot offer any assurance that we will not face product liability suits in the future, nor can we assure you that our insurance coverage will be sufficient to cover our liability under any such cases.
In addition, a liability claim may be brought against us even if our product candidates merely appear to have caused an injury. Product liability claims may be brought against us by consumers, health care providers, pharmaceutical companies or others selling or otherwise coming into contact with our product candidates, among others. If we cannot successfully defend ourselves against product liability claims we will incur substantial liabilities and reputational harm. In addition, regardless of merit or eventual outcome, product liability claims may result in:
| • |
| withdrawal of clinical trial participants; |
| • |
| termination of clinical trial sites or entire trial programs; |
| • |
| inability to gain regulatory approval of our product candidates; |
| • |
| the inability to commercialize our product candidates; |
| • |
| decreased demand for our product candidates; |
| • |
| impairment of our business reputation; |
| • |
| product recall or withdrawal from the market or labeling, marketing or promotional restrictions; |
| • |
| substantial costs of any related litigation or similar disputes; |
| • |
| distraction of management’s attention and other resources from our primary business; |
| • |
| substantial monetary awards to patients or other claimants against us that may not be covered by insurance; or |
| • |
| loss of revenue. |
We currently maintain product liability insurance coverage, which may not be sufficient to cover all of our product liability related expenses or losses and may not cover us for any expenses or losses we may suffer. Moreover, insurance coverage is becoming increasingly expensive, and, in the future, we may not be able to maintain insurance coverage at a reasonable cost, in sufficient amounts or upon adequate terms to protect us against losses due to product liability. We will need to increase our product liability coverage if any of our product candidates receive regulatory approval, which will be costly, and we may be unable to obtain this increased product liability insurance on commercially reasonable terms, or at all. A successful product liability claim or series of claims brought against us could cause our stock price to decline and, if judgments exceed our insurance coverage, could decrease our cash and could harm our business, financial condition, operating results and prospects.
| 25 |
| Table of Contents |
If any of our product candidates are approved for marketing and we are found to have improperly promoted off-label uses, or if physicians misuse our products or use our products off-label, we may become subject to prohibitions on the sale or marketing of our products, product liability claims and significant fines, penalties and sanctions, and our brand and reputation could be harmed.
The FDA and other regulatory agencies strictly regulate the marketing and promotional claims that are made about drug and biologic products. In particular, a product may not be promoted for uses or indications that are not approved by the FDA or such other regulatory agencies as reflected in the product’s approved labeling and comparative safety or efficacy claims cannot be made without direct comparative clinical data. If we are found to have promoted off-label uses of any of our product candidates, we may receive warning or untitled letters and become subject to significant liability, which would materially harm our business. Both federal and state governments have levied large civil and criminal fines against companies for alleged improper promotion and have enjoined several companies from engaging in off-label promotion. If we become the target of such an investigation or prosecution based on our marketing and promotional practices, we could face similar sanctions, which would materially harm our business. In addition, management’s attention could be diverted from our business operations, significant legal expenses could be incurred and our brand and reputation could be damaged. The FDA has also requested that companies enter into consent decrees or permanent injunctions under which specified promotional conduct is changed or curtailed. If we are deemed by the FDA to have engaged in the promotion of our products for off-label use, we could be subject to FDA regulatory or enforcement actions, including the issuance of an untitled letter, a warning letter, injunction, seizure, civil fine or criminal penalties. It is also possible that other federal, state or foreign enforcement authorities might take action if they consider our business activities constitute promotion of an off-label use, which could result in significant penalties, including criminal, civil or administrative penalties, damages, fines, disgorgement, exclusion from participation in government healthcare programs and the curtailment or restructuring of our operations.
We cannot, however, prevent a physician from using our product candidates outside of those indications for use when in the physician’s independent professional medical judgment he or she deems appropriate. Physicians may also misuse our product candidates or use improper techniques, potentially leading to adverse results, side effects or injury, which may lead to product liability claims. If our product candidates are misused or used with improper technique, we may become subject to costly litigation by physicians or their patients. Furthermore, the use of our product candidates for indications other than those cleared by the FDA may not effectively treat such conditions, which could harm our reputation among physicians and patients.
We may choose not to continue developing or commercializing any of our product candidates at any time during development or after approval, which would reduce or eliminate our potential return on investment for those product candidates.
At any time, we may decide to discontinue the development of any of our product candidates or not to continue commercializing one or more of our approved product candidates for a variety of reasons, including the appearance of new technologies that make our product obsolete, competition from a competing product or changes in or failure to comply with applicable regulatory requirements. If we terminate a program in which we have invested significant resources, we will not receive any return on our investment and we will have missed the opportunity to have allocated those resources to potentially more productive uses.
We or our current and prospective partners may be subject to product recalls in the future that could harm our brand and reputation and could negatively affect our business.
We or our current and prospective partners may be subject to product recalls, withdrawals or seizures if any of our product candidates, if approved for marketing, fail to meet specifications or are believed to cause injury or illness or if we are alleged to have violated governmental regulations including those related to the manufacture, labeling, promotion, sale or distribution. Any recall, withdrawal or seizure in the future could materially and adversely affect consumer confidence in our brands and lead to decreased demand for our approved products. In addition, a recall, withdrawal or seizure of any of our approved products would require significant management attention, would likely result in substantial and unexpected expenditures and would harm our business, financial condition and operating results.
If we or any partners with which we may collaborate are unable to achieve and maintain coverage and adequate levels of reimbursement for any of our product candidates for which we receive regulatory approval, or any future products we may seek to commercialize, their commercial success may be severely hindered.
For any of our product candidates that become available only by prescription, successful sales by us or by any partners with which we may collaborate depend on the availability of coverage and adequate reimbursement from third-party payors. Patients who are prescribed medicine for the treatment of their conditions generally rely on third-party payors to reimburse all or part of the costs associated with their prescription drugs. The availability of coverage and adequate reimbursement from governmental healthcare programs, such as Medicare and Medicaid, and private third-party payors is critical to new product acceptance. Coverage decisions may depend upon clinical and economic standards that disfavor new drug products when more established or lower cost therapeutic alternatives are already available or subsequently become available. If any of our product candidates do not demonstrate attractive efficacy profiles, they may not qualify for coverage and reimbursement. Even if we obtain coverage for a given product, the resulting reimbursement payment rates might not be adequate or may require co-payments that patients find unacceptably high. Patients are unlikely to use our products unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of our products.
| 26 |
| Table of Contents |
In addition, the market for our product candidates will depend significantly on access to third-party payors’ drug formularies, or lists of medications for which third-party payors provide coverage and reimbursement. The industry competition to be included in such formularies often leads to downward pricing pressures on pharmaceutical companies. Also, third-party payors may refuse to include a particular branded drug in their formularies or otherwise restrict patient access to a branded drug when a less costly generic equivalent or other alternative is available.
Further, third-party payors, whether foreign or domestic, or governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. In addition, in the United States, although private third-party payors tend to follow Medicare, no uniform policy of coverage and reimbursement for drug products exists among third-party payors. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our product candidates to each payor separately, with no assurance that coverage and adequate reimbursement will be obtained.
Further, we believe that future coverage and reimbursement will likely be subject to increased restrictions both in the United States and in international markets. Third-party coverage and reimbursement for any of our product candidates for which we may receive regulatory approval may not be available or adequate in either the United States or international markets, which could harm our business, financial condition, operating results and prospects.
Healthcare legislative or regulatory reform measures, including government restrictions on pricing and reimbursement, may have a negative impact on our business and results of operations.
In the United States and some foreign jurisdictions, there have been, and continue to be, several legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of product candidates, restrict or regulate post approval activities, and affect our ability to profitably sell any product candidates for which we obtain marketing approval.
Among policy makers and payors in the United States and elsewhere, there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality and/or expanding access. In the United States, the pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by major legislative initiatives. For example, in the United States, the Patient Protection and Affordable Care Act of 2010, or the ACA, substantially changed the way health care is financed by both governmental and private insurers and significantly affects the pharmaceutical industry. Many provisions of the ACA impact the biopharmaceutical industry, including that in order for a biopharmaceutical product to receive federal reimbursement under the Medicare Part B and Medicaid programs or to be sold directly to U.S. government agencies, the manufacturer must extend discounts to entities eligible to participate in the drug pricing program under the Public Health Services Act, or PHS. Since its enactment, there have been judicial and Congressional challenges and amendments to certain aspects of the ACA. There is continued uncertainty about the implementation of the ACA, including the potential for further amendments to the ACA and legal challenges to or efforts to repeal the ACA.
Additionally, there has been heightened governmental scrutiny in the United States of pharmaceutical pricing practices in light of the rising cost of prescription drugs and biologics. Such scrutiny has resulted in several recent congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for products. At the federal level, the now-departed Trump administration proposed numerous prescription drug cost control measures. Similarly, the new Biden administration has made lowering prescription drug prices one of its priorities. The Biden administration has not yet proposed any specific plans, but we expect that these will be forthcoming in the near term. At the state level, legislatures are increasingly passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing.
| 27 |
| Table of Contents |
Other examples of proposed changes include, but are not limited to, expanding post-approval requirements, changing the Orphan Drug Act, and restricting sales and promotional activities for pharmaceutical products. We cannot be sure whether additional legislative changes will be enacted, or whether government regulations, guidance or interpretations will be changed, or what the impact of such changes would be on the marketing approvals, sales, pricing, or reimbursement of our drug candidates or products, if any, may be. We expect that these and other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we receive for any approved drug. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize our drugs.
In addition, FDA regulations and guidance may be revised or reinterpreted by the FDA in ways that may significantly affect our business and our products. Any new regulations or guidance, or revisions or reinterpretations of existing regulations or guidance, may impose additional costs or lengthen FDA review times for DMT310 or any future product candidates. We cannot determine how changes in regulations, statutes, policies, or interpretations when and if issued, enacted or adopted, may affect our business in the future. Such changes could, among other things, require:
| • |
| additional clinical trials to be conducted prior to obtaining approval; |
| • |
| changes to manufacturing methods; |
| • |
| recalls, replacements, or discontinuance of one or more of our products; and |
| • |
| additional recordkeeping. |
Such changes would likely require substantial time and impose significant costs, or could reduce the potential commercial value of DMT310 or other product candidates. In addition, delays in receipt of or failure to receive regulatory clearances or approvals for any other products would harm our business, financial condition, and results of operations.
We may also be subject to healthcare laws, regulation and enforcement and our failure to comply with those laws could adversely affect our business, operations and financial condition.
Certain federal and state healthcare laws and regulations pertaining to fraud and abuse and patients’ rights are and will be applicable to our business. We are subject to regulation by both the federal government and the states in which we or our partners conduct our business. The laws and regulations that may affect our ability to operate include:
| • |
| the federal Anti-Kickback Statute, which prohibits, among other things, any person or entity from knowingly and willfully offering, soliciting, receiving or providing any remuneration (including any kickback, bribe or rebate), directly or indirectly, overtly or covertly, in cash or in kind, to induce either the referral of an individual or in return for the purchase, lease, or order of any good, facility item or service, for which payment may be made, in whole or in part, under federal healthcare programs such as the Medicare and Medicaid programs; |
| • |
| federal civil and criminal false claims laws and civil monetary penalty laws, including, for example, the federal civil False Claims Act, which impose criminal and civil penalties, including civil whistleblower or qui tam actions, against individuals or entities for, among other things, knowingly presenting, or causing to be presented, to the federal government, including the Medicare and Medicaid programs, claims for payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government; |
| 28 |
| Table of Contents |
| • |
| the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created new federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations or promises, any of the money or property owned by, or under the custody or control |
| of, any healthcare benefit program, regardless of the payor (e.g., public or private), knowingly and willfully embezzling or stealing from a health care benefit program, willfully obstructing a criminal investigation of a health care offense and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters; |
| • |
| HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, and their implementing regulations, which impose obligations on covered entities, including healthcare providers, health plans, and healthcare clearinghouses, as well as their respective business associates that create, receive, maintain or transmit individually identifiable health information for or on behalf of a covered entity, with respect to safeguarding the privacy, security and transmission of individually identifiable health information; |
| • |
| the federal physician sunshine requirements under the Affordable Care Act, which require manufacturers of drugs, devices, biologics and medical supplies to report annually to the Centers for Medicare & Medicaid Services information related to payments and other transfers of value provided to physicians and teaching hospitals, and ownership and investment interests held by physicians and their immediate family members; and |
| • |
| state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws, which may apply to items or services reimbursed by any third-party payor, including commercial insurers; state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government, or otherwise restrict payments that may be provided to healthcare providers and other potential referral sources; state laws that require drug manufacturers to report information related to payments and other transfers of value to healthcare providers or marketing expenditures; and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. |
Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws. In addition, recent health care reform legislation has strengthened these laws. For example, the recently enacted Affordable Care Act, among other things, amended the intent requirement of the federal Anti-Kickback Statute and certain criminal healthcare fraud statutes. A person or entity no longer needs to have actual knowledge of the statute or specific intent to violate it. In addition, the Affordable Care Act provided that the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act.
Achieving and sustaining compliance with these laws may prove costly. In addition, any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. If our operations are found to be in violation of any of the laws described above or any other governmental laws or regulations that apply to us, we may be subject to penalties, including administrative, civil and criminal penalties, damages, fines, disgorgement, the exclusion from participation in federal and state healthcare programs, individual imprisonment or the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our financial results.
| 29 |
| Table of Contents |
Our business involves the use of hazardous materials and we and our third-party suppliers and manufacturers must comply with environmental laws and regulations, which can be expensive and restrict how we do business.
The manufacturing activities of our third-party suppliers and manufacturers involve the controlled storage, use and disposal of hazardous materials owned by us, including the components of our product candidates and other hazardous compounds. We and our manufacturers and suppliers are subject to laws and regulations governing the use, manufacture, storage, handling, and disposal of these hazardous materials. In some cases, these hazardous materials and various wastes resulting from their use are stored at our suppliers’ or manufacturers’ facilities pending use and disposal. We and our suppliers and manufacturers cannot completely eliminate the risk of contamination, which could cause an interruption of our commercialization efforts, research and development efforts and business operations, injury to our service providers and others and environmental damage resulting in costly clean-up and liabilities under applicable laws and regulations governing the use, storage, handling and disposal of these materials and specified waste products. Although we believe that the safety procedures utilized by our third-party suppliers and manufacturers for handling and disposing of these materials generally comply with the standards prescribed by these laws and regulations, we cannot guarantee that this is the case or eliminate the risk of accidental contamination or injury from these materials. In such an event, we may be held liable for any resulting damages and such liability could exceed our resources. We do not currently carry biological or hazardous waste insurance coverage.
Our employees, independent contractors, principal investigators, consultants, vendors, CROs and any partners with which we may collaborate may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
We are exposed to the risk that our employees, independent contractors, principal investigators, consultants, vendors, CROs and any partners with which we may collaborate may engage in fraudulent or other illegal activity. Misconduct by these persons could include intentional, reckless or negligent conduct or unauthorized activity that violates: laws or regulations, including those laws requiring the reporting of true, complete and accurate information to the FDA or foreign regulatory authorities; manufacturing standards; federal, state and foreign healthcare fraud and abuse laws and data privacy; or laws that require the true, complete and accurate reporting of financial information or data. In particular, sales, marketing and other business arrangements in the healthcare industry are subject to extensive laws intended to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws may restrict or prohibit a wide range of business activities, including research, manufacturing, distribution, pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Activities subject to these laws also involve the improper use of information obtained in the course of clinical trials, or illegal misappropriation of drug product, which could result in regulatory sanctions or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations, and serious harm to our reputation. In addition, federal procurement laws impose substantial penalties for misconduct in connection with government contracts and require certain contractors to maintain a code of business ethics and conduct. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of civil, criminal and administrative penalties, damages, monetary fines, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could adversely affect our ability to operate our business and our operating results.
Our future growth depends, in part, on our ability to penetrate foreign markets, where we would be subject to additional regulatory burdens and other risks and uncertainties.
Our future profitability will depend, in part, on our ability to commercialize our product candidates in foreign markets for which we intend to rely on collaborations with third parties. If we commercialize DMT310 or our other product candidates in foreign markets, we would be subject to additional risks and uncertainties, including:
| • |
| our customers’ ability to obtain market access and appropriate reimbursement for our product candidates in foreign markets; |
| • |
| our inability to directly control commercial activities because we are relying on third parties; |
| 30 |
| Table of Contents |
| • |
| the burden of complying with complex and changing foreign regulatory, tax, accounting and legal requirements; |
| • |
| different medical practices and customs in foreign countries affecting acceptance in the marketplace |
| • |
| import or export licensing requirements; |
| • |
| longer accounts receivable collection times; |
| • |
| longer lead times for shipping; |
| • |
| language barriers for technical training; |
| • |
| reduced protection of intellectual property rights in some foreign countries; |
| • |
| foreign currency exchange rate fluctuations; and |
| • |
| the interpretation of contractual provisions governed by foreign laws in the event of a contract dispute. |
Foreign sales of our product candidates could also be adversely affected by the imposition of governmental controls, political and economic instability, trade restrictions and changes in tariffs, any of which may adversely affect our results of operations.
Development of test methodology for DMT310 presents unique challenges due to the complex mixture of constituents in the product. Determination of appropriate assay(s) for release and quality control evaluations could require significant development time and cost to successfully complete and uncertain.
DMT310 is comprised of both inorganic and organic constituents, and unlike most pharmaceutical products, there is no single active component to characterize for purposes of assay development. In order to release the drug product and test for stability we plan to develop a cell-based bioassay to assess inhibitory effects of DMT310 on pro-inflammatory cytokines known to play a role in the pathogenesis of various skin diseases. While this approach may show activity, it may not be suitable as a quality control release potency assay for DMT310. Furthermore, this technique may not have sufficient sensitivity to be considered stability-indicating and detect small changes or degradation to the product. If we are not able to develop a suitable potency assay utilizing this approach, we may have to identify and develop an alternative bioassay platform or secondary approaches that may require additional orthogonal methodologies to meet our testing requirements. This could be expensive, time consuming and its success uncertain, leading to delays in filing of the NDA.
Risks Related to Our Dependence on Third Parties
We are dependent on one supplier for the raw material used to produce DMT310 and DMT410. The termination of this contract would result in a disruption to product development and our business will be harmed.
We currently only have one qualified source of supply for the raw material used in DMT310 and DMT410. While we have an exclusive supply agreement with our supplier, our supplier may not comply with the terms of our agreement and may supply to third parties. DMT310 and DMT410 contain a wild growing freshwater sponge that grows in an area of the Volga river delta in Russia that is partially protected by a Russian government entity. The Russian government entity allocates a quantity of freshwater sponge that may be harvested each harvest season and may determine in any year that no sponge or a smaller quantity of sponge than harvested in previous years may be harvested in a particular year, which could impact our ability to obtain raw material to manufacture and supply DMT310 and DMT410. If we have not adequately stockpiled raw materials, or even if we do stockpile raw material, we could not have enough raw material to meet the quantity demands to conduct our non-clinical and clinical studies or to supply product for the market if approved.
The freshwater sponge contained in DMT310 and DMT410 can only be harvested once per year based on the presence of certain environmental conditions. If these environmental conditions are not present during the harvest season, then our supplier may not be able to harvest the raw material required, which could impact our ability to manufacture and supply DMT310 and DMT410. The ability of our supplier to harvest the sponge may also be impacted by severe weather and limit the length of time they can harvest, which could limit the amount of raw material that can be harvested, which may impact our ability to manufacture and supply DMT310 and DMT410. The portion of the Volga river delta where the sponge grows could also become contaminated from pollutants, which could contaminate the sponge to be harvested by our supplier, making it unusable in humans, impacting our ability to manufacture and supply DMT310 and DMT410.
| 31 |
| Table of Contents |
Even if we are able to obtain supply, we and our supplier are exposed to a number of environmental and geopolitical risks, including:
| • |
| risk of contamination being introduced in the Volga river, thereby polluting the Spongilla lacustris population through environmental factors that we cannot control, which could result in new impurities or reduced supply of raw materials; |
| • |
| loss of Spongilla lacustris habitat and other similar environmental risks to the sponge population whether due to climate change, over-development, or otherwise; |
| • |
| risk of disease in the Spongilla lacustris geographic area where harvested; |
| • |
| risk of trade issues between the U.S. and Russia; |
| • |
| restrictions on trade of certain items between the U.S. and Russia; and |
| • |
| other unforeseen geopolitical factors that limit our ability access our supply of raw material. |
Restrictions could be imposed on the harvesting of the raw material. Such events could have a significant impact on our cost and ability to produce DMT310 and DMT410 and anticipated line extensions. The country from which we obtain the raw material could change its laws and regulations regarding the export of the natural products or impose or increase taxes or duties payable by exporters of such products. In addition, any business, global or economic challenges our existing supplier faces, whether in the ordinary course of business or not, could impair its ability to supply our needs for raw materials. Accordingly, there is a risk that supplies of our raw materials may be significantly delayed by or may become unavailable as a result of any issues affecting our supply and production of naturally sourced products. In addition, if we need a new or additional suppliers, we may not be able to negotiate new agreements with an alternate or new supplier on terms that we deem commercially reasonable or at all, and the failure by us to enter into such agreements could harm our financial condition, business, clinical trials and prospects.
We have in the past relied and expect to continue to rely on third-party CROs and other third parties to conduct and oversee our clinical trials and other aspects of product development. If these third parties do not meet our requirements or otherwise conduct the trials as required, we may not be able to satisfy our contractual obligations or obtain regulatory approval for, or commercialize, our product candidates when expected or at all.
We have in the past relied and expect to continue to rely on third-party CROs to conduct and oversee our clinical trials and other aspects of product development. We also rely upon various medical institutions, clinical investigators and contract laboratories to conduct our trials in accordance with our clinical protocols and all applicable regulatory requirements, including the FDA’s regulations and GCPs, which are an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators and monitors, and state regulations governing the handling, storage, security and recordkeeping for drug and biologic products. These CROs and other third parties play a significant role in the conduct of these trials and the subsequent collection and analysis of data from the clinical trials. We rely heavily on these parties for the execution of our clinical trials and preclinical studies, and control only certain aspects of their activities. We and our CROs and other third-party contractors are required to comply with GCP, GLP, and GACP requirements, which are regulations and guidelines enforced by the FDA and comparable foreign regulatory authorities for products in clinical development. Regulatory authorities enforce these GCP, GLP and GACP requirements through periodic inspections of trial sponsors, principal investigators and trial sites. If we or any of these third parties fail to comply with applicable GCP, GLP and GACP requirements, the clinical data generated in our clinical trials may be deemed unreliable and the FDA or other regulatory authority may require us to perform additional clinical trials before approving our or our partners’ marketing applications. We cannot assure you that upon inspection by a given regulatory authority, such regulatory authority will determine that any of our clinical or preclinical trials complies with applicable GCP and GLP requirements. In addition, our clinical trials must generally be conducted with product produced under cGMP regulations. Our failure to comply with these regulations and policies may require us to repeat clinical trials, which would delay the regulatory approval process.
| 32 |
| Table of Contents |
Our CROs are not our employees, and we do not control whether or not they devote sufficient time and resources to our clinical trials. Our CROs may also have relationships with other commercial entities, including our competitors, for whom they may also be conducting clinical trials, or other drug development activities, which could harm our competitive position. We face the risk of potential unauthorized disclosure or misappropriation of our intellectual property by CROs, which may reduce our trade secret protection and allow our potential competitors to access and exploit our proprietary technology. If our CROs do not successfully carry out their contractual duties or obligations, fail to meet expected deadlines, or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols or regulatory requirements or for any other reason, our clinical trials may be extended, delayed or terminated, and we may not be able to obtain regulatory approval for, or successfully commercialize any product candidate that we develop. As a result, our financial results and the commercial prospects for any product candidate that we develop would be harmed, our costs could increase, and our ability to generate revenue could be delayed.
If any of our CROs or clinical trial sites terminate their involvement in one of our clinical trials for any reason, we may not be able to enter into arrangements with alternative CROs or clinical trial sites, or do so on commercially reasonable terms. In addition, if our relationship with clinical trial sites is terminated, we may experience the loss of follow-up information on patients enrolled in our ongoing clinical trials unless we are able to transfer the care of those patients to another qualified clinical trial site. In addition, principal investigators for our clinical trials may serve as scientific advisors or consultants to us from time to time and could receive cash or equity compensation in connection with such services. If these relationships and any related compensation result in perceived or actual conflicts of interest, the integrity of the data generated at the applicable clinical trial site may be questioned by the FDA
We rely completely on third-party contractors to supply, manufacture and distribute clinical drug supplies for our product candidates, including certain sole-source suppliers and manufacturers, we intend to rely on third parties for commercial supply, manufacturing and distribution if any of our product candidates receive regulatory approval and we expect to rely on third parties for supply, manufacturing and distribution of preclinical, clinical and commercial supplies of any future product candidates.
We do not currently have, nor do we plan to acquire, the infrastructure or capability to supply, manufacture or distribute preclinical, clinical or commercial quantities of drug substances or products. Our ability to develop our product candidates depends and our ability to commercially supply our products will depend, in part, on our ability to successfully obtain the raw materials and APIs and other substances and materials used in our product candidates from third parties and to have finished products manufactured by third parties in accordance with regulatory requirements and in sufficient quantities for preclinical and clinical testing and commercialization. If we fail to develop and maintain supply relationships with these third parties, we may be unable to continue to develop or commercialize our product candidates.
We rely and will continue to rely on certain third parties as the sole source of the materials they supply or the finished products they manufacture. Any of our existing suppliers or manufacturers may:
| • |
| fail to supply us with product on a timely basis or in the requested amount due to unexpected damage to or destruction of facilities or equipment or otherwise; |
| • |
| fail to increase manufacturing capacity and produce drug product and components in larger quantities and at higher yields in a timely or cost-effective manner, or at all, to sufficiently meet our commercial needs; |
| • |
| be unable to meet our production demands due to issues related to their reliance on sole-source suppliers and manufacturers; |
| • |
| supply us with product that fails to meet regulatory requirements; |
| • |
| become unavailable through business interruption or financial insolvency; |
| • |
| lose regulatory status as an approved source; |
| • |
| be unable or unwilling to renew current supply agreements when such agreements expire on a timely basis, on acceptable terms or at all; or |
| • |
| discontinue production or manufacturing of necessary drug substances or products. |
| 33 |
| Table of Contents |
In the event of any of the foregoing, if we do not have an alternative supplier or manufacturer in place, we would be required to expend substantial management time and expense to identify, qualify and transfer processes to alternative suppliers or manufacturers. Transferring technology to other sites may require additional processes, technologies and validation studies, which are costly, may take considerable amounts of time, may not be successful and, in most cases, require review and approval by the FDA. Any need to find and qualify new suppliers or manufacturers could significantly delay production of our product candidates, adversely impact our ability to market our product candidates and adversely affect our business. Replacements may not be available to us on a timely basis, on acceptable terms or at all. Additionally, we and our manufacturers do not currently maintain significant inventory of drug substances and other materials. Any interruption in the supply of a drug substance or other material or in the manufacture of our product candidates could have a material adverse effect on our business, financial condition, operating results and prospects.
We do not have direct control over the ability of our contract suppliers and manufacturers to maintain adequate capacity and capabilities to serve our needs, including quality control, quality assurance and qualified personnel. Although we are ultimately responsible for ensuring compliance with regulatory requirements such as cGMPs and GACP, we are dependent on our contract suppliers and manufacturers for day-to-day compliance with cGMPs or GACP for production of raw materials, APIs, and finished products. Facilities used by our contract suppliers and manufacturers to produce the APIs and other substances and materials or finished products for commercial sale must pass inspection and be approved by the FDA and other relevant regulatory authorities. Our contract suppliers and manufacturers must comply with cGMP and GACP requirements enforced by the FDA through its facilities inspection program and review of submitted technical information. If the safety of any product or product candidate or component is compromised due to a failure to adhere to applicable laws or for other reasons, we may not be able to successfully commercialize or obtain regulatory approval for the affected product or product candidate, and we may be held liable for injuries sustained as a result. Any of these factors could cause a delay or termination of preclinical studies, clinical trials or regulatory submissions or approvals of our product candidates, and could entail higher costs or result in our being unable to effectively commercialize our approved products on a timely basis, or at all.
In addition, these contract manufacturers are engaged with other companies to supply and manufacture materials or products for such companies, which also exposes our suppliers and manufacturers to regulatory risks for the production of such materials and products. As a result, failure to meet the regulatory requirements for the production of those materials and products may also affect the regulatory clearance of a contract supplier’s or manufacturer’s facility. If the FDA or a comparable foreign regulatory agency does not approve these facilities for the supply or manufacture of our product candidates, or if it withdraws its approval in the future, we may need to find alternative supply or manufacturing facilities, which would negatively impact our ability to develop, obtain regulatory approval of or market our product candidates, if approved.
Our reliance on contract manufacturers and suppliers further exposes us to the possibility that they, or third parties with access to their facilities, will have access to and may misappropriate our trade secrets or other proprietary information.
In addition, the manufacturing facilities of certain of our suppliers, including our supplier of Spongilla lacustris, are located outside of the United States. This may give rise to difficulties in importing our products or product candidates or their components into the United States or other countries as a result of, among other things, regulatory agency approval requirements or import inspections, incomplete or inaccurate import documentation or defective packaging.
| 34 |
| Table of Contents |
If we are not able to establish and maintain collaborations, we may have to alter our development and commercialization plans.
The development and potential commercialization of our product candidates will require substantial additional cash to fund expenses. In order to fund further development of our product candidates, we may collaborate with pharmaceutical and biotechnology companies for the development and potential commercialization of those product candidates. We face significant competition in seeking appropriate partners. Whether we reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the partner’s resources and experience, the terms and conditions of the proposed collaboration and the proposed partner’s evaluation of a number of factors. Those factors may include the design or results of clinical trials; the likelihood of approval by the FDA or other regulatory authorities; the potential market for the subject product candidate; the costs and complexities of manufacturing and delivering such product candidate to patients; the potential of competing products; any uncertainty with respect to our ownership of our intellectual property; and industry and market conditions generally. The partner may also consider alternative product candidates or technologies for similar indications that may be available for collaboration and whether such a collaboration could be more attractive than the one with us for our product candidate. We may also be restricted under future license agreements from entering into agreements on certain terms with potential partners. Collaborations are complex and time-consuming to negotiate and document. In addition, there have been a significant number of recent business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future partners.
Future collaborations we may enter into may involve the following risks:
| • |
| collaborators may have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
| • |
| collaborators may not perform their obligations as expected; |
| • |
| changes in the collaborators’ strategic focus or available funding, or external factors, such as an acquisition, may divert resources or create competing priorities; |
| • |
| collaborators may delay discovery and preclinical development, provide insufficient funding for product development of targets selected by us, stop or abandon discovery and preclinical development for a product candidate, repeat or conduct new discovery and preclinical development for a product candidate; |
| • |
| collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our products or product candidates if the collaborators believe that competitive products are more likely to be successfully developed than ours; |
| • |
| product candidates discovered in collaboration with us may be viewed by our collaborators as competitive with their own product candidates or products, which may cause collaborators to cease to devote resources to the development of our product candidates; |
| • |
| disagreements with collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development, might cause delays or termination of the discovery, preclinical development or commercialization of product candidates, might lead to additional responsibilities for us with respect to product candidates, or might result in litigation or arbitration, any of which would be time-consuming and expensive; |
| • |
| collaborators may not properly maintain or defend our intellectual property rights or intellectual property rights licensed to us or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation; |
| • |
| collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability; and |
| • |
| collaborations may be terminated for the convenience of the collaborator and, if terminated, we could be required to raise additional capital to pursue further development or commercialization of the applicable product candidates. |
| 35 |
| Table of Contents |
Collaborations typically impose detailed obligations on each party. If we were to breach our obligations, we may face substantial consequences, including potential termination of the collaboration, and our rights to our partners’ product candidates, in which we have invested substantial time and money, would be lost.
We may not be able to negotiate collaborations on a timely basis, on acceptable terms or at all. If we are unable to do so, we may have to curtail the development of a product candidate, reduce or delay its development program or one or more of our other development programs, delay its potential commercialization or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to increase our expenditures to fund development or commercialization activities on our own, we may need to obtain additional capital, which may not be available to us on acceptable terms or at all. If we do not have sufficient funds, we may not be able to further develop our product candidates or bring them to market and generate product revenue.
Risks Related to Managing Our Growth, Our Employees and Our Operations
We will need to further increase the size and complexity of our organization in the future, and we may experience difficulties in executing our growth strategy and managing any growth.
Our management, personnel, systems and facilities currently in place are not adequate to support our business plan and near-term future growth. We will need to further expand our chemistry and manufacturing team, clinical team, managerial, operational, financial, and other resources to support our planned research, development and commercialization activities.
To manage our operations, growth and various projects effectively requires that we:
| • |
| continue to improve our operational, financial, management and regulatory compliance controls and reporting systems and procedures; |
| • |
| attract and retain sufficient numbers of talented employees; |
| • |
| develop a marketing, sales and distribution capability; |
| • |
| manage our commercialization activities for our product candidates effectively and in a cost-effective manner; |
| • |
| establish and maintain relationships with development and commercialization partners; |
| • |
| manage our preclinical and clinical trials effectively; |
| • |
| manage our third-party supply and manufacturing operations effectively and in a cost-effective manner, while increasing production capabilities for our current product candidates to commercial levels; and |
| • |
| manage our development efforts effectively while carrying out our contractual obligations to partners and other third parties. |
In addition, historically, we have utilized and continue to utilize the services of part-time outside consultants to perform a number of tasks for us, including tasks related to preclinical and clinical testing. Our growth strategy may also entail expanding our use of consultants to implement these and other tasks going forward. We rely on consultants for certain functions of our business and will need to effectively manage these consultants to ensure that they successfully carry out their contractual obligations and meet expected deadlines. There can be no assurance that we will be able to manage our existing consultants or find other competent outside consultants, as needed, on economically reasonable terms, or at all. If we are not able to effectively manage our growth and expand our organization by hiring new employees and expanding our use of consultants, we might be unable to implement successfully the tasks necessary to execute effectively on our planned research, development and commercialization activities and, accordingly, might not achieve our research, development and commercialization goals.
| 36 |
| Table of Contents |
If we fail to attract and retain management and other key personnel, we may be unable to continue to successfully develop or commercialize our product candidates or otherwise implement our business plan.
Our ability to compete in the highly competitive pharmaceuticals industry depends upon our ability to attract and retain highly qualified managerial, scientific, medical, sales and marketing and other personnel. We are highly dependent on our management and scientific personnel, including: our Chief Executive Officer, President and director, Gerald T. Proehl; our Senior Vice President, Regulatory Affairs and Quality Assurance, Maria Bedoya Toro Munera, Ph.D., M.B.A.; and our Senior Vice President, Development, Christopher J. Nardo, M.P.H., Ph.D. The loss of the services of any of these individuals could impede, delay or prevent the successful development of our product pipeline, completion of our planned clinical trials, commercialization of our product candidates or in-licensing or acquisition of new assets and could negatively impact our ability to successfully implement our business plan. If we lose the services of any of these individuals, we might not be able to find suitable replacements on a timely basis or at all, and our business could be harmed as a result. We do not maintain “key man” insurance policies on the lives of these individuals or the lives of any of our other employees. In order to retain valuable employees at our company, in addition to salary and cash incentives, we provide stock options that vest over time. The value to employees of stock options that vest over time will be significantly affected by movements in our stock price that are beyond our control, and may at any time be insufficient to counteract offers from other companies.
We might not be able to attract or retain qualified management and other key personnel in the future due to the intense competition for qualified personnel among biotechnology, pharmaceutical and other businesses, particularly in the San Diego area where we are headquartered. We could have difficulty attracting experienced personnel to our company and may be required to expend significant financial resources in our employee recruitment and retention efforts. Many of the other pharmaceutical companies with whom we compete for qualified personnel have greater financial and other resources, different risk profiles and longer histories in the industry than we do. They also may provide more diverse opportunities and better chances for career advancement. If we are not able to attract and retain the necessary personnel to accomplish our business objectives, we may experience constraints that will harm our ability to implement our business strategy and achieve our business objectives.
In addition, we have scientific and clinical advisors who assist us in formulating our development and clinical strategies. These advisors are not our employees and may have commitments to, or consulting or advisory contracts with, other entities that may limit their availability to us. In addition, our advisors may have arrangements with other companies to assist those companies in developing products or technologies that may compete with ours.
Our ability to attract and retain qualified members of our board of directors may be impacted due to new state laws, including recently enacted gender and diversity quotas.
In September 2018, the state of California enacted SB 826 requiring public companies headquartered in California to maintain minimum female representation on their boards of directors as follows: by the end of 2019, at least one woman on its board, by the end of 2020, public company boards with five members will be required to have at least two female directors, and public company boards with six or more members will be required to have at least three female directors. In September 2020, the state of California enacted AB 979 requiring public companies headquartered in California to maintain minimum representation on their boards of directors from members of underrepresented communities as follow: by the end of 2021, at least one director from an underrepresented community, by end of 2022, public company boards with more than four but fewer than nine members will be required to have at least two directors from underrepresented communities, and public company board with nine or more members will be required to have at least three directors from underrepresented communities. Failure to achieve designated minimum levels in a timely manner exposes such companies to financial penalties and reputational harm. We cannot assure that we can recruit, attract and/or retain qualified members of the board and meet the above quotas as a result of the California laws, which may expose us to penalties and/or reputational harm.
| 37 |
| Table of Contents |
We currently have limited marketing capabilities and no sales organization. If we are unable to establish sales and marketing capabilities on our own or through third parties, we will be unable to successfully commercialize our product candidates, if approved, or generate product revenue.
We currently have limited marketing capabilities and no sales organization. To commercialize our product candidates, if approved, in the United States, Canada, the European Union and other jurisdictions we seek to enter, we must build our marketing, sales, distribution, managerial and other non-technical capabilities or make arrangements with third parties to perform these services, and we may not be successful in doing so. Although our management team has experience in the marketing, sale and distribution of pharmaceutical products from prior employment at other companies, we as a company have no prior experience in the marketing, sale and distribution of pharmaceutical products and there are significant risks involved in building and managing a sales organization, including our ability to hire, retain and incentivize qualified individuals, generate sufficient sales leads, provide adequate training to sales and marketing personnel and effectively manage a geographically dispersed sales and marketing team. Any failure or delay in the development of our internal sales, marketing and distribution capabilities would adversely impact the commercialization of these products. We may choose to collaborate with additional third parties that have direct sales forces and established distribution systems, either to augment our own sales force and distribution systems or in lieu of our own sales force and distribution systems. If we are unable to enter into such arrangements on acceptable terms or at all, we may not be able to successfully commercialize our product candidates. If we are unable to successfully commercialize our product candidates, either on our own or through collaborations with one or more third parties, our business, financial condition, operating results and prospects would suffer.
Our failure to successfully in-license, acquire, develop and market additional product candidates or approved products would impair our ability to grow our business.
We intend to in-license, acquire, develop and market additional products and product candidates and we may in-license or acquire commercial-stage products or engage in other strategic transactions. Because our internal research and development capabilities are limited, we may be dependent upon pharmaceutical companies, academic scientists and other researchers to sell or license products or technology to us. The success of this strategy depends partly upon our ability to identify and select promising pharmaceutical product candidates and products, negotiate licensing or acquisition agreements with their current owners and finance these arrangements.
The process of proposing, negotiating and implementing a license or acquisition of a product candidate or approved product is lengthy and complex. Other companies, including some with substantially greater financial, marketing, sales and other resources, may compete with us for the license or acquisition of product candidates and approved products. We have limited resources to identify and execute the acquisition or in-licensing of third-party products, businesses and technologies and integrate them into our current infrastructure. Moreover, we may devote resources to potential acquisitions or licensing opportunities that are never completed, or we may fail to realize the anticipated benefits of such efforts. We may not be able to acquire the rights to additional product candidates on terms that we find acceptable, or at all.
Further, any product candidate that we acquire may require additional development efforts prior to commercial sale, including preclinical or clinical testing and approval by the FDA and applicable foreign regulatory authorities. All product candidates are prone to risks of failure typical of pharmaceutical product development, including the possibility that a product candidate will not be shown to be sufficiently safe and effective for approval by regulatory authorities. In addition, we cannot provide assurance that any approved products that we acquire will be manufactured or sold profitably or achieve market acceptance.
Additional potential transactions that we may consider include a variety of different business arrangements, including spin-offs, strategic partnerships, joint ventures, restructurings, divestitures, business combinations and investments. Any such transaction may require us to incur non-recurring or other charges, may increase our near- and long-term expenditures and may pose significant integration challenges or disrupt our management or business, which could adversely affect our operations and financial results. For example, these transactions entail numerous potential operational and financial risks, including:
| • |
| exposure to unknown liabilities; |
| • |
| disruption of our business and diversion of our management’s time and attention in order to develop acquired products, product candidates or technologies; |
| • |
| incurrence of substantial debt or dilutive issuances of equity securities to pay for acquisitions; |
| • |
| substantial acquisition and integration costs; |
| • |
| write-downs of assets or impairment charges; |
| • |
| increased amortization expenses; |
| • |
| difficulty and cost in combining the operations and personnel of any acquired businesses with our operations and personnel; |
| • |
| impairment of relationships with key suppliers, partners or customers of any acquired businesses due to changes in management and ownership; and |
| • |
| inability to retain our key employees or those of any acquired businesses. |
| 38 |
| Table of Contents |
Accordingly, there can be no assurance that we will undertake or successfully complete any transactions of the nature described above, and any transaction that we do complete could harm our business, financial condition, operating results and prospects.
Manufacturing and supply of the APIs and other substances and materials used in our product candidates is a complex and technically challenging undertaking, and there is potential for failure at many points in the manufacturing, testing, quality assurance and distribution supply chain, as well as the potential for latent defects after products have been manufactured and distributed.
Manufacturing and supply of APIs, other substances and materials and finished drug products is technically challenging. Changes beyond our direct control can impact the quality, volume, price and successful delivery of our product candidates and can impede, delay, limit or prevent the successful development and commercialization of our product candidates. Mistakes and mishandling are not uncommon and can affect successful production and supply. Some of these risks include:
| • |
| failure of our manufacturers to follow cGMP or GACP requirements or mishandling of product while in production or in preparation for transit; |
| • |
| inability of our contract suppliers and manufacturers to efficiently and cost-effectively increase and maintain high yields and batch quality, consistency and stability; |
| • |
| our inability to develop an FDA approved bioassay for release of any future product; |
| • |
| difficulty in establishing optimal drug delivery substances and techniques, production and storage methods and packaging and shipment processes; |
| • |
| transportation and import/export risk, particularly given the global nature of our supply chain; |
| • |
| delays in analytical results or failure of analytical techniques that we depend on for quality control and release of any future product; |
| • |
| natural disasters, pandemics, labor disputes, financial distress, lack of raw material supply, issues with facilities and equipment or other forms of disruption to business operations of our contract manufacturers and suppliers; and |
| • |
| latent defects that may become apparent after the product has been released and which may result in recall and destruction of product. |
Any of these factors could result in delays or higher costs in connection with our clinical trials, regulatory submissions, required approvals or commercialization of our product candidates, which could harm our business, financial condition, operating results and prospects.
| 39 |
| Table of Contents |
Our operating results may fluctuate significantly, which makes our future operating results difficult to predict and could cause our operating results to fall below expectations.
Our operations to date have been primarily limited to researching and developing our product candidates and undertaking preclinical studies and clinical trials of our product candidates. We have not yet obtained regulatory approvals for any of our product candidates. Consequently, any predictions you make about our future success or viability may not be as accurate as they could be if we had a longer operating history or approved products on the market. Furthermore, our operating results may fluctuate due to a variety of other factors, many of which are outside of our control and may be difficult to predict, including the following:
| • |
| delays in the commencement, enrollment and the timing of clinical testing for our product candidates; |
| • |
| the timing and success or failure of clinical trials for our product candidates or competing product candidates, or any other change in the competitive landscape of our industry, including consolidation among our competitors or partners; |
| • |
| any delays in regulatory review and approval of product candidates in clinical development; |
| • |
| the timing and cost of, and level of investment in, research and development activities relating to our product candidates, which may change from time to time; |
| • |
| the cost of manufacturing our product candidates, which may vary depending on FDA guidelines and requirements, and the quantity of production; |
| • |
| our ability to obtain additional funding to develop our product candidates; |
| • |
| expenditures that we will or may incur to acquire or develop additional product candidates and technologies; |
| • |
| the level of demand for our product candidates, should they receive approval, which may vary significantly; |
| • |
| potential side effects of our product candidates that could delay or prevent commercialization or cause an approved drug to be taken off the market; |
| • |
| the ability of patients or healthcare providers to obtain coverage of or sufficient reimbursement for our product candidates, if approved; |
| • |
| our dependency on third-party manufacturers to supply or manufacture our product candidates; |
| • |
| our ability to establish an effective sales, marketing and distribution infrastructure in a timely manner; |
| • |
| market acceptance of our product candidates, if approved, and our ability to forecast demand for those product candidates; |
| • |
| our ability to receive approval and commercialize our product candidates outside of the United States; |
| • |
| our ability to establish and maintain collaborations, licensing or other arrangements; |
| • |
| our ability and third parties’ abilities to protect intellectual property rights; |
| • |
| costs related to and outcomes of potential litigation or other disputes; |
| • |
| our ability to adequately support future growth; |
| • |
| our ability to attract and retain key personnel to manage our business effectively; |
| • |
| potential liabilities associated with hazardous materials; |
| • |
| our ability to maintain adequate insurance policies; and |
| • |
| future accounting pronouncements or changes in our accounting policies. |
| 40 |
| Table of Contents |
Our operating results and liquidity needs could be negatively affected by market fluctuations and economic downturn.
Our operating results and liquidity could be negatively affected by economic conditions generally, both in the United States and elsewhere around the world. The market for discretionary medical products and procedures may be particularly vulnerable to unfavorable economic conditions. Some patients may consider certain of our product candidates to be discretionary, and if full reimbursement for such products is not available, demand for these products may be tied to the discretionary spending levels of our targeted patient populations. Domestic and international equity and debt markets have experienced and may continue to experience heightened volatility and turmoil based on domestic and international economic conditions and concerns. In the event these economic conditions and concerns continue or worsen and the markets continue to remain volatile, our operating results and liquidity could be adversely affected by those factors in many ways, including weakening demand for certain of our products and making it more difficult for us to raise funds if necessary, and our stock price may decline. Additionally, although we plan to market our products primarily in the United States, our partners have extensive global operations, indirectly exposing us to risk.
Our business and operations would suffer in the event of failures in our internal computer systems.
Despite the implementation of security measures, our internal computer systems and those of our current and any future partners, contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we have not experienced any such material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our manufacturing activities, development programs and our business operations. For example, the loss of manufacturing records or clinical trial data from completed or future clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further commercialization and development of our products and product candidates could be delayed.
We are increasingly dependent on information technology, and our systems and infrastructure face certain risks, including cybersecurity and data leakage risks.
Significant disruptions to our information technology systems or breaches of information security could adversely affect our business. In the ordinary course of business, we collect, store and transmit large amounts of confidential information, and it is critical that we do so in a secure manner to maintain the confidentiality and integrity of such confidential information. The size and complexity of our information technology systems, and those of our third-party vendors with whom we contract, make such systems potentially vulnerable to service interruptions and security breaches from inadvertent or intentional actions by our employees, partners or vendors, from attacks by malicious third parties, or from intentional or accidental physical damage to our systems infrastructure maintained by us or by third parties. Maintaining the secrecy of this confidential, proprietary, or trade secret information is important to our competitive business position. While we have taken steps to protect such information and invested in information technology, there can be no assurance that our efforts will prevent service interruptions or security breaches in our systems or the unauthorized or inadvertent wrongful use or disclosure of confidential information that could adversely affect our business operations or result in the loss, dissemination, or misuse of critical or sensitive information. A breach of our security measures or the accidental loss, inadvertent disclosure, unapproved dissemination, misappropriation or misuse of trade secrets, proprietary information, or other confidential information, whether as a result of theft, hacking, fraud, trickery or other forms of deception, or for any other reason, could enable others to produce competing products, use our proprietary technology or information, or adversely affect our business or financial condition. Further, any such interruption, security breach, loss or disclosure of confidential information, could result in financial, legal, business, and reputational harm to us and could have a material adverse effect on our business, financial position, results of operations or cash flow.
Risks Related to Our Intellectual Property
We may not be able to obtain or enforce patent rights or other intellectual property rights that cover our product candidates and technologies that are of sufficient breadth to prevent third parties from competing against us.
Our success with respect to our product candidates and technologies will depend in part on our ability to obtain and maintain patent protection in both the United States and other countries, to preserve our trade secrets and to prevent third parties from infringing upon our proprietary rights. Our ability to protect any of our product candidates from unauthorized or infringing use by third parties depends in substantial part on our ability to obtain and maintain valid and enforceable patents or enforce confidentiality contracts.
| 41 |
| Table of Contents |
Our patents include licensed patents and patent applications in the United States and foreign jurisdictions where we believe there is a market opportunity for our products. The covered technology and the scope of coverage vary from country to country. For those countries where we do not have granted patents, we may not have any ability to prevent the unauthorized use of our technologies. Any patents that we may obtain may be narrow in scope and thus easily circumvented by competitors. Further, in countries where we do not have granted patents, third parties may be able to make, use or sell products identical to or substantially similar to, our product candidates.
The patent application process, also known as patent prosecution, is expensive and time-consuming, and we and our current or future licensors and licensees may not be able to prepare, file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we or our current licensors, or any future licensors or licensees, will fail to identify patentable aspects of inventions made in the course of development and commercialization activities before it is too late to obtain patent protection on them. Therefore, these and any of our patents and applications may not be prosecuted and enforced in a manner consistent with the best interests of our business. It is possible that defects of form in the preparation or filing of our patents or patent applications may exist, or may arise in the future, such as with respect to proper priority claims, inventorship, claim scope or patent term adjustments. If our current licensors, or any future licensors or licensees, are not fully cooperative or disagree with us as to the prosecution, maintenance or enforcement of any patent rights, such patent rights could be compromised and we might not be able to prevent third parties from making, using and selling competing products. If there are material defects in the form or preparation of our patents or patent applications, such patents or applications may be invalid and unenforceable. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business, financial condition and operating results.
Due to legal standards relating to patentability, validity, enforceability and claim scope of patents covering pharmaceutical inventions, our ability to obtain, maintain and enforce patents is uncertain and involves complex legal and factual questions. Accordingly, rights under any existing patents or any patents we might obtain or license may not cover our product candidates, or may not provide us with sufficient protection for our product candidates to afford a commercial advantage against competitive products or processes, including those from branded and generic pharmaceutical companies. In addition, we cannot guarantee that any patents will issue from any pending or future patent applications owned by or licensed to us. Even if patents have issued or will issue, we cannot guarantee that the claims of these patents are or will be held valid or enforceable by the courts or will provide us with any significant protection against competitive products or otherwise be commercially valuable to us.
Competitors in the field of dermatologic therapeutics have created a substantial amount of prior art, including scientific publications, patents and patent applications. Our ability to obtain and maintain valid and enforceable patents depends on whether the differences between our technology and the prior art allow our technology to be patentable over the prior art. Although we believe that our technology includes certain inventions that are unique and not duplicative of any prior art, we do not have outstanding issued patents covering all of the recent developments in our technology and we are unsure of the patent protection that we will be successful in obtaining, if any. Even if the patents do successfully issue, third parties may design around or challenge the validity, enforceability or scope of such issued patents or any other issued patents we own or license, which may result in such patents being narrowed, invalidated or held unenforceable. If the breadth or strength of protection provided by the patents we hold or pursue with respect to our product candidates is challenged, it could dissuade companies from collaborating with us to develop, or threaten our ability to commercialize, our product candidates.
The laws of some foreign jurisdictions do not provide intellectual property rights to the same extent as in the United States and many companies have encountered significant difficulties in protecting and defending such rights in foreign jurisdictions. If we encounter such difficulties in protecting or are otherwise precluded from effectively protecting our intellectual property in foreign jurisdictions, our business prospects could be substantially harmed. The patent positions of pharmaceutical and biotechnology companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. Changes in either the patent laws or in the interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced in our patents or in third-party patents.
| 42 |
| Table of Contents |
The degree of future protection of our proprietary rights is uncertain. Patent protection may be unavailable or severely limited in some cases and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
| • |
| we might not have been the first to invent or the first to file the inventions covered by each of our pending patent applications and issued patents; |
| • |
| others may independently develop similar or alternative technologies or duplicate any of our technologies; |
| • |
| the patents of others may have an adverse effect on our business; |
| • |
| any patents we obtain or our licensors’ issued patents may not encompass commercially viable products, may not provide us with any competitive advantages or may be challenged by third parties; |
| • |
| any patents we obtain on our in-licensed issued patents may not be valid or enforceable; and |
| • |
| we may not develop additional proprietary technologies that are patentable. |
Patents have a limited lifespan. In the United States, the natural expiration of a patent is generally 20 years from its earliest non-provisional priority application filing date. Various extensions may be available; however the life of a patent, and the protection it affords, is limited. Without patent protection for our product candidates, we may be open to competition from generic versions of our product candidates. Further, the extensive period of time between patent filing and regulatory approval for a product candidate limits the time during which we can market a product candidate under patent protection, which may particularly affect the profitability of our early-stage product candidates. The issued U.S. patents relating to DMT310 will expire between 2022 and 2023.
Proprietary trade secrets and unpatented know-how are also very important to our business. Although we have taken steps to protect our trade secrets and unpatented know-how by entering into confidentiality agreements with third parties, and intellectual property protection agreements with certain employees, consultants and advisors, third parties may still obtain this information or we may be unable to protect our rights. We also have limited control over the protection of trade secrets used by our suppliers, manufacturers and other third parties. There can be no assurance that binding agreements will not be breached, that we would have adequate remedies for any breach or that our trade secrets and unpatented know-how will not otherwise become known or be independently discovered by our competitors. If trade secrets are independently discovered, we would not be able to prevent their use. Enforcing a claim that a third party illegally obtained and is using our trade secrets or unpatented know-how is expensive and time-consuming, and the outcome is unpredictable. In addition, courts outside the United States may be less willing to protect trade secret information.
Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our future patents.
Our ability to obtain patents is highly uncertain because, to date, some legal principles remain unresolved, there has not been a consistent policy regarding the breadth or interpretation of claims allowed in patents in the United States and the specific content of patents and patent applications that are necessary to support and interpret patent claims is highly uncertain due to the complex nature of the relevant legal, scientific, and factual issues. Changes in either patent laws or interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property or narrow the scope of our patent protection.
For example, on September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to United States patent law. These include provisions that affect the way patent applications will be prosecuted and may also affect patent litigation. The United States Patent and Trademark Office, or USPTO, has developed new and untested regulations and procedures to govern the full implementation of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, and in particular, the first to file provisions, only became effective in March 2013. The Leahy-Smith Act has also introduced procedures making it easier for third parties to challenge issued patents, as well as to intervene in the prosecution of patent applications. Finally, the Leahy-Smith Act contains new statutory provisions that require the USPTO to issue new regulations for their implementation, and it may take the courts years to interpret the provisions of the new statute. It is too early to tell what, if any, impact the Leahy-Smith Act will have on the operation of our business and the protection and enforcement of our intellectual property. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our future patents. Further, the U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on actions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce patents that we have owned or licensed or that we might obtain in the future. An inability to obtain, enforce, and defend patents covering our proprietary technologies would materially and adversely affect our business prospects and financial condition.
| 43 |
| Table of Contents |
Similarly, changes in patent laws and regulations in other countries or jurisdictions or changes in the governmental bodies that enforce them or changes in how the relevant governmental authority enforces patent laws or regulations may weaken our ability to obtain new patents or to enforce patents that we may obtain in the future. Further, the laws of some foreign countries do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant problems in protecting and defending our intellectual property both in the United States and abroad. For example, if the issuance to us, in a given country, of a patent covering an invention is not followed by the issuance, in other countries, of patents covering the same invention, or if any judicial interpretation of the validity, enforceability, or scope of the claims, or the written description or enablement, in a patent issued in one country is not similar to the interpretation given to the corresponding patent issued in another country, our ability to protect our intellectual property in those countries may be limited. Changes in either patent laws or in interpretations of patent laws in the United States and other countries may materially diminish the value of our intellectual property or narrow the scope of our patent protection.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on our product candidates in all countries throughout the world would be prohibitively expensive. The requirements for patentability may differ in certain countries, particularly developing countries. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as laws in the United States. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where we have patent protection, but enforcement on infringing activities is inadequate. These products may compete with our products, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to pharmaceuticals, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. In addition, certain countries in Europe and certain developing countries have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In those countries, we may have limited remedies if our patents are infringed or if we are compelled to grant a license to our patents to a third party, which could materially diminish the value of those patents. This could limit our potential revenue opportunities. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we own or license. Finally, our ability to protect and enforce our intellectual property rights may be adversely affected by unforeseen changes in foreign intellectual property laws.
| 44 |
| Table of Contents |
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance and annuity fees on any issued patent are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we or our licensors fail to maintain the patents and patent applications covering our product candidates, our competitors might be able to enter the market, which would have an adverse effect on our business.
If we fail to comply with our obligations under our intellectual property license agreements, we could lose license rights that are important to our business.
We are a party to certain license agreements that impose various diligence, milestone, royalty, insurance and other obligations on us. If we fail to comply with these obligations, the respective licensors may have the right to terminate the license, in which event we may not be able to develop or market the affected product candidate. The loss of such rights could materially adversely affect our business, financial condition, operating results and prospects. For more information about these license arrangements, see “Business-Collaborations and License Agreements.”
If we are sued for infringing intellectual property rights of third parties, it will be costly and time-consuming, and an unfavorable outcome in that litigation could have a material adverse effect on our business.
Our commercial success depends upon our ability to develop, manufacture, market and sell our product candidates and use our proprietary technologies without infringing the proprietary rights of third parties. We cannot guarantee that marketing and selling such candidates and using such technologies will not infringe existing or future patents. Numerous U.S. and foreign issued patents and pending patent applications owned by third parties exist in the fields relating to our product candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that others may assert that our product candidates, technologies or methods of delivery or use infringe their patent rights. Moreover, it is not always clear to industry participants, including us, which patents cover various drugs, biologics, drug delivery systems or their methods of use, and which of these patents may be valid and enforceable. Thus, because of the large number of patents issued and patent applications filed in our fields, there may be a risk that third parties may allege they have patent rights encompassing our product candidates, technologies or methods.
In addition, there may be issued patents of third parties that are infringed or are alleged to be infringed by our product candidates or proprietary technologies. Because some patent applications in the United States may be maintained in secrecy until the patents are issued, because patent applications in the United States and many foreign jurisdictions are typically not published until eighteen months after filing and because publications in the scientific literature often lag behind actual discoveries, we cannot be certain that others have not filed patent applications for technology covered by our own and in-licensed issued patents or our pending applications. Our competitors may have filed, and may in the future file, patent applications covering our product candidates or technology similar to ours. Any such patent application may have priority over our own and in-licensed patent applications or patents, which could further require us to obtain rights to issued patents covering such technologies. If another party has filed a U.S. patent application on inventions similar to those owned or in-licensed to us, we or, in the case of in-licensed technology, the licensor may have to participate, in the United States, in an interference proceeding to determine priority of invention.
We may be exposed to, or threatened with, future litigation by third parties having patent or other intellectual property rights alleging that our product candidates or proprietary technologies infringe such third parties’ intellectual property rights, including litigation resulting from filing under Paragraph IV of the Hatch-Waxman Act. These lawsuits could claim that there are existing patent rights for such drug and this type of litigation can be costly and could adversely affect our operating results and divert the attention of managerial and technical personnel, even if we do not infringe such patents or the patents asserted against us are ultimately established as invalid. There is a risk that a court would decide that we are infringing the third party’s patents and would order us to stop the activities covered by the patents. In addition, there is a risk that a court will order us to pay the other party damages for having violated the other party’s patents.
| 45 |
| Table of Contents |
As a result of patent infringement claims, or to avoid potential claims, we may choose or be required to seek licenses from third parties. These licenses may not be available on commercially acceptable terms, or at all. Even if we are able to obtain a license, the license would likely obligate us to pay license fees or royalties or both, and the rights granted to us might be nonexclusive, which could result in our competitors gaining access to the same intellectual property, or such rights might be restrictive and limit our present and future activities. Ultimately, we or a licensee could be prevented from commercializing a product, or forced to cease some aspect of our business operations, if, as a result of actual or threatened patent infringement claims, we are unable to enter into licenses on acceptable terms.
In addition to possible infringement claims against us, we may become a party to other patent litigation and other proceedings, including interference, derivation, re-examination or other post-grant proceedings declared or granted by the USPTO, and similar proceedings in foreign countries, regarding intellectual property rights with respect to our current or of our other products.
There is a substantial amount of litigation involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries generally. To date, no litigation asserting infringement claims has ever been brought against us. If a third party claims that we infringe its intellectual property rights, we may face a number of issues, including:
| • |
| infringement and other intellectual property claims which, regardless of merit, may be expensive and time-consuming to litigate and may divert our management’s attention from our core business; |
| • |
| substantial damages for infringement, which we may have to pay if a court decides that the product or technology at issue infringes or violates the third party’s rights, and if the court finds that the infringement was willful, we could be ordered to pay treble damages and the patent owner’s attorneys’ fees; |
| • |
| a court prohibiting us from selling or licensing the product or using the technology unless the third party licenses its intellectual property rights to us, which it is not required to do; |
| • |
| if a license is available from a third party, we may have to pay substantial royalties or upfront fees or grant cross-licenses to intellectual property rights for our products or technologies; and |
| • |
| redesigning our products or processes so they do not infringe, which may not be possible or may require substantial monetary expenditures and time. |
Some of our competitors may be able to sustain the costs of complex patent litigation more effectively than we can because they have substantially greater resources. In addition, any uncertainties resulting from the initiation and continuation of any litigation could harm our ability to raise additional funds or otherwise adversely affect our business, financial condition, operating results and prospects.
Because we rely on certain third-party licensors and partners, and will continue to do so in the future, if one of our licensors or partners is sued for infringing a third party’s intellectual property rights, our business, financial condition, operating results and prospects could suffer in the same manner as if we were sued directly. In addition to facing litigation risks, we have agreed to indemnify certain third-party licensors and partners against claims of infringement caused by our proprietary technologies, and we have entered or may enter into cost-sharing agreements with some our licensors and partners that could require us to pay some of the costs of patent litigation brought against those third parties whether or not the alleged infringement is caused by our proprietary technologies. In certain instances, these cost-sharing agreements could also require us to assume greater responsibility for infringement damages than would be assumed just on the basis of our technology.
The occurrence of any of the foregoing could adversely affect our business, financial condition or operating results.
| 46 |
| Table of Contents |
We may become involved in lawsuits to protect or enforce our patents or other intellectual property or the patents of our licensors, which could be expensive and time-consuming.
Competitors may infringe our intellectual property, including our patents or the patents of our licensors. As a result, we may be required to file infringement claims to stop third-party infringement or unauthorized use. This can be expensive and time-consuming, particularly for a company of our size. In addition, in an infringement proceeding, a court may decide that a patent of ours is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our patent claims do not cover its technology or that the factors necessary to grant an injunction against an infringer are not satisfied. An adverse determination of any litigation or other proceedings could put one or more of our patents at risk of being invalidated, interpreted narrowly or amended such that they do not cover our product candidates. Moreover, such adverse determinations could put our patent applications at risk of not issuing, or issuing with limited and potentially inadequate scope to cover our product candidates or to prevent others from marketing similar products.
Interference, derivation or other proceedings brought at the USPTO may be necessary to determine the priority or patentability of inventions with respect to our patent applications or those of our licensors or potential partners. Litigation or USPTO proceedings brought by us may fail or may be invoked against us by third parties. Even if we are successful, domestic or foreign litigation or USPTO or foreign patent office proceedings may result in substantial costs and distraction to our management. We may not be able, alone or with our licensors or potential partners, to prevent misappropriation of our proprietary rights, particularly in countries where the laws may not protect such rights as fully as in the United States.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation or other proceedings, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation or other proceedings. In addition, during the course of this kind of litigation or proceedings, there could be public announcements of the results of hearings, motions or other interim proceedings or developments or public access to related documents. If investors perceive these results to be negative, the market price for our common stock or warrants could be significantly harmed.
Our reliance on third parties requires us to share our trade secrets, which increases the possibility that our trade secrets will be misappropriated or disclosed, and confidentiality agreements with employees and third parties may not adequately prevent disclosure of trade secrets and protect other proprietary information.
We consider proprietary trade secrets or confidential know-how and unpatented know-how to be important to our business. We may rely on trade secrets or confidential know-how to protect our technology, especially where patent protection is believed by us to be of limited value.
To protect this type of information against disclosure or appropriation by competitors, our policy is to require our employees, consultants, collaborators, contractors and advisors to enter into confidentiality agreements and, if applicable, material transfer agreements, consulting agreements or other similar agreements with us prior to beginning research or disclosing proprietary information. These agreements typically limit the rights of the third parties to use or disclose our confidential information, including our trade secrets. However, current or former employees, consultants, collaborators, contractors and advisors may unintentionally or willfully disclose our confidential information to competitors, and confidentiality agreements may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. The need to share trade secrets and other confidential information increases the risk that such trade secrets become known by our competitors, are inadvertently incorporated into the technology of others, or are disclosed or used in violation of these agreements. Given that our proprietary position is based, in part, on our know-how and trade secrets, a competitor’s discovery of our trade secrets or other unauthorized use or disclosure would impair our competitive position and may have an adverse effect on our business and results of operations. Enforcing a claim that a third party obtained illegally and is using trade secrets or confidential know-how is expensive, time consuming and unpredictable. The enforceability of confidentiality agreements may vary from jurisdiction to jurisdiction.
In addition, these agreements typically restrict the ability of our employees, consultants, collaborators, contractors and advisors to publish data potentially relating to our trade secrets, although our agreements may contain certain limited publication rights. Despite our efforts to protect our trade secrets, our competitors may discover our trade secrets, either through breach of our agreements with third parties, independent development or publication of information by any of our third-party collaborators. A competitor’s discovery of our trade secrets would impair our competitive position and have an adverse impact on our business.
| 47 |
| Table of Contents |
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed to us alleged trade secrets of their former employers or their former or current customers.
As is common in the biotechnology and pharmaceutical industries, certain of our employees were formerly employed by other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Moreover, we engage the services of consultants to assist us in the development of our products and product candidates, many of whom were previously employed at or may have previously been or are currently providing consulting services to, other biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may be subject to claims that these employees and consultants or we have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers or their former or current customers. Although we have no knowledge of any such claims being alleged to date, if such claims were to arise, litigation may be necessary to defend against any such claims. Even if we are successful in defending against any such claims, any such litigation could be protracted, expensive, a distraction to our management team, not viewed favorably by investors and other third parties and may potentially result in an unfavorable outcome.
If our patent term expires before or soon after our products are approved, or if manufacturers of generic or biosimilar drugs successfully challenge our patents, our business may be materially harmed.
Patents have a limited duration. In the United States, if all maintenance fees are timely paid, the natural expiration of a patent is generally 20 years from its earliest U.S. non-provisional filing date. Various extensions may be available, but the life of a patent, and the protection it affords, is limited. Even if patents covering our product candidates, their manufacture, or use are obtained, once the patent life has expired, we may be open to competition from competitive medications, including generic or biosimilar medications.
Depending upon the timing, duration and conditions of FDA marketing approval of our product candidates, one or more of our United States patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Act, and similar legislation in the European Union. The Hatch-Waxman Act permits a patent term extension of up to five years for a patent covering an approved product as compensation for effective patent term lost during product development and the FDA regulatory review process. The patent term extension cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval, and only one patent applicable to an approved drug may be extended. However, we may not receive an extension if we fail to apply within applicable deadlines, fail to apply prior to expiration of relevant patents or otherwise fail to satisfy applicable requirements. Moreover, the length of the extension could be less than we request. If we are unable to obtain patent term extension or the term of any such extension is less than we request, the period during which we can enforce our patent rights for that product will be shortened and our competitors may obtain approval to market competing products sooner than we expect. Also, the scope of our right to exclude during any patent term extension period may be limited or may not cover a competitor’s product or product use. As a result, our revenue from applicable products could be reduced, possibly materially.
Given the amount of time required for the development, testing and regulatory review of new drug candidates, patents protecting such drug candidates might expire before or shortly after such drug candidates are commercialized. As a result, our patents and patent applications may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours. Any of the foregoing could have a material adverse effect on our competitive position, business, financial conditions, results of operations and prospects.
Manufacturers of generic or biosimilar drugs may challenge the scope, validity, or enforceability of our patents in court or before a patent office, and we may not be successful in enforcing or defending those intellectual property rights and, as a result, may not be able to develop or market the relevant product exclusively, which would have a material adverse effect on any potential sales of that product. Upon the expiration of our issued patents or patents that may issue from our pending patent applications, we will not be able to assert such patent rights against potential competitors and our business and results of operations may be adversely affected.
| 48 |
| Table of Contents |
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
Our unregistered trademarks or trade names may be challenged, infringed, circumvented or declared generic or determined to be infringing on other marks. We may not be able to protect our rights to these trademarks and trade names, which we need to build name recognition among potential collaborators or customers in our markets of interest. At times, competitors may adopt trade names or trademarks similar to ours, thereby impeding our ability to build brand identity and possibly leading to market confusion. In addition, there could be potential trade name or trademark infringement claims brought by owners of other registered trademarks or trademarks that incorporate variations of our unregistered trademarks or trade names. Over the long term, if we are unable to successfully register our trademarks and trade names and establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively, and our business may be adversely affected. Our efforts to enforce or protect our proprietary rights related to trademarks, trade secrets, domain names, copyrights or other intellectual property may be ineffective and could result in substantial costs and diversion of resources and could adversely impact our financial condition or results of operations.
Our proprietary information may be lost, or we may suffer security breaches.
In the ordinary course of our business, we collect and store sensitive data, including intellectual property, clinical trial data, proprietary business information, personal data and personally identifiable information of our clinical trial subjects and employees, in our data centers and on our networks. The secure processing, maintenance and transmission of this information is critical to our operations. Despite our security measures, our information technology and infrastructure may be vulnerable to attacks by hackers or breached due to employee error, malfeasance or other disruptions. Although, to our knowledge, we have not experienced any such material security breach to date, any such breach could compromise our networks and the information stored there could be accessed, publicly disclosed, lost or stolen. Any such access, disclosure or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, significant regulatory penalties, disrupt our operations, damage our reputation and cause a loss of confidence in us and our ability to conduct clinical trials, which could adversely affect our reputation and delay our clinical development of our product candidates.
Risks Related to this Offering, the Securities Markets and Ownership of Our Common Stock and Warrants
The market prices of our common stock has been volatile and can fluctuate substantially, which could result in substantial losses for purchasers of our Units in this offering.
The market price of our common stock is highly volatile, and since our initial public offering in August 2021, the market price of our common stock has ranged from $1.41 to $6.95 per share. You may be unable to sell your shares of common stock at or above the offering price. The market price for our common stock and warrants may be volatile and subject to wide fluctuations in response to factors including the following:
| • |
| actual or anticipated fluctuations in our quarterly or annual operating results; |
| • |
| actual or anticipated changes in the pace of our corporate achievements or our growth rate relative to our competitors; |
| • |
| failure to meet or exceed financial estimates and projections of the investment community or that we provide to the public; |
| • |
| issuance of new or updated research or reports by securities analysts; |
| • |
| share price and volume fluctuations attributable to inconsistent trading volume levels of our common stock or warrants; |
| • |
| additions or departures of key management or other personnel; |
| • |
| disputes or other developments related to proprietary rights, including patents, litigation matters, and our ability to obtain patent protection for our technologies; |
| • |
| announcement or expectation of additional debt or equity financing efforts; |
| • |
| sales of our common stock or warrants by us, our insiders or our other stockholders; and |
| • |
| general economic, market or political conditions in the United States or elsewhere (including, without limitation, conditions arising out the COVID-19 pandemic). |
| 49 |
| Table of Contents |
In particular, the market prices of clinical-stage companies like ours have been highly volatile due to factors, including, but not limited to:
| • |
| any delay or failure in a clinical trial for our product candidates or receive approval from the FDA and other regulatory agents; |
| • |
| developments or disputes concerning our product candidate’s intellectual property rights; |
| • |
| our or our competitors’ technological innovations; |
| • |
| fluctuations in the valuation of companies perceived by investors to be comparable to us; |
| • |
| announcements by us or our competitors of significant contracts, acquisitions, strategic partnerships, joint ventures, capital commitments, new technologies or patents; |
| • |
| failure to complete significant transactions or collaborate with vendors in manufacturing our product; and |
| • |
| proposals for legislation that would place restrictions on the price of medical therapies. |
These and other market and industry factors may cause the market price and demand for our common stock and warrants to fluctuate substantially, regardless of our actual operating performance, which may limit or prevent investors from readily selling their shares of common stock or warrants and may otherwise negatively affect the liquidity of our common stock and warrants. In addition, the stock market in general, and Nasdaq and emerging growth companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. In the past, when the market price of a security has been volatile, holders of that security have instituted securities class action litigation against the company that issued the security. If any of our stockholders brought a lawsuit against us, we could incur substantial costs defending the lawsuit. Such a lawsuit could also divert the time and attention of our management.
The Warrants offered by this prospectus may not have any value.
The Warrants offered by this prospectus will be exercisable for five years from the date of initial issuance at an initial exercise price per share of common stock equal to 100% of the offering price of the Unit in this offering. There can be no assurance that the market price of our common stock will ever equal or exceed the exercise price of the Warrants. In the event that our common stock price does not exceed the exercise price of the Warrants during the period when the Warrants are exercisable, the Warrants may not have any value.
There is no public market for the Warrants or the Pre-Funded Warrants being offered in this offering.
There is no established public trading market for the Warrants or the Pre-Funded Warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply to list the Warrants or the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system, including Nasdaq. Without an active trading market, the liquidity of the Warrants and the Pre-Funded Warrants will be limited.
Neither a Warrant nor a Pre-Funded Warrant entitle the holder to any rights as common stockholders until the holder exercises the Warrant or Pre-Funded Warrant for a share of our common stock.
Until you acquire shares of our common stock upon exercise of your Warrants or Pre-Funded Warrants, your Warrants and your Pre-Funded Warrants will not provide you any rights as a common stockholder. Upon exercise of your Warrants or Pre-Funded Warrants, you will be entitled to exercise the rights of a common stockholder only as to matters for which the record date occurs after the exercise date.
| 50 |
| Table of Contents |
We are an “emerging growth company,” and will be able take advantage of reduced disclosure requirements applicable to “emerging growth companies,” which could make our common stock and Warrants less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act and, for as long as we continue to be an “emerging growth company,” we intend to take advantage of certain exemptions from various reporting requirements applicable to other public companies but not to “emerging growth companies,” including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. We could be an “emerging growth company” for up to five years, or until the earliest of (i) the last day of the first fiscal year in which our annual gross revenues exceed $1.07 billion, (ii) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter, or (iii) the date on which we have issued more than $1 billion in non-convertible debt during the preceding three year period.
We intend to take advantage of these reporting exemptions described above until we are no longer an “emerging growth company.” Under the JOBS Act, “emerging growth companies” can also delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not “emerging growth companies.”
We cannot predict if investors will find our common stock or warrants less attractive if we choose to rely on these exemptions. If some investors find our common stock or warrants less attractive as a result of any choices to reduce future disclosure, there may be a less active trading market for our common stock and warrants and the price of our common stock and warrants may be more volatile.
There may be limitations on the effectiveness of our internal controls, and a failure of our control systems to prevent error or fraud may materially harm our company. If we fail to remediate a material weakness, or if we experience material weaknesses in the future or otherwise fail to maintain an effective system of internal controls in the future, we may not be able to accurately or timely report our financial condition or results of operations, which may adversely affect investor confidence in us and, as a result, the value of our common stock and warrants.
Prior to the completion of our initial public offering in August 2021, we had been a private company with limited accounting personnel to adequately execute our accounting processes and limited supervisory resources with which to address our internal control over financial reporting. As a newly public company, we have not designed nor maintained an effective control environment as required of public companies under the rules and regulations of the SEC. Specifically, we lack a sufficient number of professionals with an appropriate level of accounting knowledge, training and experience to appropriately analyze, record and disclose accounting matters timely and accurately while maintaining appropriate segregation of duties.
Although we are not yet subject to the certification or attestation requirements of Section 404 of the Sarbanes-Oxley Act, management and our independent registered public accounting firm identified a material weakness in our internal control over financial reporting as we did not design or implement a control to ensure all material contracts or agreements are reviewed by accounting personnel to ensure they are accounted for and disclosed. A material weakness is a deficiency, or a combination of control deficiencies, in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis.
| 51 |
| Table of Contents |
Proper systems of internal controls over financial accounting and disclosure controls and procedures are critical to the operation of a public company. We may be unable to effectively establish such systems, especially in light of the fact that we expect to operate as a publicly reporting company. This would leave us without the ability to reliably assimilate and compile financial information about our company and significantly impair our ability to prevent error and detect fraud, all of which would have a negative impact on our company from many perspectives. Moreover, we do not expect that disclosure controls or internal control over financial reporting, even if established, will prevent all error and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, have been detected. Failure of our control systems to prevent error or fraud could materially adversely impact us.
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
The trading market for our common stock will depend in part on the research and reports that securities or industry analysts publish about us or our business. We do not currently have and may never obtain research coverage by securities and industry analysts. If no securities or industry analysts commence coverage of our company, the trading price for our stock would be negatively impacted. If we obtain securities or industry analyst coverage and if one or more of the analysts who covers us downgrades our stock or publishes inaccurate or unfavorable research about our business, our stock price would likely decline. If one or more of these analysts ceases coverage of us or fails to publish reports on us regularly, demand for our stock could decrease, which could cause our stock price and trading volume to decline.
Future sales of our common stock, warrants or securities convertible into our common stock may depress our stock price.
Sales of a substantial number of shares of our common stock, warrants or securities convertible into our common stock in the public market could occur at any time. These sales, or the perception in the market that the holders of a large number of shares intend to sell shares, could reduce the market price of our common stock and warrants. After this offering, we will have 12,773,073 outstanding shares of common stock, based on the number of shares outstanding as of the date of this prospectus. Moreover, we have registered all shares of common stock that we may issue after this offering under our equity compensation plans. If a large number of shares of our common stock, warrants or securities convertible into our common stock are sold in the public market after they become eligible for sale, the sales could reduce the trading price of our common stock and warrants and impede our ability to raise future capital.
Our failure to meet the continued listing requirements of Nasdaq could result in a delisting of our common stock and warrants.
If we fail to satisfy the continued listing requirements of Nasdaq, such as the corporate governance requirements or the minimum closing bid price requirement, Nasdaq may take steps to delist our common stock and warrants. Such a delisting would likely have a negative effect on the price of our common stock and warrants and would impair your ability to sell or purchase our common stock and warrants when you wish to do so. In the event of a delisting, we can provide no assurance that any action taken by us to restore compliance with listing requirements would allow our common stock and warrants to become listed again, stabilize the market price or improve the liquidity of our common stock and warrants, prevent our common stock and warrants from dropping below the Nasdaq minimum bid price requirement or prevent future non-compliance with Nasdaq’s listing requirements.
| 52 |
| Table of Contents |
Our directors, executive officers and principal stockholders will continue to have substantial control over us after this offering and could delay or prevent a change of corporate control.
Our directors, executive officers and holders of more than 5% of our common stock, together with their affiliates, beneficially own, in the aggregate, approximately 50.9% of our outstanding common stock. As a result, these stockholders, acting together, have the ability to significantly influence the outcome of matters submitted to our stockholders for approval, including the election of directors and any merger, consolidation or sale of all or substantially all of our assets. In addition, these stockholders, acting together, have the ability to control the management and affairs of our company. Accordingly, this concentration of ownership could harm the market price of our common stock and warrants by:
| • |
| delaying, deferring or preventing a change of control of us; |
| • |
| impeding a merger, consolidation, takeover or other business combination involving us; or |
| • |
| discouraging a potential acquiror from making a tender offer or otherwise attempting to obtain control of us. |
See “Principal Stockholders” below for more information regarding the ownership of our outstanding stock by our executive officers, directors and holders of more than 5% of our common stock, together with their affiliates.
Anti-takeover provisions contained in our certificate of incorporation and bylaws, as well as provisions of Delaware law, could impair a takeover attempt.
Our amended and restated certificate of incorporation, bylaws and Delaware law contain provisions which could have the effect of rendering more difficult, delaying or preventing an acquisition deemed undesirable by our board of directors. Our corporate governance documents include provisions:
| • |
| classifying our board of directors into three classes; |
| • |
| authorizing “blank check” preferred stock, which could be issued by our board of directors without stockholder approval and may contain voting, liquidation, dividend, and other rights superior to our common stock; |
| • |
| limiting the liability of, and providing indemnification to, our directors and officers; |
| • |
| limiting the ability of our stockholders to call and bring business before special meetings; |
| • |
| requiring advance notice of stockholder proposals for business to be conducted at meetings of our stockholders and for nominations of candidates for election to our board of directors; |
| • |
| controlling the procedures for the conduct and scheduling of board of directors and stockholder meetings; and |
| • |
| providing our board of directors with the express power to postpone previously scheduled annual meetings and to cancel previously scheduled special meetings. |
These provisions, alone or together, could delay or prevent hostile takeovers and changes in control or changes in our management.
As a Delaware corporation, we are also subject to provisions of Delaware law, including Section 203 of the Delaware General Corporation law, which prevents some stockholders holding more than 15% of our outstanding common stock from engaging in certain business combinations without approval of the holders of substantially all of our outstanding common stock.
Any provision of our amended and restated certificate of incorporation, bylaws or Delaware law that has the effect of delaying or deterring a change in control could limit the opportunity for our stockholders to receive a premium for their shares of our common stock or warrants, and could also affect the price that some investors are willing to pay for our common stock and warrants.
| 53 |
| Table of Contents |
We qualify as an “emerging growth company” as defined in the JOBS Act and will be able to avail ourselves of reduced disclosure requirements applicable to emerging growth companies, which could make our common stock and warrants less attractive to investors.
We qualify as an “emerging growth company” as defined in the JOBS Act, and we intend to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging growth companies” including certain reduced financial statement reporting obligations, reduced disclosure obligations about our executive compensation arrangements, exemptions from the requirement that we solicit non-binding advisory votes on executive compensation or golden parachute arrangements and exemption from the auditor’s attestation requirements of Section 404(b) of the Sarbanes-Oxley Act. We may take advantage of these reporting exemptions until we are no longer an “emerging growth company.” We will remain an “emerging growth company” until the earliest of (1) the last day of the fiscal year in which we have total annual gross revenue of $1 billion or more, (2) the last day of our fiscal year following the fifth anniversary of the date of the completion of this offering, (3) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years or (4) the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
Because management has broad discretion as to the use of the net proceeds from this offering, you may not agree with how we use them, and such proceeds may not be applied successfully.
Our management will have considerable discretion over the use of proceeds from this offering. We currently intend to use the net proceeds from this offering for external research and development expenses associated with the development of our DMT310 and DMT410 product candidates, manufacturing drug product for our clinical programs, with the balance primarily used to fund internal research and development expenses associated with all of our product candidates, working capital, capital expenditures and other general corporate purposes. In addition, a portion of the net proceeds may also be used to acquire or in-license, as applicable, product candidates, technologies, compounds, other assets or complementary businesses. However, our management will have broad discretion in the application of the net proceeds from this offering and could spend the proceeds in ways that do not necessarily improve our operating results or enhance the value of our common stock or warrants, or that you otherwise do not agree with. You will be relying on the judgment of our management concerning these uses and you will not have the opportunity, as part of your investment decision, to assess whether the proceeds are being used appropriately. The failure of our management to apply these funds effectively could, among other things, result in unfavorable returns and uncertainty about our prospects, each of which could cause the price of our common stock and warrants to decline.
If you purchase Units sold in this offering, you will incur immediate and substantial dilution of your investment in the shares of common stock comprising such Units.
If you purchase Units in this offering, you will experience substantial and immediate dilution in the pro forma net tangible book value per share after giving effect to this offering of $1.60 per share as of September 30, 2021, based on the public offering price of $2.25 per Unit, because the price that you pay will be substantially greater than the pro forma net tangible book value per share of the common stock in the Unit that you acquire. This dilution is due in large part to the fact that our earlier investors paid substantially less than the initial public offering price when they purchased shares of our capital stock. You will experience additional dilution upon exercise of the outstanding stock options and other equity awards that may be granted under our equity incentive plans, and when we otherwise issue additional shares of our common stock. For more information, see “Dilution.”
We have never paid dividends on our capital stock, and we do not anticipate paying any cash dividends in the foreseeable future.
We have never declared nor paid cash dividends on our capital stock. We currently intend to retain any future earnings to finance the operation and expansion of our business, and we do not expect to declare or pay any dividends in the foreseeable future. Consequently, stockholders must rely on sales of their common stock and warrants after price appreciation, which may never occur, as the only way to realize any future gains on their investment.
| 54 |
| Table of Contents |
Our amended and restated certificate of incorporation designates the Court of Chancery of the State of Delaware as the sole and exclusive forum for certain types of actions and proceedings that may be initiated by our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or other employees.
Our amended and restated certificate of incorporation requires that, unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware will, to the fullest extent permitted by law, be the sole and exclusive forum for each of the following:
| • |
| any derivative action or proceeding brought on our behalf; |
| • |
| any action asserting a claim for breach of any fiduciary duty owed by any director, officer or other employee of ours to the Company or our stockholders; |
| • |
| any action asserting a claim against us or any director or officer of ours arising pursuant to, or a claim against us or any of our directors or officers, with respect to the interpretation or application of any provision of, the DGCL, our certificate of incorporation or bylaws; or |
| • |
| any action asserting a claim governed by the internal affairs doctrine; |
provided, that, if and only if the Court of Chancery of the State of Delaware dismisses any of the foregoing actions for lack of subject matter jurisdiction, any such action or actions may be brought in another state court sitting in the State of Delaware.
The exclusive forum provision is limited to the extent permitted by law, and it will not apply to claims arising under the Securities Exchange Act of 1934, as amended, or the Exchange Act, or for any other federal securities laws which provide for exclusive federal jurisdiction.
Furthermore, Section 22 of the Securities Act creates concurrent jurisdiction for federal and state courts over all such Securities Act actions. Accordingly, both state and federal courts have jurisdiction to entertain such claims. To prevent having to litigate claims in multiple jurisdictions and the threat of inconsistent or contrary rulings by different courts, among other considerations, our amended and restated certificate of incorporation provides that the federal district courts of the United States of America will be the exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act. While the Delaware courts have determined that such choice of forum provisions are facially valid, a stockholder may nevertheless seek to bring such a claim arising under the Securities Act against us, our directors, officers, or other employees in a venue other than in the federal district courts of the United States of America. In such instance, we would expect to vigorously assert the validity and enforceability of the exclusive forum provisions of our amended and restated certificate of incorporation.
Although we believe this provision benefits us by providing increased consistency in the application of Delaware law in the types of lawsuits to which it applies, this provision may limit or discourage a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees, which may discourage such lawsuits against us and our directors, officers and other employees, and may result in increased costs for investors to bring a claim. Alternatively, if a court were to find the choice of forum provision contained in our amended and restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions, which could adversely affect our business and financial condition.
We note that there is uncertainty as to whether a court would enforce the provision and that investors cannot waive compliance with the federal securities laws and the rules and regulations thereunder. Although we believe this provision benefits us by providing increased consistency in the application of Delaware law in the types of lawsuits to which it applies, the provision may have the effect of discouraging lawsuits against our directors and officers.
| 55 |
| Table of Contents |
CAUTIONARY NOTE REGARDING FORWARD LOOKING STATEMENTS
Except for historical information, this prospectus contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Exchange Act of 1934, as amended. The forward-looking statements involve substantial risks and uncertainties. The forward-looking statements are contained principally in the sections entitled “Prospectus Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” “Business” and elsewhere in this prospectus. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “estimate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions intended to identify statements about the future. These statements speak only as of the date of this prospectus and involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking statements include, without limitation, statements about the following:
| • |
| our lack of operating history; |
| • |
| our plans to develop and commercialize our product candidates; |
| • |
| the timing of our planned clinical trials for DMT310 and our other product candidates; |
| • |
| the timing of our NDA submission for DMT310 for the treatment of acne vulgaris; |
| • |
| the timing of and our ability to obtain and maintain regulatory approvals for DMT310 and our other product candidates; |
| • |
| the clinical utility of our product candidates; |
| • |
| our commercialization, marketing and manufacturing capabilities and strategy; |
| • |
| our expectations about the willingness of healthcare professionals to use DMT310; |
| • |
| our intellectual property position; |
| • |
| our plans to in-license, acquire, develop and commercialize additional product candidates for other dermatological conditions to build a fully integrated dermatology company; |
| • |
| our expected use of proceeds from this offering; our competitive position and the development of and projections relating to our competitors or our industry; |
| • |
| our ability to identify, recruit and retain key personnel; |
| • |
| the impact of laws and regulations; |
| • |
| our expectations regarding the time during which we will be an emerging growth company under the JOBS Act; |
| • |
| our plans to identify additional product candidates with significant commercial potential that are consistent with our commercial objectives; and |
| • |
| our estimates regarding future revenue, expenses and needs for additional financing. |
| 56 |
| Table of Contents |
Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. You should refer to the “Risk Factors” section of this prospectus for a discussion of important factors that may cause our actual results to differ materially from those expressed or implied by our forward-looking statements. Moreover, we operate in an evolving environment. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties. As a result of these factors, we cannot assure you that the forward-looking statements in this prospectus will prove to be accurate. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. You should, however, review the factors and risks and other information we describe in the reports we will file from time to time with the SEC after the date of this prospectus.
You should read this prospectus and the documents that we reference in this prospectus and have filed as exhibits to the registration statement, of which this prospectus is a part, completely and with the understanding that our actual future results may be materially different from what we expect. We qualify all of our forward-looking statements by these cautionary statements.
INDUSTRY AND OTHER DATA
We obtained the industry, statistical and market data in this prospectus from our own internal estimates and research as well as from industry and general publications and research, surveys and studies conducted by third parties. All of the market data used in this prospectus involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such data. While we believe that each of these studies and publications is reliable, the industry in which we operate is subject to a high degree of uncertainty and risk due to a variety of important factors, including those described in the section titled “Risk Factors.” These and other factors could cause results to differ materially from those expressed in the estimates made by third parties and by us.
| 57 |
| Table of Contents |
USE OF PROCEEDS
We estimate that the net proceeds we will receive from the sale of our common stock and Warrants in this offering, after deducting underwriting discounts and commissions and estimated expenses payable by us and excluding the proceeds, if any, from the exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant, will be approximately $9.0 million (or $10.4 million if the underwriter exercises its option to purchase additional shares and/or Warrants in full and no sale of Units including a Pre-Funded Warrant), based on the public offering price of $2.25 per Unit (the last reported sale price of our common stock on Nasdaq on January 31, 2022). The public offering price per Unit will be determined between us, the underwriter and investors based on market conditions at the time of pricing and may be at a discount to the current market price of our common stock.
We intend to use substantially all of the net proceeds from this offering to complete FDA required studies to hold an End of Phase 2 meeting for the DMT310 acne program, initiate a DMT310 Phase 2 psoriasis trial, complete our DMT310 Phase 2 rosacea trial, advance the clinical development of DMT410 for the treatment of various aesthetic indications, manufacture drug product for our DMT310 Phase 3 acne program, and for working capital and other general corporate purposes.
We may also use a portion of the net proceeds of this offering for the acquisition or licensing, as the case may be, of additional technologies, other assets or businesses, or for other strategic investments or opportunities, although we currently have no understandings, agreements or commitments to do so.
Although we currently anticipate that we will use the net proceeds from this offering as described above, there may be circumstances where a reallocation of funds is necessary. The amounts and timing of our actual expenditures will depend upon numerous factors, including our sales and marketing and commercialization efforts, demand for our products, our operating costs and the other factors described under “Risk Factors” in this prospectus. Accordingly, our management will have flexibility in applying the net proceeds from this offering. An investor will not have the opportunity to evaluate the economic, financial or other information on which we base our decisions on how to use the proceeds.
The net proceeds from this offering, together with our cash and marketable securities, may not be sufficient for us to fund any of our product candidates through regulatory approval, and we will need to raise additional capital to complete the development and commercialization of our product candidates.
A $1.00 increase (decrease) in the assumed public offering price of $2.25 per Unit (the last reported sale price of our common stock on Nasdaq on January 31, 2022), would increase (decrease) the net proceeds to us from this offering by approximately $4.1 million (or approximately $6.1 million if the underwriters exercise their option to purchase additional shares and/or Warrants in full and no sale of Units including a Pre-Funded Warrant), assuming the number of Units offered by us, as set forth on the cover of this prospectus, remains the same and, after deducting underwriting discounts and commissions and estimated expenses payable by us and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant. An increase (decrease) of 100,000 in the number of Units offered by us in this offering, would increase (decrease) the net proceeds to us from this offering by approximately $200,000 (or approximately $1.6 million if the underwriters exercise their option to purchase additional shares and/or Warrants in full and no sale of Units including a Pre-Funded Warrant), assuming the public offering price of $2.25 per share (the last reported sale price of our common stock on Nasdaq on January 31, 2022), remains the same and after deducting underwriting discounts and commissions and estimated expenses payable by us and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant. The information above is illustrative only and will change based on the actual public offering price and other terms of this offering determined at pricing.
Pending our use of the net proceeds from this offering, we intend to invest the net proceeds in a variety of capital preservation investments, including short-term, investment-grade, interest-bearing instruments, and U.S. government securities.
| 58 |
| Table of Contents |
DIVIDEND POLICY
We have never declared or paid any cash dividends on our capital stock. We intend to retain future earnings, if any, to finance the operation of our business and do not anticipate paying any cash dividends in the foreseeable future. Any future determination related to dividend policy will be made at the discretion of our board of directors after considering our financial condition, results of operations, capital requirements, business prospects and other factors the board of directors deems relevant, and subject to the restrictions contained in any future financing instruments.
| 59 |
| Table of Contents |
CAPITALIZATION
The following table sets forth our cash and capitalization as of September 30, 2021:
| • |
| on an actual basis; |
| • |
| on an as adjusted basis, giving effect to our issuance and sale of 4,444,444 Units (assuming no sale of Units including a Pre-Funded Warrant) in this offering based on the public offering price of $2.25 per Unit (the last reported sale price of our common stock on Nasdaq on January 31, 2022), after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us and the receipt by us of the proceeds of such sale. |
You should read this table together with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our audited financial statements and related notes and unaudited interim condensed financial statements and related notes thereto included elsewhere in this prospectus.
|
| As of September 30, 2021 |
| |||||
|
| Actual |
|
| As Adjusted |
| ||
|
| (unaudited) |
|
| (unaudited) |
| ||
Cash |
| $ | 12,603,341 |
|
| $ | 21,583,340 |
|
Common stock, par value $0.0001, 90,000,000 shares authorized, 8,328,629 shares issued and outstanding, actual; 90,000,000 shares authorized and 12,773,073 shares issued and outstanding, as adjusted |
|
| 833 |
|
|
| 1,277 |
|
Additional paid-in capital |
|
| 45,919,140 |
|
|
| 54,898,695 |
|
Accumulated deficit |
| $ | (33,429,418 | ) |
| $ | (33,429,418 | ) |
Total stockholders' equity |
|
| 12,490,555 |
|
|
| 21,470,554 |
|
Total capitalization |
| $ | 12,490,555 |
|
| $ | 21,470,554 |
|
The table above is based on 8,328,629 shares of our common stock outstanding as of September 30, 2021 and does not include:
| • |
| 523,199 shares of common stock issuable upon exercise of stock options outstanding, at a weighted-average an exercise price of $5.84 per share; |
| • |
| 2,957,142 shares of common stock issuable upon exercise of warrants issued in our initial public offering, at an exercise price of $7.00 per share; |
|
|
|
|
• |
| 134,515 shares of common stock issuable upon exercise of other warrants outstanding, at a weighted-average exercise price of $13.33 per share; and |
| • |
| 1,125,014 shares of our common stock that are available for future issuance under our 2021 Plan, or shares that will become available under our 2021 Plan. |
A $1.00 increase or decrease in the assumed public offering price of $2.25 per Unit (the last reported sale price of our common stock on Nasdaq on January 31, 2022), would increase or decrease the pro forma as adjusted amount of each of cash and cash equivalents, additional paid-in capital, total stockholders’ equity (deficit) and total capitalization by approximately $4.1 million, assuming that the number of Units offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant. An increase or decrease of 100,000 Units in the number of Units offered by us, as set forth on the cover page of this prospectus, would increase or decrease the pro forma as adjusted amount of each of cash and cash equivalents, additional paid-in capital, total stockholders’ equity (deficit) and total capitalization by approximately $200,000, assuming no change in the assumed public offering price per Unit and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant.
| 60 |
| Table of Contents |
DILUTION
If you invest in our securities in this offering, your ownership interest will be immediately diluted to the extent of the difference between the public offering price per share of common stock included in each Unit (attributing no value to the Warrants and assuming no sale of Units including a Pre-Funded Warrant) and the as adjusted net tangible book value per share of our common stock after this offering. As of September 30, 2021, we had a historical net tangible book value of approximately $11.4 million, or $1.37 per share of common stock. Our historical net tangible book value per share represents total tangible assets less total liabilities, divided by the number of shares of our common stock outstanding as of September 30, 2021.
After giving further effect to the sale of shares of common stock included in each Unit in this offering at a public offering price of $2.25 per share of common stock included in each Unit (the last reported sale price of our common stock on Nasdaq on January 31, 2022), and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant, our as adjusted net tangible book value as of September 30, 2021 would have been approximately $20.4 million, or approximately $1.60 per share of common stock. This amount represents an immediate increase in actual book value of $0.23 per share to our existing stockholders and immediate dilution of approximately $0.65 per share to new investors in this offering (attributing no value to the Warrants and assuming no sale of Units including a Pre-Funded Warrant). We determine dilution by subtracting the as adjusted net tangible book value per share after this offering from the amount of cash that a new investor paid for a share of common stock in this offering. The following table illustrates this dilution:
Offering price per unit |
|
|
|
| $ | 2.25 |
| |
Historical net tangible book value per share as of September 30, 2021 |
| $ | 1.37 |
|
|
|
|
|
Pro forma increase in net tangible book value per share attributable to new investors |
| $ | 0.23 |
|
|
|
|
|
Pro forma as adjusted net tangible book value per share after this offering |
|
|
|
|
| $ | 1.60 |
|
Dilution per share to new investors purchasing common stock in this offering |
|
|
|
|
| $ | 0.65 |
|
The dilution information discussed above is illustrative only and will change based on the actual public offering price and other terms of this offering determined at pricing. A $1.00 decrease in the assumed public offering price of $2.25 per Unit (the last reported sale price of our common stock on Nasdaq on January 31, 2022), would decrease our as adjusted net tangible book value per share after this offering by $0.10 and increase dilution per share to new investors purchasing Units in this offering by $0.02, assuming that the number of Units offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant. A $1.00 increase in the assumed public offering price of $2.25 per Unit (the last reported sale price of our common stock on Nasdaq on January 31, 2022), would increase our as adjusted net tangible book value per share after this offering by $0.55 and dilution per share to new investors purchasing Units in this offering by $1.33, assuming that the number of Units offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant. An increase or decrease of 100,000 Units in the number of Units offered by us, as set forth on the cover page of this prospectus, would increase or decrease our as adjusted net tangible book value per share after this offering by $0.23 and dilution per share to new investors purchasing Units in this offering by $0.65, assuming no change in the assumed public offering price per Unit and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and assuming no exercise of the Warrants issued as part of the Units and assuming no sale of Units including a Pre-Funded Warrant.
If the underwriters exercise their option to purchase additional shares of our common stock and/or Warrants in full and assuming no sale of Units including a Pre-Funded Warrant, the pro forma as adjusted net tangible book value after this offering would be $1.62 per share, the increase in pro forma net tangible book value per share would be $0.25 and dilution per share to new investors would be $0.63 per share.
The above discussion and table are based on 8,328,629 shares of our common stock outstanding as of September 30, 2021 and excludes as of such date:
| • |
| 523,199 shares of common stock issuable upon exercise of stock options outstanding, at a weighted-average an exercise price of $5.84 per share; |
| • |
| 2,957,142 shares of common stock issuable upon exercise of warrants issued in our initial public offering, at an exercise price of $7.00 per share; |
|
|
|
|
• |
| 134,515 shares of common stock issuable upon exercise of other warrants outstanding, at a weighted-average exercise price of $13.33 per share; and |
| • |
| 1,125,014 shares of our common stock that are available for future issuance under our 2021 Plan, or shares that will become available under our 2021 Plan. |
| 61 |
| Table of Contents |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
You should read the following discussion and analysis of our financial condition and results of operations together with our financial statements and the related notes and other financial information included elsewhere in this prospectus. Some of the information contained in this discussion and analysis or set forth elsewhere in this prospectus, including information with respect to our plans and strategy for our business, includes forward-looking statements that involve risks and uncertainties. You should review the “Risk Factors” section of this prospectus for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.
Overview
We are a clinical-stage medical dermatology company focused on identifying, developing and commercializing innovative pharmaceutical product candidates for the treatment of medical and aesthetic skin diseases and conditions we believe have significant unmet needs.
Dermatological diseases such as acne vulgaris (or acne), psoriasis vulgaris (or psoriasis), papulopustular rosacea (or rosacea), hyperhidrosis, and various aesthetic indications affect millions of people worldwide each year and may negatively impact their quality of life and emotional well-being. While there are multiple current treatment options for these indications on the market, we believe that most have significant drawbacks, including underwhelming treatment results, cumbersome application regimens and varying negative side effects leading to a lack of patient compliance. While a majority of these indications are first treated with topical products, many patients frequently switch treatments or discontinue treatment altogether due to patient dissatisfaction with slow and modest response rates, early onset of negative side effects, onerous application schedules and typically long duration of therapy. For diseases like psoriasis, a small percentage of patients may be candidates for biologic or systemic therapies, but these patients are typically required to try topical or oral treatment options prior to qualifying for these expensive systemic therapies. Given the limitations with current topical therapies and the restricted usability of systemic therapies, we believe there is a significant opportunity to address the needs of frustrated patients searching for effective topical products that satisfy their dermatological and lifestyle needs.
Our two product candidates, DMT310 and DMT410, both incorporate our proprietary, multifaceted, Spongilla technology to topically treat a variety of dermatological conditions. Our Spongilla technology is a multifactorial, naturally-derived product that is processed from a wholly naturally grown freshwater sponge, Spongilla lacustris or Spongilla, which is processed into a powder and is mixed with a fluidizing agent immediately prior to application by the patient to form an easily applicable paste. Spongilla is a unique freshwater sponge that only grows in commercial quantities in select regions of the world and under specific environmental conditions, all of which give it its distinctive anti-microbial, anti-inflammatory, and mechanical properties. The combination of these ideal environmental conditions, the proprietary harvesting protocols developed with our supplier, and our post-harvest processing procedures optimize the mechanical component as well as the chemical components of Spongilla for product candidates with multiple mechanisms of action for the treatment of inflammatory skin conditions.
Our lead product candidate DMT310, utilizes our Spongilla technology for a once-weekly treatment of a variety of skin diseases. Our initial focus is the treatment of acne vulgaris. However, due to the multiple mechanisms of action and anti-inflammatory effect seen with DMT310, we have moved into clinical trials of two additional indications, psoriasis and rosacea. In October 2021, we completed a Phase 1b proof of concept, or POC, trial in psoriasis and in November 2021 we initiated a Phase 2 clinical trial of DMT310 for the treatment of rosacea.
Our second product candidate utilizing our Spongilla technology is our combination treatment, DMT410. DMT410 consists of one treatment of our proprietary sponge powder followed by topical application of botulinum toxin for delivery into the dermis. We first tested this program in a Phase 1 POC trial of axillary hyperhidrosis patients, which was completed in July 2019. We reported that 80% of patients achieved a reduction in gravimetric sweat production greater than 50%, four weeks after treatment. Based on the results of this trial, we initiated a Phase 1 POC trial of DMT410 for the intradermal treatment of multiple aesthetic skin conditions. In November 2021, we announced top-line results from this trial where we obtained data that we believe warrants further investigation of this program. We believe these two DMT410 trials indicate we have been able to topically deliver botulinum toxin into the dermis subsequent to the application of our Spongilla technology.
| 62 |
| Table of Contents |
We believe our Spongilla technology platform will enable us to develop and formulate singular and combination products that are able to target topical delivery of chemical compounds into the dermis for maximum treatment effect for a variety of inflammatory skin diseases. One mechanism of our technology is its mechanical ability to allow for the intradermal delivery of a variety of large and small molecules through microchannels to a targeted treatment site via topical application. In addition to its mechanical components, the Spongilla technology also utilizes multiple naturally occurring chemical compounds that we believe have demonstrated in-vitro anti-microbial, and anti-inflammatory properties. We believe the combination of these mechanical and chemical components make our platform versatile for the treatment of a wide variety of medical and aesthetic skin conditions and diseases.
We have a limited operating history. Since our inception, our operations have focused on developing DMT310 and DMT410, organizing and staffing our company, raising capital, establishing our supply chain and manufacturing processes, further characterizing the multiple mechanisms of action of our Spongilla technology, building an intellectual property portfolio, and conducting non-clinical and clinical trials. We do not have any product candidates approved for marketing and have not generated any revenue from product sales. We have funded our operations primarily through the sale of our equity securities and debt securities. Since inception, we have raised an aggregate of approximately $48 million of gross proceeds from the sale of our debt and equity securities, including the securities sold in our IPO.
We have not generated any revenue to date and have incurred significant operating losses. Our net loss was $4.7 million and $3.2 million for the years ended December 31, 2019 and 2020, respectively, and as of December 31, 2020, we had an accumulated deficit of $28.1 million. We incurred a net loss of $5.3 million and $2.8 million for the nine months ended September 30, 2021 and 2020, respectively, and had an accumulated deficit of approximately $33.4 million as of September 30, 2021. We expect to continue to incur significant expenses and operating losses for the foreseeable future. We anticipate that our expenses will increase significantly in connection with our ongoing activities, as we:
| • |
| complete development of DMT310 for the treatment of acne, including non-clinical studies and Phase 3 clinical trials; |
| • |
| prepare and file for regulatory approval of DMT310 for the treatment of moderate to severe acne; |
| • |
| continue development of DMT310 for the treatment of rosacea, including a Phase 2 clinical trial and Phase 3 clinical trials; |
| • |
| continue development of DMT310 for the treatment of psoriasis, including a Phase 2 clinical trial and Phase 3 clinical trials; |
| • |
| identify a botulinum toxin partner for DMT410 for the treatment of aesthetic and medical skin conditions; |
| • |
| prepare for commercialization of DMT310, if approved, including the hiring of sales and marketing personnel; |
| • |
| begin to manufacture our product candidates for Phase 2 and Phase 3 trials and commercial sale; |
| • |
| hire additional research and development and selling, general and administrative personnel; |
| • |
| maintain, expand and protect our intellectual property portfolio; and |
| • |
| incur additional costs associated with operating as a public company following the completion of this offering. |
We will likely need additional financing after this offering to support our operations. We may seek to fund our operations through public or private equity or debt financings or other sources. Adequate additional financing may not be available to us on acceptable terms, or at all. Our failure to raise capital when needed or on favorable terms would have a negative impact on our financial condition and our ability to pursue our business strategy. We will need to generate significant revenues to achieve profitability, and we may never do so.
| 63 |
| Table of Contents |
Trends and Uncertainties - COVID-19
We are subject to risks and uncertainties as a result of the COVID-19 pandemic. The extent of the impact of the COVID-19 pandemic on our business is highly uncertain and difficult to predict, as the responses that we, other businesses and governments are taking continue to evolve. Furthermore, capital markets and economies worldwide have also been negatively impacted by the COVID-19 pandemic, and it is possible that it could cause a local and/or global economic recession. Policymakers around the globe have responded with fiscal policy actions to support the healthcare industry and economy as a whole. The magnitude and overall effectiveness of these actions remain uncertain.
The severity of the impact of the COVID-19 pandemic on our business will depend on a number of factors, including, but not limited to, the duration and severity of the pandemic and the extent and severity of the impact on our service providers, suppliers, contract research organizations and our preclinical and clinical trials, all of which are uncertain and cannot be predicted.
As of the date of this prospectus, the extent to which the COVID-19 pandemic may in the future materially impact our financial condition, liquidity or results of operations is uncertain.
Components of Results of Operations
Revenue
We have not generated any revenue since inception and do not expect to generate any revenue from the sale of products in the near future until we obtain regulatory approval of, and commercialize, our product candidates.
Operating Expenses
Research and Development Expenses
Research and development activities are central to our business model. Product candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of late-stage clinical trials. We expect research and development costs to increase significantly for the foreseeable future as our pipeline of product candidates progress further into clinical trials. However, we do not believe it is possible at this time to accurately project total program-related expenses to reach commercialization based on numerous factors. In addition, there are numerous unknown expenses related to the commercialization of our product candidates including continued regulatory requirements, many of which cannot be determined with accuracy at this time.
Research and development expenses consist of expenses incurred in connection with the development of our product candidates. We expense development costs as incurred. These expenses include:
| • |
| expenses incurred under agreements with CROs, as well as investigative sites and consultants that conduct our clinical trials and preclinical studies; |
| • |
| manufacturing and supply scale-up expenses and the cost of acquiring and manufacturing preclinical and clinical trial supply and commercial supply, including manufacturing validation batches; and |
| • |
| outsourced laboratory services, including materials and supplies used to support our research and development activities, including payments made for license fees and milestone payments. |
| 64 |
| Table of Contents |
The successful development of our product candidates is highly uncertain. At this time, we cannot reasonably estimate or know the nature, timing and costs of the efforts that will be necessary to complete the remainder of the development of, or when, if ever, material net cash inflows may commence from our product candidates. This uncertainty is due to the numerous risks and uncertainties associated with the duration and cost of clinical trials, which vary significantly over the life of a project as a result of many factors, including:
| • |
| the number of clinical sites included in the clinical trials; |
| • |
| the length of time required to enroll suitable patients; |
| • |
| the size of patient populations participating in the clinical trials; |
| • |
| the number of doses a patient receives; |
| • |
| the duration of patient follow-ups; |
| • |
| the development state of the product candidates; and |
| • |
| the efficacy and safety profile of the product candidates. |
Our expenditures are subject to additional uncertainties, including the terms and timing of regulatory approvals, and the expense of filing, prosecuting, defending, and enforcing any patent claims or other intellectual property rights. We may never succeed in achieving regulatory approval for our product candidates. We may obtain unexpected results from our clinical trials. We may elect to discontinue, delay, or modify clinical trials of our product candidates. A change in the outcome of any of these variables with respect to the development of a product candidate could mean a significant change in the costs and timing associated with the development of that product candidate. For example, if the FDA or other regulatory authorities were to require us to conduct clinical trials beyond those that we currently anticipate, or if we experience significant delays in enrollment in any of our clinical trials, we could be required to expend significant additional financial resources and time on the completion of clinical development. Product commercialization will take several years and millions of dollars in development costs.
General and Administrative Expenses
General and administrative expenses consist principally of salaries and related costs for personnel in executive and administrative functions, travel expenses and recruiting expenses. Other general and administrative expenses include stock based compensation expenses, professional fees for legal, accounting and tax-related services, insurance costs, as well as payments made to consultants. We expense all general and administrative expenses as incurred.
We anticipate that our general and administrative expenses will increase as a result of increased payroll, expanded infrastructure and higher consulting, legal and tax-related services associated with maintaining compliance with stock exchange listing and SEC requirements, accounting and investor relations costs, and director and officer insurance premiums associated with being a public company. As a result, we expect to report higher general and administrative expenses in 2022.
Interest Expense
Interest expense has consisted primarily of interest expense on our previously outstanding convertible debt and loan with Silicon Valley Bank, amortization of debt discount costs, and interest on milestone payments under the License Amendment and Settlement Agreement.
Income Taxes
From inception until March 24, 2021, we operated as a limited liability company taxed as a partnership. Therefore, any income tax liability or benefit through that date accrued to our members. Since March 24, 2021, we have operated as a C-Corporation and account for income taxes under the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements. Under this method, deferred tax assets and liabilities are determined based on the differences between the financial statements and tax basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. We recognize net deferred tax assets to the extent that we believe these assets are more likely than not to be realized. In making such a determination, we consider all available positive and negative evidence. We record the difference between the benefit recognized and measured pursuant to the accounting guidance on accounting for uncertain tax positions taken or expected to be taken on our tax return. We record uncertain tax positions on the basis of a two-step process whereby (1) management determines whether it is more likely than not that the tax positions will be sustained on the basis of the technical merits of the position and (2) for those tax positions that meet the more-likely-than-not recognition threshold, management recognizes the largest amount of tax benefit that is more than 50% likely to be realized upon ultimate settlement with the related tax authority. The liabilities are adjusted in light of changing facts and circumstances, such as the outcome of tax audits. To the extent that the assessment of such tax positions changes, the change in estimate is recorded in the period in which the determination is made. As of December 31, 2020 and 2019, we had no uncertain income tax positions.
| 65 |
| Table of Contents |
Critical Accounting Policies and Significant Judgments and Estimates
We have based our management’s discussion and analysis of financial condition and results of operations on our financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States. The preparation of these financial statements requires us to make estimates that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements as well as the reported revenues and expenses during the reporting periods. On an ongoing basis, we evaluate our estimates and judgments, including those related to clinical development expenses and the fair value of units and common stock. We base our estimates on historical experience and on various other factors that we believe to be appropriate under the circumstances. Actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully discussed in the notes to our audited financial statements appearing at the end of this prospectus, we believe that the following accounting policies are critical to the process of making significant judgments and estimates in the preparation of our financial statements.
Research and Development Expenses
As part of the process of preparing our financial statements, we are required to record actual research and development expenses and to estimate accrued research and development expenses, current assets and other current liabilities. This process involves reviewing open contracts and commitments, communicating with our personnel to identify services that have been performed for us and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of the actual cost. The majority of our service providers invoice us monthly in arrears for services performed or when contractual milestones are met. We make estimates of our accrued research and development expenses, current assets and other current liabilities as of each balance sheet date in our financial statements based on facts and circumstances known to us at that time. Example of estimated accrued research and development expenses, prepaid assets and other current liabilities include fees paid to contract manufacturers made in connection with the manufacturing of clinical trials materials and contract research organizations made in connection the performance of clinical trials on our behalf.
We base our expenses related to clinical manufacturing and clinical trials on our estimates of the services performed pursuant to contracts with the entities performing those services on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. Payments under these types of contracts depend heavily upon the successful completion of many separate tasks involved in the manufacturing of drug product and the performance of clinical trials. In accruing service fees, we estimate the time period over which services will be performed, and the actual services performed in each period. If our estimates of the status and timing of services performed differs from the actual status and timing of services performed, we may report amounts that are too high or too low in any particular period. To date, there have been no material differences from our estimates to the amount actually incurred.
Fair Value of Common Stock and Stock-Based Compensation
Stock-based compensation cost is measured at the grant date based on the fair value of the award and is recognized as expense over the requisite service period, which is generally the vesting period. Our policy permits the valuation of stock-based awards granted to non-employees to be measured at fair value at the grant date.
| 66 |
| Table of Contents |
Determining the appropriate fair value of share-based awards requires the use of subjective assumptions, including the fair value of our common shares for awards prior to our IPO, and for options, the expected life of the option and expected share price volatility. We use the Black-Scholes option pricing model to value our option awards. The assumptions used in calculating the fair value of share-based awards represents our best estimates and involve inherent uncertainties and the application of judgment. As a result, if factors change and management uses different assumptions, share-based compensation expense could be materially different for future awards.
Prior to our initial public offering in August 2021, we were required to periodically estimate the fair value of our common stock when issuing stock options and computing their estimated stock-based compensation expense. The fair value of common stock was determined by management on a periodic basis, with the assistance of an independent third-party valuation firm. The assumptions underlying these valuations represented management’s best estimates, which involved inherent uncertainties and the application of significant levels of management judgment.
In order to determine the fair value, we considered, among other things, contemporaneous transactions involving the sale of our common stock to unrelated third parties, the lack of marketability of our common stock and the market performance of comparable publicly traded companies.
Fair Value of Common Units
In the absence of a public trading market for our common units prior to our IPO, on each grant date, we developed an estimate of the fair value of our common units underlying the unit grants. We estimated the fair value of our common units by referencing arms-length transactions with third parties, inclusive of the common units underlying which occurred on or near the valuation date(s).
During the year ended December 31, 2019, we issued 161,142 Class B Common Units to employees, officers, and directors. During the year ended December 31, 2020, we issued 2,926 Class B Common Units to consultants and 2,643 Class B Common Units to an employee. All of the Class B Common Units issued in 2019 and 2020 represent a profits interest, as that term is defined in Revenue Procedure 93-27, 1993-2 C.B. 343, as clarified by Revenue Procedure 2001-43, 2001-2 C.B. 191. For performance-based awards, if and when the achievement of the predetermined performance criteria become probable, expense will be recognized. To date unit based compensation expense has not been material. The assumptions used in calculating the fair value of unit-based awards represents management’s best estimates and involve inherent uncertainties and the application of management’s judgment. As a result, if factors change and management uses different assumptions, unit-based compensation expense could be materially different for future awards. All of such securities were exchanged for shares of our capital units in connection with our conversion from a limited liability company to a corporation in March 2021.
Results of Operations for the Nine Months Ended September 30, 2021 and 2020
The following table summarizes our results of operations for the nine months ended September 30, 2021 and 2020:
|
| Nine Months Ended September 30, |
|
|
|
| ||||||
|
| 2021 |
|
| 2020 |
|
| Difference |
| |||
Operating expenses: |
|
|
|
|
|
|
|
|
| |||
Research and development |
| $ | 2,347,564 |
|
| $ | 1,493,520 |
|
| $ | 854,044 |
|
General and administrative |
|
| 2,956,444 |
|
|
| 1,187,906 |
|
|
| 1,768,538 |
|
Total operating expenses |
|
| 5,304,008 |
|
|
| 2,681,426 |
|
|
| 2,622,582 |
|
Loss from operations |
|
| (5,304,008 | ) |
|
| (2,681,426 | ) |
|
| (2,622,582 | ) |
Interest expense, net |
|
| 45,613 |
|
|
| 158,791 |
|
|
| (113,178 | ) |
Net loss |
| $ | (5,349,621 | ) |
| $ | (2,840,217 | ) |
| $ | (2,509,404 | ) |
Research and Development Expenses
Research and development expenses increased by $0.8 million from $1.5 million for the nine months ended September 30, 2020 to $2.3 million for the nine months ended September 30, 2021. The increase was the result of increased salaries, benefits, and stock-based compensation of $0.9 million, increased manufacturing costs of $0.2 million, and increased nonclinical studies of $0.1 million, offset by $0.4 million in decreased clinical costs.
General and Administrative Expenses
General and administrative expenses increased by $1.8 million from $1.2 million for the nine months ended September 30, 2020 to $3.0 million for the nine months ended September 30, 2021. The increase resulted from increased legal and professional fees of $0.7 million, increased salaries, benefits, and stock-based compensation expense of $0.9 million and increased insurance costs of $0.2 million.
| 67 |
| Table of Contents |
Other Income and Expenses
Other income and expenses decreased by $113,178 from $158,791 for the nine months ended September 30, 2020 to $45,613 for the nine months ended September 30, 2021. The decrease was the result of decreased debt discount amortization of $74,167 and decreased interest expense of $39,011.
Cash Flows
The following table summarizes our cash flows from operating and financing activities:
|
| For the Nine Months Ended |
| |||||
|
| September 30, |
| |||||
|
| 2021 |
|
| 2020 |
| ||
|
| (unaudited) |
|
| (unaudited) |
| ||
Total net cash provided by (used in): |
|
|
|
|
|
| ||
Operating activities |
| $ | (3,889,483 | ) |
| $ | (3,492,567 | ) |
Financing activities |
|
| 15,962,424 |
|
|
| 2,253,811 |
|
Increase (decrease) in cash |
|
| 12,072,941 |
|
|
| (1,238,756 | ) |
Cash used in operations of $3.9 million for the nine months ended September 30, 2021 was the result of the net loss of $5.3 million and an increase in prepaid expenses and other current assets of $1.0 million, offset by non-cash stock-based compensation of $1.4 million and an increase in accounts payable and accrued and other current liabilities of $1.0 million.
Cash used in operations of $3.5 million for the nine months ended September 30, 2020 was the result of the net loss of $2.8 million, payment of $0.5 million for a license and settlement liability, as well as an increase in accounts payable and accrued and other current liabilities of $0.2 million.
Financing Activities
Cash provided by financing activities of $16.0 million for the nine months ended September 30, 2021 was the result of net proceeds of $15.4 million from our initial public offering, $1.6 million from the issuance of convertible subordinated promissory notes, proceeds of $0.6 million from the issuance of Series 1d Preferred Units, offset by a $1.0 million payment for the redemption of 5,221,156 shares of Series 1c preferred stock and $0.6 million of principal and final payments on debt.
Cash provided by financing activities of $2.3 million for the nine months ended September 30, 2020 was the result of proceeds of $2.3 million from the issuance of convertible subordinated promissory notes, proceeds of $0.3 million from the issuance of Series 1c Preferred Units, and proceeds of $0.1 million from the Paycheck Protection Plan loan, offset by $0.5 million of principal payments on debt.
Results of Operations for the Year Ended December 31, 2020 and 2019
The following table summarizes our results of operations for the years ended December 31, 2020 and 2019:
|
| Year Ended December 31, |
|
|
|
| ||||||
|
| 2020 |
|
| 2019 |
|
| Difference |
| |||
Operating expenses: |
|
|
|
|
|
|
|
|
| |||
Research and development |
| $ | 1,607,819 |
|
| $ | 2,623,280 |
|
| $ | (1,015,461 | ) |
General and administrative |
|
| 1,565,034 |
|
|
| 1,847,328 |
|
|
| (282,294 | ) |
Total operating expenses |
|
| 3,172,853 |
|
|
| 4,470,608 |
|
|
| (1,297,755 | ) |
Loss from operations |
|
| (3,172,853 | ) |
|
| (4,470,608 | ) |
|
| 1,297,755 |
|
Interest expense, net |
|
| 63,677 |
|
|
| 250,748 |
|
|
| (187,071 | ) |
Net loss |
| $ | (3,236,530 | ) |
| $ | (4,721,356 | ) |
| $ | 1,484,826 |
|
Research and Development Expenses
Research and development expenses were $1,607,819 for the year ended December 31, 2020, compared to $2,623,280 for the year ended December 31, 2019. The decrease of $1,015,461 is primarily related to product manufacturing costs of $323,786 and the $730,962 estimated fair value of Series 1c Preferred Units issued in connection with the Villani License Agreement (defined below) that were not incurred in 2020, offset by increased clinical trial costs of $195,937 in 2020. Of the $1,607,819 in research and development expenses incurred during the year ended December 31, 2020, $1,396,308, was associated with activities related to clinical trials and $107,636 was related to product storage and stability testing. These activities included, but were not limited to, our Phase 2b clinical trial of DMT310 for the once-weekly treatment of moderate-to-severe acne as well as an open-label, 10 patient, Phase 1b clinical trial of DMT410 for the treatment of moderate-to-severe glabellar, lateral canthal and forehead lines along with multiple secondary aesthetic endpoints.
| 68 |
| Table of Contents |
We do not track research and development expenses by product candidate. Instead, because DMT310 and DMT410 utilize the same drug product developed from our Spongilla technology, we track research and development expenses based on the drug product used in a clinical trial.
Of the $2,623,280 in research and development incurred during the year ended December 31, 2019, $730,962 related to the estimated fair value of Series 1c Preferred Units issued in connection with the Villani License Agreement, $1,200,371 was associated with activities related to clinical trials, $323,786 was associated with product manufacturing costs and $160,853 was related to product storage and stability testing. These activities included, but were not limited to, our Phase 2a clinical trial of DMT310 for the once-weekly treatment of moderate-to-severe acne as well as an open-label, 10 patient, Phase 1b proof of concept clinical trial of DMT410 for the treatment of axillary hyperhidrosis.
General and Administrative Expenses
General and administrative expenses were $1,565,034 for the year ended December 31, 2020, compared to $1,847,328 for the year ended December 31, 2019. The decrease of $282,294 was primarily attributable to legal and professional fees related to the Villani License Agreement that were not incurred in 2020.
Cash Flows
The following table summarizes our cash flows from operating and financing activities:
|
| For the Year Ended |
| |||||
|
| December 31, |
| |||||
|
| 2020 |
|
| 2019 |
| ||
|
|
|
|
|
|
| ||
Total net cash provided by (used in): |
|
|
|
|
|
| ||
Operating activities |
| $ | (4,028,541 | ) |
| $ | (3,922,272 | ) |
Financing activities |
|
| 2,567,139 |
|
|
| 4,734,461 |
|
Increase (decrease) in cash |
|
| (1,461,402 | ) |
|
| 812,189 |
|
Cash used in operations of $4,028,541 in 2020 was primarily the result of the net loss of $3,236,530, the payment of a $500,000 license and settlement liability, a decrease in accounts payable and accrued liabilities of $254,471, and the gain from forgiveness of the Payroll Protection Plan of $133,592, offset by debt amortization of $97,104.
Cash used in operations of $3,922,272 in 2019 was primarily the result of the net loss of $4,721,356 and the payment of a $250,000 license and settlement liability, offset by the estimated fair value of $730,962 related to Series 1c Preferred Units issued in connection with the Villani License Agreement, debt discount amortization expense of $141,753, and decreases in accounts payable and accrued liabilities of $124,956.
Financing Activities
Cash provided by financing activities was $2,567,139 and $4,734,461 during the year ended December 31, 2020 and 2019, respectively. The primary reason for the decrease of $2,167,322 during 2020 was attributable to the issuance of 44,767,474 preferred units in 2019 for aggregate net proceeds of $5,510,630 (which includes the conversion of $150,000 in principal amount of convertible note from a Managing Member), compared to the issuance of 1,785,714 preferred units for an aggregate net proceeds of $250,000, and the issuance of the Notes for an aggregate net proceeds of $2,974,982 in 2020, as well as the proceeds in 2020 from the Payroll Protection Plan loan for net proceeds of $133,592.
Liquidity and Capital Resources
Since our inception, we have not generated any revenue or commercialized any products. As of September 30, 2021, our cash totaled $12.6 million and we had an accumulated deficit of $33.4 million. For the year ended December 31, 2020 and the nine months ended September 30, 2021, we used cash of $4.0 million and $3.9 million, respectively, in operations. As a result of our initial public offering of common stock and warrants in August 2021 for net proceeds to us of $15.4 million, our current cash balances are expected to fund operations into August 2022. We anticipate that we will continue to incur net losses for the foreseeable future.
| 69 |
| Table of Contents |
Historically, our principal sources of cash have included proceeds from the issuance of common and preferred equity and proceeds from the issuance of debt. Our principal uses of cash have included cash used in operations (including clinical development of our product candidates and general and administrative expenses) and payments for license rights. We expect that the principal uses of cash in the future will be for continuing operations, funding of research and development, and general working capital requirements. We expect that as research and development expenses continue to grow, we will need to raise additional capital to sustain operations and research and development activities.
Future Capital Requirements
We plan to focus in the near term on the development, regulatory approval, and potential commercialization of DMT310 for the treatment of acne, psoriasis, and rosacea. We anticipate we will incur net losses for the next several years as we complete clinical development of DMT310 for the treatment of acne, psoriasis and rosacea and continue research and development of DMT410 for the treatment of aesthetic and medical skin conditions. In addition, we plan to identify, acquire or in-license and develop additional drug candidates, potentially build commercial capabilities, and expand our corporate infrastructure. We may not be able to complete the development and initiate commercialization of these programs if, among other things, our clinical trials are not successful or if the FDA does not approve our drug candidate arising out of our current clinical trials when we expect, or at all.
Our primary uses of capital are, and we expect will continue to be, compensation and related expenses, clinical costs, external research and development services, legal and other regulatory expenses, and administrative and overhead costs. Our future funding requirements will be heavily determined by the resources needed to support development of our drug candidates.
As a publicly traded company, we will incur significant legal, accounting, and other expenses that we were not required to incur as a private company. In addition, the Sarbanes-Oxley Act of 2002, as well as rules adopted by the SEC and Nasdaq, requires public companies to implement specified corporate governance practices that were not applicable to us as a private company. We expect these rules and regulations will increase our legal and financial compliance costs and will make some activities more time-consuming and costly.
We believe that the net proceeds from this offering, together with our existing cash, will be sufficient to fund our operating expenses and capital expenditure requirements into February 2023. We expect that our cash funds will fund the completion of our Phase 2 clinical trials for DMT310 for the treatment of rosacea, the initiation of our Phase 2 clinical trial for DMT310 for the treatment of psoriasis and the completion of the studies required to hold an End of Phase 2 meeting with the FDA for DMT310 for the acne program. We have based this estimate of cash runway on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we expect. Even with the net proceeds from this offering, together with our existing cash, we expect that we will require additional capital to conduct Phase 3 studies for DMT310 for the treatment of acne, rosacea, and psoriasis, to pursue in-licenses or acquisitions of other drug candidates, and to complete the commercialization of our product candidates. Additional funds may not be available on a timely basis, on favorable terms, or at all, and such funds, if raised, may not be sufficient to enable us to continue to implement our long-term business strategy. If we are unable to raise sufficient additional capital, we may need to substantially curtail our planned operations and the pursuit of our growth strategy.
We may raise additional capital through the sale of equity or convertible debt securities. In such an event, the terms of these securities may include liquidation or other preferences that adversely affect the rights of a holder of our common stock.
Because of the numerous risks and uncertainties associated with research, development, and commercialization of pharmaceutical drugs, we are unable to estimate the exact amount of our working capital requirements. Our future funding requirements will depend on many factors, including:
| • | the number and characteristics of the drug candidates we pursue; | |
|
|
| |
| • | the scope, progress, results, and costs of researching and developing our drug candidates, and conducting preclinical studies and clinical trials; | |
|
|
| |
| • | the timing of, and the costs involved in, obtaining regulatory approvals for our drug candidates; | |
|
|
| |
| • | the cost of manufacturing our drug candidates and any drugs we successfully commercialize; | |
|
|
| |
| • | our ability to establish and maintain strategic collaborations, licensing or other arrangements and the financial terms of such agreements; | |
|
|
| |
| • | the costs involved in preparing, filing, prosecuting, maintaining, defending, and enforcing patent claims, including litigation costs and the outcome of such litigation; and | |
|
|
| |
| • | the timing, receipt and amount of sales of, or milestone payments related to or royalties on, our current or future drug candidates, if any. | |
| 70 |
| Table of Contents |
To continue to grow our business over the longer term, we plan to commit substantial resources to research and development, clinical trials of our product candidates, and other operations and potential product acquisitions and in-licensing. We have evaluated and expect to continue to evaluate a wide array of strategic transactions as part of our plan to acquire or in-license and develop additional products and product candidates to augment our internal development pipeline. Strategic transaction opportunities that we may pursue could materially affect our liquidity and capital resources and may require us to incur additional indebtedness, seek equity capital or both. In addition, we may pursue development, acquisition or in-licensing of approved or development products in new or existing therapeutic areas or continue the expansion of our existing operations. Accordingly, we expect to continue to opportunistically seek access to additional capital to license or acquire additional products, product candidates or companies to expand our operations, or for general corporate purposes. Strategic transactions may require us to raise additional capital through one or more public or private debt or equity financings or could be structured as a collaboration or partnering arrangement. We have no arrangements, agreements, or understandings in place at the present time to enter into any acquisition, in-licensing or similar strategic business transaction.
If we raise additional funds by issuing equity securities, our stockholder will experience dilution. Debt financing, if available, would result in increased fixed payment obligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. Any debt financing or additional equity that we raise may contain terms, such as liquidation and other preferences that are not favorable to us or our stockholder. If we raise additional funds through collaboration and licensing arrangements with third parties, it may be necessary to relinquish valuable rights to our technologies, future revenue streams or product candidates or to grant licenses on terms that may not be favorable to us.
Going Concern
Since inception we have been engaged in organizational activities, including raising capital and research and development activities. We have not generated revenues and have not yet achieved profitable operations, nor have we ever generated positive cash flow from operations. There is no assurance that profitable operations, if achieved, could be sustained on a continuing basis. We are subject to those risks associated with any pre-clinical stage pharmaceutical company that has substantial expenditures for research and development. There can be no assurance that our research and development projects will be successful, that products developed will obtain necessary regulatory approval, or that any approved product will be commercially viable. In addition, we operate in an environment of rapid technological change and is largely dependent on the services of our employees and consultants. Further, our future operations are dependent on the success of the Company’s efforts to raise additional capital. These uncertainties raise substantial doubt about our ability to continue as a going concern for 12 months after the issuance date of our financial statements. The accompanying financial statements have been prepared on a going concern basis. The financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from the possible inability of the company to continue as a going concern, which contemplates the continuation of operations, realization of assets and liquidation of liabilities in the ordinary course of business. We incurred a net loss of $3.9 million for the year ended December 31, 2020 and $4.0 million for the nine months ended September 30, 2021, and had an accumulated deficit of $33.4 million at September 30, 2021. We anticipate incurring additional losses until such time, if ever, that we can generate significant revenue from our product candidates currently in development. Our primary source of capital has been the issuance of debt and equity securities.
| 71 |
| Table of Contents |
Global Pandemic Outbreak
In March 2020, the World Health Organization declared COVID-19 a global pandemic. The outbreak has become increasingly widespread in the United States, impacting the markets in which we operate. While the full impact of the pandemic continues to evolve, the financial markets have been subject to significant volatility that adversely impacts our ability to enter into, modify, and negotiate favorable terms and conditions relative to equity and debt financing initiatives. The uncertain financial markets, disruptions in supply chains, mobility restraints, and changing priorities as well as volatile asset values also affect our ability to enter into collaborations, joint ventures, and license and royalty agreements. The outbreak and government measures taken in response to the pandemic have also had a significant impact, both direct and indirect, on businesses and commerce, as worker shortages have occurred; supply chains have been disrupted; facilities and production have been suspended; and demand for certain goods and services, such as medical services and supplies, have spiked, while demand for other goods and services, such as travel, have fallen. The future progression of the pandemic and its effects on our business and operations are uncertain. We may face difficulties recruiting or retaining patients in our ongoing and planned preclinical and clinical trials if patients are affected by the virus or are fearful of traveling to our clinical trial sites because of the outbreak. We and our third-party contract manufacturers, CROs, and clinical sites may also face disruptions in procuring items that are essential to our research and development activities, including, for example, medical and laboratory supplies used in our clinical trials or preclinical studies, in each case, that are sourced from abroad or for which there are shortages because of ongoing efforts to address the outbreak. While expected to be temporary, these disruptions may negatively impact our results of operations, financial condition, and liquidity in 2021, and potentially beyond.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements.
Recently Issued Accounting Standards
For a discussion of recent accounting pronouncements, please see the Summary of Significant Accounting Policies in the Notes to our financial statements included elsewhere in this prospectus.
JOBS Act
On April 5, 2012, the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, was signed into law. The JOBS Act contains provisions that, among other things, reduce certain reporting requirements for an “emerging growth company”. As an “emerging growth company,” we are electing to take advantage of the extended transition period afforded by the JOBS Act for the implementation of new or revised accounting standards, and as a result, we will comply with new or revised accounting standards on the relevant dates on which adoption of such standards is required for non-emerging growth companies. Section 107 of the JOBS Act provides that our decision not to take advantage of the extended transition period is irrevocable.
Subject to certain conditions set forth in the JOBS Act, as an “emerging growth company,” we are not required to, among other things, (i) provide an auditor’s attestation report on our system of internal controls over financial reporting pursuant to Section 404, (ii) provide all of the compensation disclosure that may be required of non-emerging growth public companies under the Dodd-Frank Wall Street Reform and Consumer Protection Act, (iii) comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements (auditor discussion and analysis), and (iv) disclose certain executive compensation-related items such as the correlation between executive compensation and performance and comparisons of the chief executive officer’s compensation to median employee compensation. These exemptions will apply until the fifth anniversary of the completion of our initial public offering or until we no longer meet the requirements for being an “emerging growth company,” whichever occurs first.
| 72 |
| Table of Contents |
BUSINESS
Overview
We are a clinical-stage medical dermatology company focused on identifying, developing, and commercializing innovative pharmaceutical product candidates for the treatment of medical and aesthetic skin diseases and conditions we believe represent significant market opportunities because current therapies are inadequate or non-existent.
Dermatological diseases such as acne vulgaris (or acne), psoriasis vulgaris (or psoriasis), papulopustular rosacea (or rosacea), hyperhidrosis and various aesthetic indications, affect millions of people worldwide each year, and may negatively impact their quality of life and emotional well-being. While there are multiple current treatment options for these indications on the market, we believe that most have significant drawbacks, including underwhelming efficacy, cumbersome application regimens and varying negative side effects, all of which we believe lead to decreased patient compliance. While a majority of these indications are first treated with topical products, many patients frequently switch treatments or discontinue treatment altogether due to patient dissatisfaction stemming from slow and modest response rates, early onset of negative side effects, onerous application schedules and typically long duration of therapy. For diseases like psoriasis, a small percentage of patients may be candidates for biologic or systemic therapies, but these patients are typically required to try topical or oral treatment options prior to qualifying for these expensive biologic or systemic therapies. Given the limitations with current topical therapies and the restricted usability of biologic and systemic therapies, we believe there is a significant opportunity to address the needs of frustrated patients searching for topical products that satisfy their dermatological and lifestyle needs.
Our two product candidates, DMT310 and DMT410, both incorporate our proprietary, multifaceted, Spongilla technology to topically treat a variety of dermatological conditions. Our Spongilla technology is a multifactorial, naturally-derived product that is processed from a naturally grown freshwater sponge, Spongilla lacustris or Spongilla, which is processed into a powder that is mixed with a fluidizing agent immediately prior to application by the patient to form an easily applicable paste. Spongilla is a unique freshwater sponge that only grows in commercial quantities in select regions of the world and under specific environmental conditions, all of which give it its distinctive anti-microbial, anti-inflammatory and mechanical properties. The combination of these environmental conditions, the proprietary harvesting protocols developed with our supplier, and our post-harvest processing procedures produce a pharmaceutical product candidate that optimizes the mechanical component as well as the chemical components of the sponge for a product candidate with multiple mechanisms of action for the treatment of inflammatory skin conditions.
Our lead product candidate DMT310, is intended to utilize our Spongilla technology for a once-weekly treatment of a variety of skin diseases with our initial focus being for the treatment of acne vulgaris. However, due to the multiple mechanisms of action and anti-inflammatory effect seen with DMT310 throughout our various clinical trials for acne, we have quickly moved DMT310 into clinical trials for two additional indications, psoriasis and rosacea. We recently completed a Phase 1b proof of concept, or POC, trial in psoriasis and in November 2021 we initiated a Phase 2 clinical trial of DMT310 for the treatment of rosacea.
Our second product candidate utilizing our Spongilla technology is our combination treatment, DMT410. DMT410 is intended to consist of one treatment of our proprietary sponge powder followed by one topical application of botulinum toxin for delivery into the dermis. We first tested this program in a Phase 1 POC trial of axillary hyperhidrosis patients which saw 80% of patients achieve a reduction in gravimetric sweat production greater than 50% four weeks after treatment. Based on DMT410’s ability to effectively deliver botulinum toxin to the dermis, we initiated a Phase 1 POC trial of DMT410 for the treatment of multiple aesthetic skin conditions. On November 19, 2021, we announced top-line results from this trial where we saw encouraging data that we believe warrants further investigation of this program.
We believe our Spongilla technology platform will enable us to develop and formulate singular and combination products that are able to target topical delivery of chemical compounds into the dermis for a variety of indications. We believe the mechanical ability of the Spongilla technology allows for the intradermal delivery of a variety of large and small molecules to a targeted treatment site, through topical application as seen in our Phase 1a DMT410 hyperhidrosis trial. We believe the combination of Spongilla’s mechanical and chemical components (which we believe have demonstrated, in-vitro, anti-microbial and anti-inflammatory properties), add to the versatility of our Spongilla technology platform, allowing for application in the treatment of a wide variety of medical and aesthetic skin conditions and diseases.
| 73 |
| Table of Contents |
DMT310 is intended to consist of two grams of powder processed from a naturally grown freshwater sponge, Spongilla lacustris, which powder is then mixed with a fluidizing agent (hydrogen peroxide) immediately prior to application by the patient to form an easily applicable paste, similar to a mud mask. The paste is left on the skin for approximately fifteen minutes until dry, after which time it is washed off with water. Due to the unique combination of DMT310’s mechanical components and chemical components, we believe patients will only need to apply the product once-weekly to produce a treatment effect. The mechanical components of the Spongilla powder consist of a large number of microscopic siliceous spicules that, when massaged into the skin, penetrate the stratum corneum (the skin’s outermost protective layer) and create microchannels into the dermis where many facets of inflammatory skin diseases reside. We believe that the penetration of the spicules not only increase collagen production, but also cause the opening of microchannels which allow oxygen to enter clogged pilosebaceous glands, eliminating the anaerobic environment in which C.acnes grow (C. acnes is the bacteria that cause inflammatory lesions in acne patients). The spicules may also provide a channel for DMT310’s naturally occurring chemical compounds to be delivered to the dermis and pilosebaceous glands, helping to kill the C. acnes. In addition to these anti-microbial compounds, DMT310 also appears to have anti-inflammatory chemical compounds, as evidenced in-vitro, that inhibit inflammation through the reduction of C.acnes stimulated IL-8 production and by inhibiting IL-17A and IL-17F expression in human cell lines. During in-vitro studies of DMT310’s organic compounds, we observed the inhibition of lipogenesis of sebocytes, which may translate to a reduction in sebum (an oily and waxy substance produced by the human body’s sebaceous glands) production and oiliness of the skin in patients. We believe the combination of these biological effects along with the mechanical effects of the spicules are important factors to treat multiple inflammatory skin diseases.
Application of DMT310

Image 1: The Spongilla is processed into a fine powder and packaged into 2g pouches with a 6mL bottle of 3% H2O2 (hydrogen peroxide). Once per week, patients mix the powder with hydrogen peroxide, and massage the mixture onto their skin; after 10-15 minutes the product is easily removed with water.
DMT410 is the second product candidate from our Spongilla platform. We are developing DMT410 for the topical treatment of skin diseases and conditions typically treated with multiple intradermal injections of botulinum toxin. DMT410’s combination treatment regimen uses one application of our unique Spongilla powder followed by one topical application of botulinum toxin to enable the topical delivery of botulinum toxin into the dermis for the treatment of a variety of indications, including for the treatment of hyperhidrosis and multiple aesthetic skin conditions such as overall skin quality, pore size and count, sebum production, brightness, luminosity, and fine lines. If approved by the FDA, we believe DMT410’s topical delivery of botulinum toxin can greatly increase its market opportunities due DMT410’s needle-free application, targeted intradermal delivery and potential for expanded indications.
We believe that the current medical and aesthetic dermatology landscape lacks innovative treatment options, mainly seeing the introduction of reformulations and combinations of old molecules. This provides an ideal opportunity to change how patients treat their skin conditions. With our anticipated once-weekly treatment schedule, we believe we can become a leader in the space with a product candidate derived from a natural sponge that may improve patient compliance with minimal side effects and a rapid time to treatment effect, as seen in our multiple clinical trials in acne and psoriasis. If we are able to successfully develop our product candidates, receive FDA approval, develop a concentrated prescribing base of dermatologists, and utilize our management’s prior experience, we believe we have the ability to build a commercial organization to develop and commercialize treatment options in our core areas of focus within the dermatology space.
| 74 |
| Table of Contents |
Our Clinical Development Pipeline and Product Candidates
Our clinical development pipeline currently consists of DMT310 and DMT410, each in development for multiple skin diseases and conditions. In the accompanying section we will describe each product candidate, its benefits, and our market strategy for each product candidate. The dates reflected in the below table and sections are estimates only, and there can be no assurances that the events included in the below table or sections will be completed on the anticipated timeline presented, or at all.
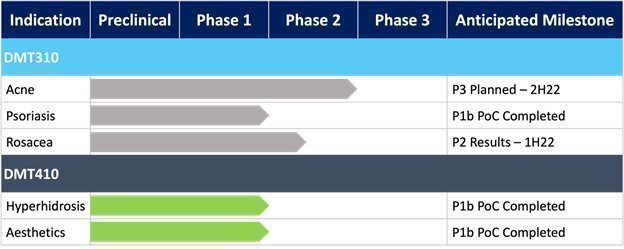
DMT310
Moderate-to-Severe Acne. In June 2020, we completed a randomized, double-blind, multicenter, placebo-controlled Phase 2b clinical trial of DMT310 for the once-weekly treatment of moderate-to-severe acne. DMT310 showed statistically significant improvement versus placebo for all three endpoints (inflammatory lesion counts, non-inflammatory lesion counts, and the Investigators Global Assessment), after only four topical treatments and continued until the end of study at week 12. We plan on using these same endpoints in our two upcoming Phase 3 trials to support the filing of our NDA for DMT310 for the treatment of moderate-to-severe acne vulgaris. We believe these results from once-weekly applications may favorably position DMT310 in the market for the treatment of moderate-to-severe acne. We intend to initiate the two required Phase 3 clinical trials in late 2022 after holding our End of Phase 2 meeting with the FDA in the second half of 2022.
Mild-to-Moderate Psoriasis. In October 2021, we completed a Phase 1b POC trial of DMT310 for the once-weekly treatment of mild-to-moderate psoriasis. Plaque psoriasis is a chronic, inflammatory skin disease that comprises approximately 80% of the psoriasis market as of 2019, according to Fortune Business Insights Market Research Report, with a majority of patients having mild-to-moderate disease which makes them less likely to receive an approved biologic treatment, that are only indicated for patients with moderate to severe disease, as a first line therapy. Due to the large population of patients who suffer from mild-to-moderate psoriasis, and lack of effective topical therapies for more mild disease, we believe there is a large unmet need for an effective topical product with limited side effects. Based on the data in our Phase 1b POC trial, the in-vitro data of DMT310’s reduction of IL-17A and IL-17F, and the anti-inflammatory effects we observed in its Phase 2b trial for acne, we believe DMT310 may be used as a first-line therapy for patients suffering from mild-to-moderate psoriasis who are not candidates for biologic treatments.
In October 2021, we announced top-line results from our Phase 1b POC trial of DMT310 for the treatment of 30 mild-to-moderate patients with psoriatic lesions covering between 2% to 30% of their body surface area. Patients were treated with DMT310 once a week for 12 weeks. Based on the efficacy, safety and tolerability profile observed in the POC trial, we initiated additional work to better inform our clinical trial design prior to moving into a larger Phase 2, placebo-controlled, clinical trial, which we anticipate initiating in the first half of 2022.
| 75 |
| Table of Contents |
Moderate-to-Severe Rosacea. We enrolled our first patient in a Phase 2 clinical trial of DMT310 for the once-weekly treatment of moderate-to-severe rosacea in November 2021. Rosacea, or subtype II, is a chronic skin disease characterized by inflammatory lesions on the face that typically affects adults of all ages, but particularly affects women aged 30-50 and can cause great emotional and psychological distress. Due to the lack of effective therapies currently on the market, we believe there is a significant need for an effective topical product with limited side effects. Based on the effect against inflammatory lesions observed in the Phase 2b acne trial, along with multiple biomarkers seen in-vitro, we believe DMT310 may have similar anti-inflammatory effects in rosacea as seen in acne due to it also being a disease of inflammatory lesions on the face and may be a potential treatment option for patients suffering from this disease. We plan to enroll 180 patients, 18 years of age or older, with moderate-to-severe rosacea and provide them with once-weekly treatments of DMT310 for 12 weeks and plan to report top-line results from this trial in the second half of 2022. The clinical endpoints for this trial are intended to be the absolute reduction in inflammatory lesion count, and an Investigator’s Global Assessment score on a scale of 0 to 4. Assuming this trial is successful, we plan on requesting an End of Phase 2 meeting with the FDA in the first half of 2023 and initiate a Phase 3 rosacea program in the second half of 2023.
There can be no assurance that DMT310 will receive FDA approval for any of the foregoing indications.
DMT410
We are developing the second product candidate from our Spongilla platform, DMT410, for the topical treatment of skin diseases and aesthetic conditions typically treated with multiple injections of botulinum toxin. Currently, botulinum toxin must be injected multiple times to successfully deliver sufficient quantities of botulinum toxin to the desired treatment area. While injections are successful for many different diseases and aesthetic conditions, they limit botulinum toxin’s use for additional conditions where injections are difficult, painful or otherwise not viable. DMT410’s combination treatment regimen uses one application of our unique Spongilla powder followed by one topical application of botulinum toxin. The Spongilla powder is mixed with a fluidizing agent and is massaged into a patient’s treatment area by the treating physician to enhance spicule penetration to create microchannels into the dermis. After 10 to 15 minutes, the physician removes the Spongilla mask with water. The physician then expresses botulinum toxin from a syringe in precise amounts and onto the patient’s skin. The botulinum toxin is then massaged into the treatment area to take advantage of the microchannels created by Spongilla’s spicules, which provide access to the dermis passed the stratum corneum. We believe this treatment application will enable the topical delivery of botulinum toxin into the dermis for the treatment of a variety of medical and aesthetic skin conditions, including for the treatment of hyperhidrosis, acne, rosacea, overall skin quality, pore size and count, luminosity, brightness, and fine lines. We believe DMT410’s topical delivery of botulinum toxin can greatly increase market opportunities for botulinum toxin due DMT410’s needle-free application, targeted intradermal delivery and potential for expanded indications that do not require injections.
To date, we have completed two open-label Phase 1b POC clinical trials of DMT410 for the treatment of axillary hyperhidrosis and multiple aesthetic skin conditions. The Phase 1b POC trial for axillary hyperhidrosis consisted of 10 patients receiving one treatment of DMT410 on each axilla. Four weeks after one treatment with DMT410, patients exhibited reduction in sweat production. The clinical endpoints for this trial included (i) percent of patients with greater than 50% reduction in gravimetrically measured sweat production from baseline, (ii) percent of patients with gravimetric sweat production less than 50mg, and (iii) percent change in gravimetric sweat production. Four weeks after one treatment with DMT410, 80% of patients saw a decrease in gravimetric sweat production greater than 50%, 85% of patients had gravimetric sweat production of less than 50mg, and patients had an average decrease in gravimetric sweat production of 75% from baseline. We believe these results support that DMT410 may aid in the topical delivery of botulinum toxin into the dermis for a treatment effect similar to multiple injections of botulinum toxin. With DMT410, we believe botulinum toxin may be applied in a topical manner to penetrate the skin so as to be targeted to the dermis without the need for multiple injections.
| 76 |
| Table of Contents |
We recently completed the second open-label, ten (10) patient, Phase 1b POC trial of DMT410 for the treatment of multiple aesthetic skin conditions such as pore size, Global Aesthetic Improvement, brightness, luminosity, sebum production, fine lines under the eye, glabellar lines, forehead lines, and lateral canthal lines. If approved, we believe DMT410 could expand the market for botulinum toxin by adding new indications that require intradermal administration to botulinum toxin’s approved uses. In our Phase 1b POC trial of DMT410, patients received one treatment of DMT410 and were evaluated every four weeks for a total duration of 16 weeks to determine DMT410’s safety and tolerability profile, effectiveness, and its duration of treatment effect. We announced top-line results in November 2021, where we observed an improvement in many of the trial’s endpoints. At week 8, 80% of patients had at least a 25% improvement in their Global Aesthetic and 60% of patients had a 25% improvement in pore size. Also at week 8, 90% of patients had at least a one-point improvement in luminosity and 60% of patients had at least a one-point improvement in brightness. These physician graded results were supported by objective analysis provided by Canfield Scientific’s VISIA and PRIMOS visual analysis camera systems. Based on these results, we are actively seeking a partner with a botulinum toxin to continue development of DMT410 in a larger placebo controlled Phase 2 trial where we can study multiple doses of botulinum toxin applied to the entire face. We believe these results, combined with our results in hyperhidrosis, further demonstrate how DMT410’s combination regimen could greatly expand the potential indications for botulinum toxins for aesthetic skin conditions, as well as other dermatologic skin diseases such as acne or rosacea.
There can be no assurance that DMT410 will receive FDA approval for any of the foregoing indications.
Our Strategy
We plan on in-licensing, developing and commercializing differentiated medical and aesthetic dermatology product candidates for the treatment of various skin diseases and conditions, which we believe have significant unmet needs in the market. The key components of this strategy are as follows:
| • |
| Complete development and regulatory approval of DMT310 for acne. We recently completed a Phase 2b clinical trial of DMT310 for the treatment of moderate-to-severe acne. We plan to hold an End of Phase 2 meeting with the FDA, in the second half of 2022, and subject to the end of this Phase 2 meeting, we plan to initiate two pivotal Phase 3 clinical trials for DMT310 in the second half of 2022, with top-line results expected in the first half of 2024. Assuming we receive positive clinical results, we would expect to submit a new drug application, or NDA, for DMT310 for the treatment of moderate-to-severe acne in the second half of 2024. |
| • |
| Complete a Phase 2 trial of DMT310 for the treatment of psoriasis. In October 2021, we announced top-line results of our Phase 1b POC trial in patients with mild-to-moderate psoriasis. We believe the results of this POC study warrant the continued development of DMT310 for the treatment of psoriasis. We plan to initiate a Phase 2 trial for DMT310 in psoriasis in the first half of 2022. If successfully developed and commercialized, we believe DMT310 would be the first once-weekly topical product available to treat psoriasis. |
| • |
| Complete a Phase 2 trial of DMT310 in rosacea. Based on the data obtained from our Phase 2 trials in acne and the anti-inflammatory results seen in-vitro, we initiated a Phase 2 trial of DMT310 for the treatment of rosacea in November 2021, with top-line results expected in the second half of 2022. We believe this will help bolster our pipeline and could establish the broader use of DMT310 in multiple inflammatory skin diseases. |
| • |
| Explore mutually beneficial partnership opportunities for our DMT410 program in hyperhidrosis and aesthetic skin conditions. In November 2021, we announced top-line results of our Phase 1b POC trial of DMT410 for the treatment of multiple aesthetic skin conditions using our Spongilla technology for the topical application of OnabotulinumtoxinA (brand name BOTOX®). We believe this trial provides further data of the ability for DMT410 to topically deliver botulinum toxin into the dermis for skin conditions and diseases. Based on the results of DMT410 in hyperhidrosis and aesthetics, we are currently exploring partnership opportunities to further develop DMT410 for the topical treatment of skin diseases and aesthetic skin conditions. |
| • |
| Acquire or in-license additional dermatology programs to our portfolio that complement our current product candidates. We continuously evaluate potential partnering opportunities that will bolster our current product candidate portfolio and provide substantial value to our organization. We intend to focus on early to mid-stage development product candidates to generate clinical data and potentially move to later stages of development and ultimately on to commercialization. |
| • |
| Maximize the value of our portfolio by commercializing our product candidates in territories where we can do so effectively and partner for other territories to help us reach new markets. If we receive FDA approval for our product candidates, we plan to maximize the territories where they could be sold by partnering with established companies in new territories outside of the U.S. market, if possible. |
| • |
| Further strengthen our intellectual property portfolio, path to new chemical entity, or NCE, exclusivity, raw material supply and advance our regulatory filings. We plan to continue to strengthen our IP portfolio, seek NCE exclusivity for DMT310, maintain our exclusive supply agreement for our raw material requirements, and continue to protect our proprietary information. We believe these activities will be our primary competitive advantages if our product candidates receive regulatory approval. |
| 77 |
| Table of Contents |
The dates reflected in the foregoing are estimates only, and there can be no assurances that the events included will be completed on the anticipated timeline presented, or at all. Further, there can be no assurance that we will be successful in the development of DMT310 or DMT410, or any other product candidate we may develop in the future, or that DMT310 or DMT410, or any other product candidate we may develop in the future, will receive FDA approval for any indication.
Dermatology Market Overview
We are currently focused on the medical and aesthetics dermatology markets, which include multiple common and undertreated skin diseases and conditions such as acne, rosacea, psoriasis, hyperhidrosis, and multiple aesthetic conditions, some with no currently approved products, including the reduction of fine lines, pore size, sebum production and improvement in luminosity and overall skin quality. We believe these diseases and conditions cause significant negative impacts on patients’ quality of life, including physical and emotional trauma and social stigmatism, causing patients to constantly seek better treatment options to help alleviate their conditions. We also believe these markets have not experienced the same level of development and advances as other markets, as there have been few novel or innovative topical products recently approved other than reformulations or combinations of existing compounds. We believe our product candidates will be well situated within the market and offer the innovative solutions to the underserved medical and aesthetic dermatology markets.
The U.S. medical dermatology market has experienced significant growth in recent years based on the new treatment options and greater patient access to care. Based on current market data, the U.S. medical dermatology market (excluding biologics) was valued at over $16 billion dollars in prescription pharmaceutical sales in 2020.
The American Society of Plastic Surgeons estimates that over 15.4 million cosmetic procedures were performed in the U.S. in 2016, of which about 7 million used botulinum toxin. There are many factors that continue to drive growth in the aesthetics dermatology market such as greater patient acceptance, including from an increase in younger patients, and the discretionary cash that patients are willing to spend on aesthetic care. We also believe patients have a growing willingness to pay out-of-pocket for effective skin treatments in order to achieve their desired personal aesthetic look, which further supports the demand and pricing in those markets.
Based on the foregoing, we believe the dermatology market, both aesthetics and medical, offers a low-cost commercialization opportunity compared to many other prescription based specialty markets, due to the relatively small number of specialists in the dermatology field. According to the American Academy of Dermatology, in 2020 there were approximately 18,000 dermatologists in the U.S. We plan to target a subset of these dermatologists, who are larger prescribers of competitive products and who treat a large percentage of patients with our approved indications by creating a targeted sales force, in order to cover a significant portion of our target physicians. We believe the combination of a prescription based and cash-pay based product lines is an attractive business opportunity, as it incorporates multiple aspects of the dermatology market that move independent of the greater healthcare market.
| 78 |
| Table of Contents |
Background of Our Spongilla Technology
Spongilla Lacustris Overview
Spongilla lacustris, or Spongilla, is a freshwater sponge from the Spongillidea family that grows in freshwater rivers and lakes in commercial quantities in select regions of the world. It becomes dormant during the winter months and regrows each year to growth forms ranging from encrusting, to digitate, to branched, depending on its habitat’s growth conditions. While it grows in many parts of the northern hemisphere, there are only certain locations where it grows in the quantities and of the quality to viably support a commercial pharmaceutical product. One such location is the Volga River in central Russia, where we have signed an exclusive supply agreement with one of the larger known suppliers of Spongilla raw material for DMT310, which guarantees our supply of Spongilla raw material. Traditionally, locals would harvest small amounts of Spongilla for its perceived medicinal properties and use it as a folk medicine to treat a variety of inflammatory conditions, including arthritis. Over the last 18 years, our exclusive supplier has refined its harvesting methods and procedures and is now capable of supplying a high quality raw material. Our supplier has the capacity to collect and process large quantities of Spongilla per year. We believe our supplier will be able to supply a raw material in the quantities and of the quality necessary to support our clinical and commercial needs.
The traditional use of Spongilla in Russia has provided a large amount of safety data. In 2003, the Russian Ministry of Health indicated that Spongilla has been used by over one million people per year, with few reported safety issues. In 2017, we submitted this safety information, along with various other publications and non-clinical studies, in an Investigational New Drug, or IND, application to the FDA’s Division of Dermatology and Dental Products with reference to the FDA’s Botanical Drug Development Guidance for Industry, or Botanical Guidance. This submission enabled the FDA to approve our IND for DMT310, allowing us to proceed directly into a Phase 2 clinical trial in patients due in part to historical human exposure. While we are still required to complete a majority of required non-clinical and pharmacokinetic studies, we were able to strategically conserve resources while gathering human clinical efficacy and safety data prior to beginning such work.
Spongilla’s Multiple Mechanism of Actions
While Spongilla is technically a part of the animal kingdom, it grows and acts more similarly to a plant in that it can completely regenerate every year, even in harsh environmental conditions. This unique property of Spongilla not only allows us to reference the Botanical Guidance, but also helps ensure the regrowth of sufficient supply of raw material each year. In addition to causing a regrowth of the sponge each year, the harsh environmental conditions the sponge lives in helps contribute to our Spongilla technologies’ multiple mechanisms of actions. Based on knowledge gained from over almost 18 years of harvesting Spongilla, our supplier has learned the necessary environmental conditions and Spongilla characteristics that must be present for optimal raw material harvest and to ensure the raw material contains the necessary properties for an effective pharmaceutical product. These particular properties include both mechanical and chemical components that are a naturally occurring part of the sponge raw material and contribute to our Spongilla technology’s mechanisms of action in the treatment of skin diseases and conditions.
The mechanical components of DMT310 come from the Spongilla’s skeletal structure, which is made up of a large number of siliceous spicules that are bound together by organic material, as seen in Image 2 below. These spicules are smooth, rod-like shapes which come to a point on each end, and if the Spongilla is harvested under certain proprietary environmental conditions, the spicules can average between 150-300 micrometers in length and about 10-15 micrometers in diameter. While there are other types of freshwater and marine sponges, many of their spicules can be covered in barbs or hooks which we believe would get stuck in the skin or are blunt on each end, making skin penetration difficult.
| 79 |
| Table of Contents |

Image 2: Siliceous Spicules Present in Spongilla
After harvesting and further processing in the U.S., the shape and size of our spicules make them the ideal mechanism to penetrate the stratum corneum, the skins barrier, and temporarily create a micro-channel into the dermis without penetrating into the subcutaneous tissue, where the larger blood vessels are located. These newly created microchannels temporarily open the skin’s barrier to allow for the targeted delivery of large and small chemical compounds into the dermis. Most topically applied products currently contain various penetration enhancers that help force the active molecule through the stratum corneum and into the dermis, such as Dimethyl Sulfoxide (DMSO). However, these penetration enhancers are only able to help smaller molecules penetrate and are usually unable to aid larger molecules, such as botulinum toxin, in topical delivery. These penetration enhancers can also cause unwanted side effects such as dry skin or garlic like taste, breath and body odor. We believe our Spongilla technology is differentiated by enabling the delivery of both small and large molecules through topical application with less irritation and side effects than other topically applied products.
In addition to creating many microchannels in the skin, we believe the penetration of the spicules can open closed comedones allowing oxygen into the anaerobic environment of the clogged pilosebaceous glands, where C.acnes and other bacteria survive. Lastly, we believe the spicules promote collagen production within the skin which accelerates the skin’s rejuvenation period, thus bringing refreshed skin to the surface at a quicker rate than the skins normal turnover cycle. Typically, the skin takes between three to four weeks to bring a new layer to the surface, while we believe our Spongilla technology may allow this process to complete in less than one week. We believe this decreases the time to treat inflammatory skin diseases and conditions while also enhancing the look of a patient’s skin.
Our Spongilla technology also contains multiple active chemical compounds that we believe may aid in our product candidates’ treatment of multiple dermatology skin diseases and conditions. We believe part of Spongilla’s natural defense mechanism is the creation of organic material to fight off natural enemies present in the water in which it grows. This organic material binds its spicules together to form the skeletal structure of the sponge. Based on multiple in-vitro studies, we believe the organic compounds within the sponge, when separated from the spicules, have both anti-inflammatory and antimicrobial properties. We have observed anti-inflammatory activity including reduction of C. acnes, stimulated IL-8 production and the downregulation of the production of IL-17A and IL-17F in human cell lines. Additionally, in our in-vitro studies, we observed the inhibition of lipogenesis of sebocytes, which may translate to a reduction in sebum production and oiliness of the skin in patients.
While we believe each of the mechanical or chemical components of our Spongilla technology may be beneficial in treating various diseases in its own right, we believe the impact of each mechanism may be greatly enhanced when combined with the other. The large number spicules contained in each treatment create many microchannels through the stratum corneum, allowing for sufficient penetration and delivery of the chemical components into the treatment area to fight inflammation and bacteria.
| 80 |
| Table of Contents |
FDA Botanical Drug Development Guidance for Industry
Most currently approved topical dermatology products are reviewed solely by the FDA’s Office of Dermatology and Dental Products and follow a standard approval pathway. However, due to our lead product candidate, DMT310, being derived from a natural source, it will be reviewed by the FDA Office of Dermatology and Dental Products with input from the FDA Botanical Review Division. While Spongilla is not a botanical, the FDA has allowed us to reference the Botanical Guidance for raw material quality control and batch to batch consistency through development and into commercialization. We believe our ability to reference the Botanical Guidance and receive input from the Botanical Review Division on DMT310 provides us with key advantages in DMT310’s regulatory pathway to approval, if achieved. These advantages include being able to move into human clinical studies upon the FDA’s acknowledged receipt of our IND letter and subsequent study may proceed, saving us substantial financial resources to achieve human clinical data. Additionally, while we believe that our sponge contains multiple active chemical compounds, based on our regulatory analysis of the feedback from the FDA and the Botanical Guidance, we believe we are only required to provide identifiable and quantifiable active components to show quality control and batch to batch consistency. We believe this will make it more difficult for potential competitors to replicate DMT310 due to their inability to know every component of our product candidate and to show their product is similar in its composition. Thus, we believe a competitor with a similar product or product candidate would have to follow all of the manufacturing, development, and regulatory steps we must complete for approval. However, there can be no assurance that we successfully navigate the development of DMT310 or that DMT310 will receive FDA approval.
Our Product Candidates
DMT310
Our lead product candidate, DMT310, is a unique, once-weekly, naturally derived topical product, first being developed for the treatment of moderate-to-severe acne vulgaris, or acne. It is derived from freshwater Spongilla lacustris, or Spongilla, which grows under certain environmental conditions in select locations throughout the northern hemisphere. Our Spongilla raw material is harvested by our exclusive partner in Russia abiding by strict protocols which our supplier has developed over 18 years. The result of these strict protocols is a consistent chemical structure that is reproducible year after year, which is critical in producing a material able to be used in a pharmaceutical product. After harvesting, the Spongilla is shipped to our manufacturing facility in the U.S. for further processing into a uniform powder before being packaged into sachets. Immediately prior to treatment the patient will mix the powder with a diluent (hydrogen peroxide) to form a paste which the patient can then apply to the treatment area to treat the multiple facets of their disease. DMT310 utilizes the Spongilla’s mechanical spicules to help resurface a patient’s skin while also creating microchannels through the stratum corneum to allow the penetration of the Spongilla’s naturally created organic compounds to help treat various skin diseases. We believe these organic compounds are able to travel through the newly created microchannels into the dermis and sebaceous gland where both inflammatory and non-inflammatory acne lesions originate. DMT310 targets treatment of the multiple facets of acne by combining the substantial mechanical and chemical activity of Spongilla into an easy to apply product that only needs to be applied once-weekly. If approved by the FDA, we believe the combination of the mechanical and chemical properties of DMT310 has the potential for a more rapid time to treatment effect with fewer treatments, less side effects and better tolerability than other currently marketed topical acne products.
DMT310 for Treatment of Acne Vulgaris
Market Opportunity. Acne is characterized by areas of scaly red skin, non-inflammatory blackheads and whiteheads, inflammatory lesions, papules and pustules and occasionally cysts and scarring that occur on the face, neck, chest, back, shoulders, and upper arms. It affects approximately 50 million people in the U.S., with about 85% of teenagers experiencing some form of acne. The U.S. prescription acne market had approximately $2.3 billion in prescription pharmaceutical sales in 2019, according to IQVIA Inc. market data.
A majority of patients experience some form of acne during their teenage years and for some, their acne may diminish over time, or at least tends to decrease by age 25. There is, however, no way to predict how long it will take for acne to disappear entirely, with some individuals suffering from acne well into their 30s, 40s and beyond. While not life-threatening, acne causes significant trauma for those suffering from it due to social stigmas, substantial risk of permanent facial scarring, lowered self-esteem and social withdrawal. Therefore, we believe early and aggressive treatment with an effective once-weekly product may lessen the overall long-term impact of this disease and may lead to an increase in a patient’s quality of life.
| 81 |
| Table of Contents |
Due to acne’s negative impact on a patient’s quality of life and negative impact on facial aesthetic, patients suffering from acne tend to be highly motivated to rid themselves of acne and we believe more willing to pay more out-of-pocket for higher priced and highly effective treatments. It is our belief that patients seeking an easy to use and effective topical product will tolerate less favorable reimbursement rates than for other prescription products for other indications, allowing for favorable pricing if we are able to eventually obtain approval for and successfully commercialize DMT310 for acne. Furthermore, if approved, we believe that DMT310’s natural characteristics may allow us to expand our addressable acne market to include those patients who value using naturally derived products, such as DMT310.
The overall acne market can currently be broken into three separate classes based on the severity of the acne:
| • |
| Mild Acne: characterized by few papules or pustules; typically treated with over-the-counter products or topical prescription therapies. |
| • |
| Moderate Acne: characterized by multiple papules and pustules with moderate inflammation; typically treated with a combination of oral and topical prescription therapies. |
| • |
| Severe Acne: characterized by substantial papules and pustules, with many nodules and/or cysts and significant inflammation; currently treated with oral and topical combination treatments and photodynamic therapy as a third-line treatment option. |
Limitations of Current Standard of Care. While current treatment options may be effective for some patients, there are many limitations and drawbacks of current acne products which cause poor patient adherence. All currently approved topical therapies for the treatment of acne must be applied once or even twice a day in order to allow an accumulation of product within the skin to effectively treat the disease. This requirement to apply multiple times per day becomes very onerous and time consuming for patients, causing many patients to fail to comply with the strict application regimen and/or skip multiple treatments. Proper use and application schedules are particularly important for topical acne products and poor patient adherence may lead to reduced treatment effect and ultimately discontinuation of treatment due to lack of effect.
Many current acne products, such as retinoids, must be applied at least once-a-day and may cause significant stinging, burning and peeling after each application. These tolerability issues, which may start occurring after the first application, and the substantial discomfort they cause, lead many patients to discontinue the necessary daily application schedule. It is well known that benzoyl peroxide, or BPO, leads to drying of the skin and that retinoids result in many local skin reactions including erythema, burning, and peeling, after the first treatment. It has been observed in the combination study of adapalene/BPO, where more than 20% of the subjects reported moderate or severe erythema and stinging/burning.
Lastly, most topical products have an unavoidable latency period of 6-8 weeks until patients have a definite improvement. This means they may have to endure 30 to 60 applications before observing that their acne is improving (assuming a daily, or twice daily regimen), all while dealing with the burning, stinging and peeling that may accompany these topical products. We believe that teenagers, which make up the largest segment of the acne market, become impatient with the lack of rapid perceived effect leading to premature discontinuation of treatment. The lack of rapid treatment effect, side effects, and onerous application schedules all greatly contribute to patient compliance issues and could ultimately lead to treatment failure. We believe patients are more concerned with efficacy outcomes than costs, thus we believe patients will be more willing to pay higher out of pocket costs.
Our Solution for Moderate-to-Severe Acne. If approved, we believe DMT310’s once-weekly application regimen will increase patient compliance, potentially increasing the likelihood of improved acne results. Using our multifaceted, once-weekly Spongilla treatment technology, we are developing DMT310 to create a paradigm shift in how acne is treated by dermatologists by attempting to make DMT310 the preferred treatment option for all acne patients. We have designed DMT310 to treat the multiple factors of acne while also attempting to increase patient compliance.
| 82 |
| Table of Contents |
If approved, we believe DMT310 has the potential to remedy many of the negative characteristics associated with current topical therapies for moderate-to-severe acne vulgaris, including cumbersome treatment regimens, negative side effects (including burning, stinging, itching or dryness, which may occur as early as the first treatment and continue daily thereafter), and delayed time to effectiveness (which may take up to eight weeks). DMT310 is designed to be applied only once a week, rather than once or twice a day. We believe a once-weekly schedule may be conducive to high patient compliance. In addition, in our recent Phase 2b acne trial, on average, patients experienced an approximately 45% reduction in inflammatory acne lesions after just four treatments, with continued improvement of up to 62% reduction at 12 weeks. Further, approximately 90% of patients had no, or mild, tolerability issues at the end of the 12-week trial and no patients experienced any severe tolerability issues.
In addition, in our Phase 2b trial we observed that DMT310 started showing a statistically significant difference from placebo after just four treatments while also having a rapid reduction on inflammatory and non-inflammatory lesions. We believe this rapid visible response encouraged patients to continue to adhere to the once-weekly application schedule leading to a continued reduction in their lesions until the end of trial at week 12. Thus, we believe that a topical product that needs to be applied once-weekly with a quicker time to perceived treatment effect and fewer tolerability issues has the opportunity to exhibit greater treatment success due to improved patient adherence leading to loyal and repeat users.
DMT310 for the Treatment of Mild-to-Moderate Psoriasis
We believe that DMT310 could also be an effective treatment for mild-to-moderate psoriasis based on the data received from our recently completed Phase 1b POC trial and the in-vitro effect seen on the down regulation of IL-17A and IL-17F, as well as its ease of application to mild-to-moderate psoriatic lesions with smaller surface areas.
Psoriasis is characterized by “plaques,” or raised red areas of skin covered with a silver or white layer of dead skin cells referred to as “scales.” Psoriatic plaques can appear on any area of the body, but most often appear on the scalp, knees, elbows, trunk, and limbs, and the plaques are often itchy and sometimes painful. The psoriasis lesions are characterized by hyperproliferation of keratinocytes and a lymphocyte-rich infiltrate consisting primarily of T cells. In the dermis and epidermis, T lymphocytes interact with antigen-presenting cells and secrete Th1 and Th17 cytokines. These activated T cells and the inflammatory cytokines they secrete are believed to induce the skin lesions seen in psoriasis. In addition to the broad anti-inflammatory properties, we have observed in our clinical acne studies, DMT310’s ability exhibited in-vitro a dose dependent inhibition of both IL-17A and IL-17F, key cytokines implicated in the pathogenesis of psoriasis. Thus, DMT310 may provide a method to topically deliver targeted anti-inflammatory therapy directly to psoriatic lesions with good local tolerability in an easy to apply regimen.
Market Opportunity. Patients are either underdiagnosed, undertreated or left untreated. This leaves patients seeking new treatment options. Psoriasis is a chronic, inflammatory skin disorder estimated to affect up to 3.2% of the world’s population with global sales of $14.2 billion in 2020, which is projected to increase to $27.5 billion by 2030. Plaque-type psoriasis is the most common form of psoriasis, occurring in more than 80-90% of cases of psoriasis with approximately 80% of patients experiencing the mild disease form and 20% experiencing moderate-to-severe form of the disease. In addition to the disfigurement caused by psoriatic lesions, patients also may experience pruritus, or itching, which can be particularly common and bothersome for patients. Not only does psoriasis cause direct clinical challenges, but patients also suffer a negative impact on their quality of life. Patients can suffer substantial psychological impacts from their disease, including, social stigmas, feelings of rejections and shame, discrimination in the workplace, and reduced productivity, among many others. These patients are commonly looking for a safe and effective product to treat their disease.
Limitations of Current Standard of Care. Most of the available therapies target moderate-to-severe disease, meaning that mild patients are undertreated with one in five not being happy with their current treatments. The treatments for mild psoriasis patients are highly genericized, but are often inadequate to control a patient’s disease. Mild psoriasis patients are first treated with topical therapies due to the reduced systemic exposure. However, patients often feel that topical treatments are one of the negative aspects of psoriasis, which we believe is partly due to the limited options available like, coal tar, retinoids, calcineurin inhibitors and corticosteroids. While topical steroids are a very common treatment, drawbacks include being able to be used only for a short period of time and are associated with Hypothalamic pituitary adrena axis suppression, skin atrophy (thinning), striae (stretch marks), and telangiectasia (spider veins), among other side effects. Furthermore, some of these side effects are irreversible, persisting even after therapy is discontinued. Consequently, high-potency topical steroids are not recommended for chronic use and physicians generally will not prescribe them for treatment on the face. Also, rebound is a known challenge with steroids, where after steroid discontinuation, the psoriasis returns even worse than it was before steroid treatment was initiated.
| 83 |
| Table of Contents |
While biologic therapies, including drugs such as Enbrel, Cosentyx, Humira, and Stelara, are available for treatment of psoriasis, their use remains highly restricted to patients with moderate-to-severe disease. In the U.S., with less than 20% of patients having moderate-to-severe psoriasis, we believe a vast majority of the psoriasis market are left without long-term effective treatment options. While additional data has caused an uptake of biologics, they still remain limited due to multiple factors, including being indicated only for use in moderate-to-severe patients, high costs, consequent reimbursement and access restrictions, frequent high patient co-pays, perceived risk of side effects, and patient fear of injection. Additionally, we believe there is room for topical products that treat mild psoriasis to be priced at a premium compared to other topical products for other inflammatory skin diseases like acne. This is due to the fact that when comparing the cost of biologic therapy, which can cost $50,000 per year, an effective and safe topical product for mild psoriasis could have a large impact on the market. Therefore, we believe physicians would be more likely to prescribe a topical product for psoriasis long before biologic treatment, and patients experiencing milder psoriasis would prefer using a topical product over a systemic treatment.
Non-biologic systemic therapy options for psoriasis exist, but their use is also limited due to unfavorable side effects. Apremilast (Otezla), an oral PDE4 inhibitor, generated more than $1 billion in sales in all indications in 2019, but has only achieved a small patient share in psoriasis due to limitations on its use to moderate-to-severe patients, its modest symptomatic improvement, and frequent adverse events. We believe there is still a great need to bring to market a product that addresses mild-to-moderate psoriasis.
Due to the shortcomings of existing topical therapies and the lack of options providing robust symptomatic improvement with chronic treatment, especially in the last 25 years, we believe there remains a need for a safe, effective and easily applicable topical treatment for chronic disease, that has a low risk of side effects, is well tolerated, and can be easily applied on all anatomical areas.
Our solution for Mild-to-Moderate Psoriasis. Similar to the needs of patients with acne, we believe patients suffering with psoriasis might comply better with a treatment that is easy to apply and requires less application time than current treatment options. DMT310, if approved, could be used as a first line therapy for patients with mild-to-moderate psoriasis. We believe the spicules within DMT310 will help break up the psoriatic lesions, while the anti-inflammatory components of DMT310 assist with the healing of the lesions. Due to the historical use of Spongilla and the human safety data collected to date in our clinical studies, we believe DMT310 may be suitable for long term treatment of chronic psoriasis due to its unique treatment effect and acceptable safety and tolerability profile.
In addition to the mechanical effects of DMT310, extracts of the organic material have shown in-vitro to have a dose dependent inhibition of IL-17A and IL-17F secretion. In the dermis and epidermis, T lymphocytes interact with antigen-presenting cells and secrete Th1 and Th17 cytokines, including interferon-gamma (IFN-g), interleukin (IL)-2, IL-17, IL-22, and tumor necrosis factor alpha (TNF-α). These activated T cells and the inflammatory cytokines they secrete are believed to induce the skin lesions seen in psoriasis and be a fundamental contributor in the disease’s immune pathway. We know that there are multiple approved IL-17A inhibitors on the market, such as secukinumab (Cosentyx®, Novartis) and ixekizumab (Taltz®, Eli Lilly and Co.), but these are both biologics and only indicated for patients with moderate-to-severe psoriasis who are candidates for systemic therapy. The limited patient population who are actually candidates for these biologic treatments is a small percentage of the overall psoriasis market. Therefore, a topical product that can inhibit the IL-17 pathway in the skin with minimal systemic exposure would be an ideal option for both physicians and patients.
Based on clinical and non-clinical data generated to date for DMT310, and anecdotal evidence of DMT310’s effect on psoriatic lesions, we completed a Phase 1b, open label, POC study in mild-to-moderate psoriasis patients in October 2021. This trial included once-weekly treatments of DMT310 for 12 weeks in 30 mild-to-moderate psoriasis patients with lesions covering 2% to 30% of their body surface area. The primary endpoints in this trial were the Physician’s Global Assessment, which is a 6-point scale measuring the physician’s assessment of psoriasis severity of the target lesion site, the Psoriasis Area Severity Index scale is also a 6-point scale measuring the psoriatic disease severity taking into account qualitative lesion characteristics (erythema, thickness, and scaling) and degree of surface area involvement and the Pruritus Visual Analog Scale consists of the patient’s measurement of pruritus, or itch, in addition to normal tolerability and safety assessments. We announced top-line results in October 2021, and based on the efficacy, safety and tolerability profile seen in the POC trial we initiated additional work to better inform the clinical trial design prior to moving into a larger Phase 2, placebo-controlled, clinical trial, which is anticipated to begin in the first half of 2022.
| 84 |
| Table of Contents |
DMT310 for the Treatment of Moderate-to-Severe Rosacea
Based on the positive clinical data we observed against inflammatory lesions in our Phase 2b acne trial and the many similarities between acne vulgaris and papulopustular rosacea, we are also developing our DMT310 product for the treatment of papulopustular rosacea, or rosacea. We believe the broad anti-inflammatory results seen by DMT310 in clinical trials for acne may also be seen in papulopustular rosacea patients, as it is also a disease of papules and pustules, which are inflammatory lesions on the face. We believe there currently remains a high unmet need in rosacea for a safe and effective treatment and we believe a once-weekly, naturally derived topical product could be an ideal candidate to fill this void. Thus, in November 2021, we started enrolling moderate-to-severe rosacea patients in a Phase 2 trial of the once-weekly treatment of DMT310. We expect to have top-line results in the second half of 2022.
Market Opportunity. Rosacea is a chronic, inflammatory skin disorder characterized by repeated remissions and exacerbations of facial redness and inflammatory lesions that can be challenging to treat. The pathogenesis of rosacea is not completely understood, but factors such as microbial overgrowth, vascular abnormalities, and abnormally high levels of cathelicidin in the skin have been suggested to be involved. Rosacea is further divided into four subtypes; erythematotelangiectatic, papulopustular, rhinophymatous, and ocular, with many patients having a combination of the various subtypes.
There are over 16 million patients diagnosed with one or more forms of rosacea in the U.S. and it typically affects women, although more severe forms tend to affect men due lack of treatment. Depending on the presenting symptoms and their severity, treatment ranges from oral antibiotics to topical preparations including brimonidine, oxymetazoline, azelaic acid and others, all with substandard efficacy and associated side effects including stinging and burning, pruritus, as well as dryness and erythema. These issues with current treatment options can often lead to discontinuation of treatment and a prolongation of symptoms and patients suffering. Even with such drawbacks to current therapies, current topical products most commonly used to treat rosacea generate approximately $374 million in aggregate annual revenues in the U.S. in 2019, while the oral drugs typically prescribed for more severe cases are oral antibiotics and generate about $305 million in aggregate annual revenues in the U.S. in 2019 according to IQVIA Inc. market data.
Current rosacea treatments have some of the same limitations and compliance issues as current acne products, like cumbersome applications regimens and unwanted side effects. We believe this creates a large opportunity in the market for products that are easy to apply, such as DMT310.
Limitations of Current Standard of Care. Since rosacea is a complex chronic disease combination therapy is typically required. Current topical treatment options for rosacea must be applied once or twice a day, and rosacea can have a significant impact on a patient’s quality of life due both to the unwanted cosmetic appearance of the disease and intolerable and painful symptoms associated with the disease. Moreover, with a once or twice a day application schedule, patients may experience side effects (such as dryness, stinging, burning, irritation and pruritis) after each treatment, resulting in poor patient compliance. Similar to acne, compliance with a product’s application regimen is critical in the treatment success of rosacea, especially because rosacea tends to intensify in the presence of certain environmental triggers. If left untreated, the hypertrophied tissue may require surgery to reshape and debulk the excess tissue. Despite the negative impact of the disease, very few rosacea patients seek treatment. We believe this is due to the limited treatment options available for these patients. Therefore, we believe an easy to apply once-weekly treatment with minimal side effects and good tolerability could help alleviate some of the drawbacks with the current standard of care.
| 85 |
| Table of Contents |
Our Solution for Moderate-to-Severe Rosacea. Similar to the needs of patients with acne, based on a recent survey of rosacea patients, many indicated a low level of satisfaction with current treatment options leading to intermittent use of current therapies. We believe that a once-weekly product derived from a natural source that effectively treats the inflammation associated with papulopustular rosacea may be the ideal solution for patients suffering from this disease. The formation of rosacea lesions is thought to be driven by early recruitment of TH1 and TH17 lymphocytes which ultimately causes neutrophil infiltration and lesion formation. Once the T-cells arrive to area, T cell derived cytokines such as IL-17 may, together with UV radiation, activate keratinocytes to produce the chemokines CXCL1 and CXCL8 and these chemokine cause neutrophils to migrate to the area, leading to the formation of inflammatory lesions. TH17 cells preferentially express the chemokine receptor CCR6, and its specific ligand CCL20 is upregulated by UVB irradiation stimulation in keratinocytes. This stimulation of the immune response by UV radiation may explain why sunlight exposure and IL-17 may exacerbate the lesions of rosacea.
In addition to the promotion of neutrophil infiltration, IL-17 also promotes angiogenesis via the induction of VEGF, producing erythema and subsequent telangiectasia. Based on in-vitro data, DMT310 has shown a dose dependent inhibition of IL-17A and IL-17F along with potent anti-inflammatory properties seen in our Phase 2b acne trial. We believe the combination of these features, coupled with a once-weekly application schedule, could allow for DMT310 to be a leading treatment option for patients suffering with rosacea.
We recently enrolled our first patients in a Phase 2 clinical trial of DMT310 for the treatment of moderate-to-severe rosacea in November 2021. This trial will enroll approximately 180 patients across the U.S., where patients will be treated once-weekly with a topical application of either DMT310 or placebo. Patients will be followed for 12 weeks and the primary endpoints will include Investigator’s Global Assessment and inflammatory lesion counts along with tolerability and safety assessments. Conditioned upon the receipt of positive data from this Phase 2 trial, which we expect in the second half of 2022, we plan to hold an End of Phase 2 meeting with the FDA in the first half of 2023, and then plan to initiate a Phase 3 program to prepare for an NDA submission. We expect to initiate the Phase 3 program in the second half of 2023, with top-line trial results expected in 2025. However, the foregoing timeline is subject to potential delay pending the resolution of our request to the FDA for a waiver of the requirement to conduct a 90-day dermal minipig study, as discussed below with respect to DMT310 for the treatment of acne. We will leverage the nonclinical studies completed for our acne program to help support the NDA submission for rosacea. There can be no assurance that DMT310 will receive FDA approval in rosacea.
DMT400 for the Topical Delivery of Macromolecules
DMT400 is our combination treatment regimen that utilizes the unique mechanical features of our Spongilla technology to facilitate the intradermal delivery of macromolecules, such as botulinum toxin, monoclonal antibodies or dermal fillers, by topical application rather than with injections. These macromolecules are highly effective and approved for the treatment of multiple medical and aesthetic skin conditions, but currently are not approved in a topical form because the molecular structures are too large to penetrate the stratum corneum, the skin’s outermost defense barrier. Thus, all current macromolecule treatment options for skin conditions and diseases must be injected, sometimes requiring numerous injections. We believe that DMT400’s topical application regimen may provide patients with a topical treatment option for both medical and aesthetic dermatology conditions using products previously unavailable to them in a topical treatment.
DMT400 works by first topically applying our proprietary sponge powder to the treatment area wherein the mechanical spicules of the sponge penetrate the skin thereby creating microchannels into the dermis. Unlike a derma roller or other microneedle technology, our unique spicules remain in the skin for one to two days allowing the microchannel to remain open rather than close, as they would after using a derma roller, during which time a macromolecule can be applied topically to the skin. We believe this topical application of a macromolecule can be massaged into the newly created microchannels thereby facilitating the delivery of the macromolecule, through the microchannel and into the dermis, without the need for injections. This targeted delivery to the dermis rather than delivery to the systemic circulation, may decrease the systemic spread of these macromolecules thus potentially reducing side effects seen with injections, while increasing targeted application to where the disease resides.
DMT410 for the Treatment of Primary Axillary Hyperhidrosis
We initially tested our DMT400 treatment with our DMT410 program, which consists of a topical application of our proprietary sponge powder followed by a topical application of botulinum toxin. DMT410 was initially tested in a Phase 1b POC trial of ten (10) patients with primary axillary hyperhidrosis to determine if our Spongilla sponge powder could successfully facilitate the intradermal delivery of botulinum toxin and potentially other macromolecules. Based on the results seen from this study we believe we were successful in delivering active botulinum toxin to the dermis for the treatment of hyperhidrosis.
| 86 |
| Table of Contents |
Market Opportunity. Hyperhidrosis is a life-altering disorder of excessive sweating out of proportion with thermoregulatory requirements. While many patients may exhibit this excessive sweating in response to specific triggers, such as emotional stress, others may exhibit symptoms spontaneously. Typically, the diagnosis of hyperhidrosis is based partly on subjective measures that measure how the excessive sweating affects a patient’s quality of life. Physicians also gravimetrically measure the amount of sweat produced, though there is no standardized threshold which defines hyperhidrosis. It is believed to affect an estimated 15 million people in the U.S. alone. According to a 2016 update on the prevalence and severity of hyperhidrosis in the U.S., axillary (underarm) hyperhidrosis, is the most common form of the disorder. However, patients are affected by other forms like palmar (hands) and plantar (feet) hyperhidrosis, which we believe DMT410 may be able to treat and avoid the side effects seen in studies of poor administration of intradermal injections.
Limitations of Current Standards of Care. While the prevalence of hyperhidrosis is significant, treatment options are limited, and many come with unwanted side effects making patient acceptance low. Typical first line therapy is usually with aluminum chloride based antiperspirants, but many have potential drawbacks. First, daily applications can be time consuming leading to poor compliance among patients. Second, many antiperspirants are irritating to the skin leading to treatment discontinuation. Lastly, topical aluminum chloride treatment has a transient duration of effect and requires frequent reapplication to maintain sweat control. More recently, topical anticholinergics have been investigated by companies such as Brickell Biotech, Inc. and Journey Medical Corporation (formerly developed by Dermira, Inc.), but we believe they tend to have the same side effects as systemic anticholinergics which are used off-label. These side effects include dry mouth, dry eyes, blurred vision, headache, urinary retention, among others. The unwanted side effects are often so intolerable that up to one third of patients are forced to withdraw from treatment. If topical or systemic treatments fail, then patients are able to get intradermal injections of botulinum toxin which has been shown to have a great treatment effect, but treatment is very technique driven, requiring a trained physician to administer the toxin to the thin layer of the dermis. Many times, poor treatment response with botulinum toxin is due to incorrect or insufficient dosing or incorrect administration. Patients may also experience injection site pain or discomfort, which may be accompanied by swelling and bruising. However, for the treatment of palmar hyperhidrosis with intradermal injections of botulinum toxin, the most notable adverse event is transient hand weakness, if administered incorrectly. As a last resort, patients may also seek surgery to treat their hyperhidrosis, if less invasive treatment options fail. While there are treatment options available for hyperhidrosis patients, only about half of affected individuals seek treatment due to social embarrassment associated with the diagnosis of the disease. We believe this leaves a wide gap in the market for a product that combines the efficacy of botulinum toxin with the safety and tolerability profile of topical therapies. We believe DMT410, if successfully commercialized, could address this market opportunity.
Our Solution for Primary Axillary Hyperhidrosis. While primary axillary hyperhidrosis is idiopathic, the mechanism is thought to be neurogenic overactivity of the eccrine (sweat) glands in the affected area. Based on the summary basis for approval of BOTOX, we know that botulinum toxin type A has a clinical effect on hyperhidrosis, which acts by disrupting sympathetic stimulation to the eccrine glands resulting in considerably reduced axillary sweating from four to 12 months. Based on the package insert for BOTOX, intradermal injections of 50 units of BOTOX saw a greater than 50% decrease in axillary sweat production in 81% and 41% of patients treated with BOTOX or placebo, respectively, at four weeks. While intradermal injections of botulinum toxin appears to be very effective, the treatment requires multiple injections into each axilla, which is time consuming for the treating physician and administration is very technique sensitive due to the thin nature of the dermis. Additionally, given the nature of the target tissue being more sensitive, and the number of injections required, it is believed that injection site pain is a major cause for the lack of compliance. Thus, we believe a topical application regimen, capable of penetrating the stratum corneum to deliver botulinum toxin into the dermis, may be able to exhibit similar efficacy with greater compliance and adoption. In a Phase 1b POC study of DMT410 for the treatment of axillary hyperhidrosis, we saw a greater than 50% decrease in axillary sweat production in 80% of patients at week four. This result supports our belief that topical application of botulinum toxin via our Spongilla technology to the dermis could be a viable alternative to intradermal injections. If approved, DMT410 could eliminate the need for intradermal injections of botulinum toxin. This is why we believe DMT410 could potentially be a favorable treatment option for patients suffering with primary axillary hyperhidrosis. Additionally, we also believe that DMT410 may be an effective treatment for palmar or plantar hyperhidrosis due to DMT410’s delivery of botulinum toxin to the dermis without the risk of distance spread of toxin to the muscle as with intradermal injections of botulinum toxin. DMT410 could limit the side effects seen with intradermal injections including hand weakness and administration pain. There can be no assurance that DMT410 will receive FDA approval for hyperhidrosis.
| 87 |
| Table of Contents |
DMT410 for the Treatment of Aesthetic Conditions In addition to the use of DMT410 in the treatment of hyperhidrosis and other medical dermatology conditions such as acne and rosacea, based on the data from our recent Phase 1b POC trial of DMT410 for the treatment of multiple aesthetic skin conditions such as pore size, sebum production, fine lines, luminosity, and brightness of the skin, we believe DMT410 has an opportunity to be used for the treatment of multiple aesthetic skin conditions. Botulinum toxin is known to treat a variety of aesthetic skin conditions, but to achieve these positive effects, the botulinum toxin needs to be delivered to the dermis rather than the muscle to have the desired effect. DMT410’s uniquely sized spicules create microchannels through the stratum corneum and into the dermis that are large enough for botulinum toxin to be delivered to the dermis. However, the spicules are not long enough to reach the muscle layer, which limits the potential distant spread of toxin and potential side effects. Botulinum toxin acts by blocking the release of acetylcholine into the synaptic cleft, where it binds to a cholinergic receptor, thereby inhibiting sympathetic nerve function. This ability to inhibit cholinergic transmission makes it useful to treat indications with glandular hypersecretion, like sebum production or hyperhidrosis, which are partly caused by hyperactive sympathetic nerves. For aesthetic indications, since botulinum toxin must be delivered to the dermis, intradermal injections are typically required but can require numerous injections in order to cover the larger surface area to treat these aesthetic skin conditions as compared to targeted injections to the muscle. Intradermal injections can also be difficult to effectively administer, and may be painful for patients. This tends to lead to poor adoption of this therapy, which is why we believe there are currently no approved aesthetic indications utilizing intradermal injections. Additionally, no topical formulations of a botulinum toxin have been approved, likely due to the size of the molecule and its difficulty in penetrating the stratum corneum to reach the dermis. Therefore, with no currently approved intradermal injections or topical applications methods of botulinum toxin for aesthetic skin conditions, we believe there is a large market opportunity for a product that can successfully deliver botulinum toxin by topical application into the dermis to improve a patient’s aesthetic appearance. If approved, we believe DMT410 can address this market.
Limitations of Standards of Care While injections of botulinum toxin into the muscles have been approved for many years for aesthetic treatments such as the reduction of glabellar, lateral canthal or forehead lines, there are many other aesthetic skin conditions, such as enlarged pore size, excess sebum production, fine lines, decreased luminosity, and decreased brightness that botulinum toxin has been demonstrated to improve but a botulinum toxin product, whether via intradermal injections or topical application, has yet to be approved for these indications. This may be due to the fact that these aesthetic indications typically require botulinum toxin to be delivered to the dermis rather than the muscle, which due to the thin nature of the dermis, can be more difficult than injecting into the muscle. Additionally, the areas of the face requiring intradermal injections are much more sensitive and thus can be more painful for patients. These intradermal aesthetic indications typically require a wider dispersion of botulinum toxin to be delivered to the dermis rather than the few injections into the muscle needed for the deeper facial lines. Some intradermal studies have required 25-30 intradermal injections in the face in order to deliver sufficient quantities of botulinum toxin to the dermis. With some patients having a fear of needles, a treatment that can avoid the use of needles would be desirable for this population. Additionally, topical application of botulinum toxin has been difficult due to the size of the molecule making it difficult for the botulinum toxin to penetrate the stratum corneum, resulting in many topical applications being ineffective and discontinued. An example is Revance’s RT001 product which did not achieve the primary or other secondary endpoints in a Phase 3 trial for the treatment of crow’s feet and therefore, Revance does not plan to continue development of this program at this time. Another botulinum toxin company, Allergan (now part of AbbVie), purchased a company in 2016 to pursue development of a topical botulinum toxin program. However, Allergan has not conducted any studies with this program. We believe most botulinum toxin companies remain interested in developing a topical means of administering botulinum toxin that is less painful, easy to apply, provides wider coverage of toxin, and limits potential distant spread of toxin, but no product has yet been successfully developed.
Our Solution for the Treatment of Aesthetic Skin Conditions We believe a product candidate like DMT410, which may be able to successfully deliver botulinum toxin to the dermis covering a larger facial area than injections, would provide a new treatment option for a variety of aesthetic skin conditions, such as reduction in pore size, sebum production, and fine lines, and improvement in skin luminosity and brightness, thus potentially expanding the market for uses of botulinum toxin beyond injections into the muscle for treatment of deep lines. We believe DMT410 may be able to provide patients with a topical treatment option without the pain and discomfort typically associated with injections of botulinum toxin. Additionally, we believe administration will be easier and less time consuming for dermatologist, making it an additional value driven treatment option they can offer. While current botulinum toxins are approved for injections into the facial muscles to treat deeper wrinkles such as glabellar lines, lateral canthal lines and forehead lines, there remain many other aesthetic conditions of the face which could greatly benefit from an intradermal administration of botulinum toxin, especially via topical application. Certain aesthetic indications such as reduction in fine lines, pore size, and sebum production, and improvement in luminosity and brightness are typically not treated with injections into the muscles but need botulinum toxin to be delivered to the dermis to have the proper effect. With DMT410’s uniquely sized spicules, we believe it can create numerous microchannels into the dermis allowing a pathway for the topical application of botulinum toxin. Once in the dermis botulinum toxin is able to act on reducing sebum production which in turn may reduce pore size and overall oiliness of the skin. Additionally, botulinum toxin has been demonstrated to have a beneficial effect on the mean volume and depth of facial lines giving the skin a smoother appearance. Therefore, we believe there may be a need for a product, such as DMT410, that can facilitate the topical application of botulinum toxin into the dermis, to treat a variety of these aesthetic skin conditions.
| 88 |
| Table of Contents |
If approved, we believe DMT410 has the potential to expand the market for botulinum toxins’ treatment to multiple additional aesthetic skin conditions. We believe DMT410 can be an effective product at delivering bioactive botulinum toxin with a topical application for treatment of pore size, sebum production, fine lines, luminosity, brightness, overall aesthetic appearance and possibly more. We recently completed a Phase 1b POC trial of DMT410 for the treatment of multiple aesthetic skin conditions where we examined improvements in pore size, sebum product, luminosity, brightness and Global Aesthetic improvement after one treatment of DMT410. We believe this POC trial produced data which demonstrated that DMT410 was able to deliver botulinum toxin to the dermis and showed a reduction in pore size, reduce sebum production, improved luminosity, improved brightness, and improved the patients’ Global Aesthetics. This study also produced no adverse events and provided acceptable tolerability data with only mild tolerability effects seen fifteen (15) minutes post treatment. We believe this data warrants further development of this program and we are currently seeking a partner who has a botulinum toxin they are looking to develop for aesthetic skin conditions which require delivery of botulinum toxin to the dermis. There can be no assurance that DMT410 will receive FDA approval for any aesthetic indication or that we will be able to find a partner for development.
Clinical Progress of our Lead Product Candidates
DMT310 Phase 2b Clinical Results for Acne
In June 2020, we received results from our randomized, double-blind, placebo-controlled, Phase 2b clinical trial of once-weekly treatments for twelve weeks of DMT310 for acne. We enrolled 181 moderate-to-severe acne patients at fourteen (14) sites across the U.S. Patients were required to be 12 years of age or older, have at least twenty (20) non-inflammatory lesions, twenty (20) inflammatory lesions, no more than two (2) nodules or cysts and be a moderate or severe (meaning a 3 or 4) on the IGA scale of acne. The IGA scale consists of a 5-point scale, 0-4, with 0 being clear, 1 being almost clear and 4 being severe acne as graded by the treating physician. Patients were randomly divided into two treatment groups, either to receive DMT310 or placebo. Patients were required to apply the product, whether DMT310 or placebo, to the entire face, once-weekly for 12 weeks with the first two weeks of treatment applied in office under the supervision of trained study staff, then the remaining 10 weekly treatments were applied at home by the patient.
The primary clinical endpoints of the trial included the absolute reduction in inflammatory lesions from baseline. The secondary clinical endpoints included:
| • |
| the absolute reduction in non-inflammatory lesions from baseline; |
| • |
| the IGA with a responder being a patient with a 2-grade change in IGA scale and being a 0 or 1 at study exit; and |
| • |
| safety and tolerability. |
| 89 |
| Table of Contents |
All statistical analyses and data shown for our Phase 2b study are on the intent-to-treat, or ITT, population. The ITT population included all randomized subjects in the group to which they were randomized, regardless of study drug received or if they completed the study. The ITT approach provides an unbiased comparison among the treatment groups.
The trial was completed in June 2020 and showed a statistically significant and we believe, a clinically meaningful effect for all efficacy endpoints of the trial, namely reduction in inflammatory and non-inflammatory lesions and IGA responders. Notably, the treatment effect for all efficacy endpoints was statistically significant at the four-week mark, after only four treatments, and continued to be statistically significant for week eight and week 12 when compared with placebo. Patients saw a rapid and sustained therapeutics treatment effect in percentage change in inflammatory lesions with a 45% reduction in inflammatory lesions at week four and reached 62% reduction in inflammatory lesions at week 12 compared to 24% and 42% reduction of inflammatory lesions for placebo at weeks four and 12, respectively. The effect on non-inflammatory lesions were also statistically significant after four weeks, with the therapeutic effect of an approximately 36% reduction in non-inflammatory lesions at week four reaching a therapeutic effect of 58% at 12 weeks, which was statistically significant when compared with placebo. In addition, both inflammatory and non-inflammatory percent reduction in lesion counts had P-values of less than 0.001 at week four and week 12 when compared to placebo.
We also saw an early statistically significant separation in IGA with 15% of patients in the DMT310 group considered responders on the IGA scale, or who had an IGA score of 0, “clear,” or 1, “almost clear,” after only 4 treatments as compared with just over 2% of patients on placebo. This statistical separation continued for the remainder of the trial where at the completion of the trial, or week 12, 44% of patients in the DMT310 group, compared with 17% in the placebo group were IGA responders. This difference was statistically significant with a P-value of less than 0.001.
No reported drug-related severe adverse events were reported in the trial. The drug also appeared to be tolerable by a majority of patients with greater than 92% of patients experiencing no or mild tolerability with no severe dryness, scaling, erythema, or burning/stinging reported at week 12. Of those patients who did report tolerability issues, they also reported that the issues were usually transient and resolved quickly without intervention. Based on this data, after meeting with the FDA, we expect to proceed to Phase 3 clinical trials with the same clinical endpoints and the same formulation of DMT310 for moderate-to-severe acne.
The following diagrams and tables show the absolute reduction of inflammatory and non-inflammatory lesions from baseline until the end of study, or week 12, for both DMT310 and placebo (Image 3), and percent reduction of inflammatory and non-inflammatory lesions from baseline until end of study, or week 12, for both DMT310 and Placebo (Image 4). Although reduction in non-inflammatory lesions was a secondary endpoint of this trial, it is a required metric for the Phase 3 acne studies necessary for FDA approval.
| 90 |
| Table of Contents |
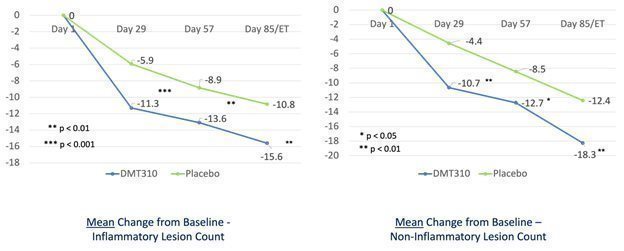
Image 3: Mean reduction of inflammatory and non-inflammatory lesions from baseline until end of study, or week 12, for both DMT310 and Placebo
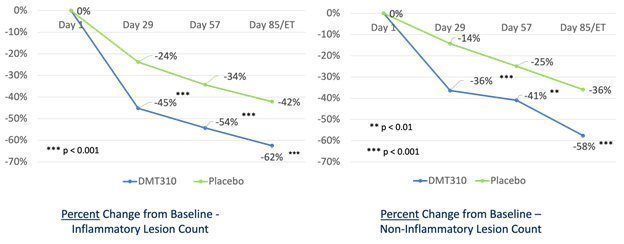
Image 4. Percent reduction of inflammatory and non-inflammatory lesions from baseline until end of study, or week 12, for both DMT310 and Placebo
| 91 |
| Table of Contents |
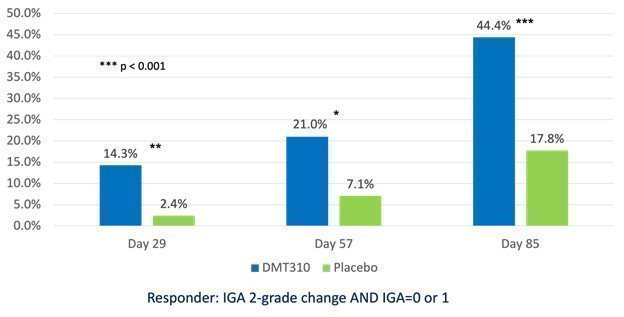
Image 5. Investigator Global Assessment response rate at Day 29, or week 4, Day 57, or week 8 and Day 85, or week 12
(1) | As used in the diagrams and table above, the reference to “P-value” (relative to placebo) means the probability of being wrong when asserting that a true difference exists between the results for the relevant patient group and the placebo group. For example, a “P-value” of less than 0.001 indicates that there is a less than one in 10,000 chance that the observed result in the treatment group and the observed result in the placebo group are the same. A “P-value” equal to or less than 0.05 means that a given difference is statistically significant. |
(2) | “Success” is defined as an IGA score of “clear” or “almost clear” upon completion of the study. |
Additionally, the safety and tolerability profile of DMT310 appeared to be acceptable with a small number of patients experiencing treatment emergent adverse events as seen in Image 6 below. In this Phase 2b trial, no subject receiving treatment with DMT310 experienced a severe local skin reaction at study end nor did any patient undergo a dose modification. Most tolerability issues were mild and resolved shortly after application without any rescue medication as seen in Image 7 below. Overall, DMT310 was generally safe and well tolerated by patients when applied once-weekly for 12 weeks.
System Organ Class Preferred Team |
| DMT310 |
| Placebo |
General disorders and administration site conditions |
| 5(5.5) |
| 2(2.2) |
Application site erythema |
| 4(4.4) |
| 1(1.1) |
Application site pruritus |
| 2(2.2) |
| 2(2.2) |
Application site dryness |
| 1(1.1) |
| 0(0.0) |
Application site exfoliation |
| 1(1.1) |
| 0(0.0) |
Image 6. Treatment Emergent Adverse Events
| 92 |
| Table of Contents |
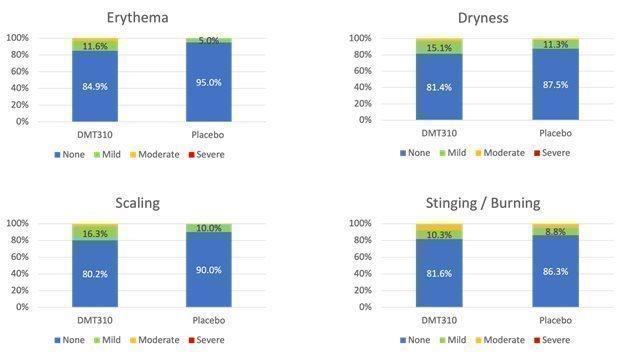
Image 7: Local Tolerability
DMT310 Phase 2a Clinical Results for Acne
In 2018 we conducted the first clinical trial of DMT310, a randomized, double-blind, 2x2 factorial, placebo-controlled, Phase 2a clinical trial of DMT310 for the treatment of acne. We enrolled 121 patients to evaluate the tolerability, safety, and efficacy of DMT310 mixed with 3% H2O2 following 12 weeks of topical administration in male and female patients with moderate-to-severe facial acne. The study employed a 2x2 factorial design in order to assess the contribution of each component of the investigational product (i.e., Spongilla lacustris topical powder and 3% H2O2 USP). This Phase 2a clinical trial employed the same clinical endpoints as our Phase 2b clinical trial of DMT310 for the treatment of acne, as discussed above.
Patients were randomly divided into one of four treatment groups, DMT310 + 3% H2O2, DMT310 + Water, Placebo + 3% H2O2, or Placebo + Water (control). The patients were required to apply the assigned study drug to their entire face up to once-weekly for 12 weeks (84 days), beginning on Day 1 and through Day 78 (as applicable). During study center visits on Days 29 and 57, a determination was made for each patient, based on the Investigator’s Global Assessment (IGA) score, as to whether study drug application would continue once-weekly or at a lower biweekly frequency (once every 2 weeks). Specifically, patients with an IGA > 1 at the Day 29 or Day 57 visits continued with once-weekly study drug applications, while patients with an IGA 1 at these same visits were instructed to subsequently apply their assigned study drug biweekly (see Image 8 below for a presentation of the study drug application frequency algorithm).
| 93 |
| Table of Contents |
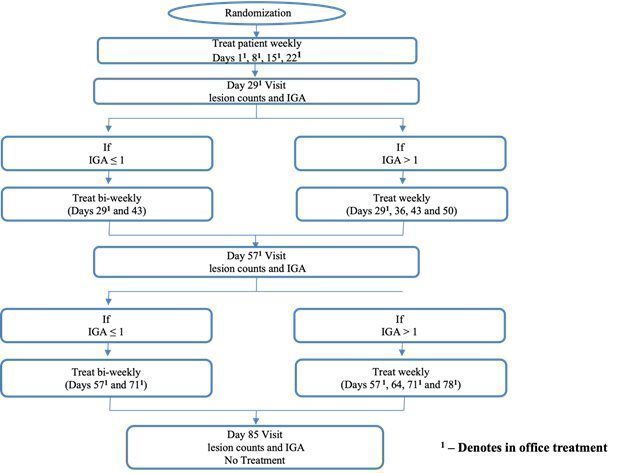
Image 8 Application Frequency Algorithm by Study Visit
This Phase 2a trial showed statistically significant and clinically meaningful effects for its endpoint, absolute reduction in inflammatory lesions when comparing DMT310 + 3% H2O2 and placebo + water (control) groups at both week 8 and week 12 or end of study. At week 12, DMT310 + 3% H2O2 had a 16 lesion reduction from baseline while placebo + water had an 11 lesion reduction from baseline, with a p-value of less than 0.05. Across all the treatment groups, there was a mean decrease from baseline (i.e., improvement) in the inflammatory and non-inflammatory lesion counts at Days 15, 29, 57, and 85. However, there were no statistically significant differences between the DMT310 + 3% H2O2 group versus the placebo + water (control) group for the mean changes from baseline in non-inflammatory lesion counts.
We did not observe a statistically significant difference between the DMT310 + 3% H2O2 group and the placebo + water (control) group in the percentages of patients with IGA treatment success (IGA score of 1 or 0 and a 2-grade change) or patients with 1-grade or 2-grade improvements from baseline in IGA at any visit. At Day 85 in the DMT310 + 3% H2O2, DMT310 + water, placebo + 3% H2O2, and placebo + water groups, 29.6%, 20.0%, 27.6%, and 34.5% of patients had IGA treatment responders, respectively; 66.7%, 66.7%, 65.5%, and 55.2% of subjects had a 1-grade improvement from baseline in the IGA, respectively; and 37.0%, 20.0%, 27.6%, and 34.5% of subjects had a 2-grade improvement from baseline in the IGA, respectively.
Analysis of Placebo: After speaking with the clinical investigators in the trial, it was determined that the higher than normal placebo response in non-inflammatory lesions may have been partially due to the stickiness of the placebo, which required patient to scrub their face, resulting in exfoliation of the skin and removal of sebaceous plugs (whiteheads and blackheads), thereby reducing non-inflammatory lesion counts. We believe this caused a placebo response rate that was much higher than the placebo response rates seen in other topical Phase 2 acne trials. As a result of the inadequate placebo, we developed a new proprietary placebo formulation for our Phase 2b clinical trial.
| 94 |
| Table of Contents |
Once-weekly vs. Biweekly Treatment Schedule: Due to this being the first time DMT310 was studied in moderate-to-severe acne patients for 12 weeks, a conservative study design was chosen to ensure patient tolerability and safety while attempting to maintain efficacy. Therefore, we allowed IGA treatment responders (IGA score of 0 or 1) to move to a biweekly or every other week application schedule as discussed above. This resulted in 3 out of 27 patients in the DMT310 + 3% H2O2 group and 0 out of 29 patients in the placebo + water group moving from an IGA score of 1 (treatment responder) at week 8 and regressing to an IGA score of 2 (treatment failure) at week 12. Therefore, our Phase 2b clinical trial of DMT310 in moderate-to-severe acne patients incorporated only once-weekly applications for 12 weeks with no biweekly option.
No statistical or clinical difference was seen between the placebo + 3% H2O2 and the placebo + water groups, which we believe indicates that 3% H2O2 by itself does not have a treatment effect.
DMT310 next steps for acne
We are currently preceding with the necessary non-clinical and pharmacokinetic studies and once complete we plan to schedule an End of Phase 2 meeting with the FDA in the second half of 2022 to discuss and finalize the protocol for the Phase 3 clinical trials in moderate-to-severe acne.
We have submitted requests to the FDA to waive the conduct of a 90-day dermal minipig study and a standard dermal pharmacokinetic study. While we have already planned and budgeted to conduct both studies, we submitted the waiver requests because DMT310 has been used in completed clinical trials of over 170 human patients in which it exhibited an acceptable safety and tolerability profile. Further, FDA has also approved use of DMT310 in our ongoing 180 human patient Phase 2 study for the treatment of rosacea prior to initiation of these dermal minipig and dermal pharmacokinetic studies.
Typically, the FDA expects the 90-day dermal minipig study to be conducted prior to filing an IND and proceeding to trials in humans of a topical dermatology development product candidate. However, due in part to DMT310’s historical safety profile and our ability to reference the FDA’s Botanical Drug Development Guidance for Industry for certain aspects of the development of DMT310, the FDA allowed DMT310 to proceed directly into human trials without first conducting this 90-day dermal minipig study. Considering the human safety data collected from DMT310 clinical trials and the historical safety data collected to date, we believe the FDA has an adequate scientific basis for granting the waiver request with respect to the 90-day dermal minipig study. If the FDA grants the dermal minipig study waiver, we believe it would provide a cost savings of approximately $600,000 in development costs. Alternatively, the FDA may allow us to conduct the 90-day dermal minipig study concurrently with our Phase 3 studies of DMT310 in acne, provided that the dermal minipig study is completed prior to submission of a New Drug Application.
We have also submitted a request to the FDA to waive conduct of the standard dermal pharmacokinetic study. As with the minipig study, this request is based on the human tolerability and safety profile of DMT310 observed to date.
If the FDA grants both waiver requests, we plan to immediately request an End of Phase 2 meeting with the FDA and begin planning to initiate our Phase 3 acne program in the second half of 2022. If the FDA grants our waiver request only with respect to the 90-day dermal minipig study, we believe we can still complete the standard dermal pharmacokinetic study and initiate the Phase 3 program in 2022, as planned. If the FDA grants the waiver request only with respect to the standard dermal pharmacokinetic study, or rejects both waiver requests, we may be forced to delay the start of our Phase 3 program into 2023 due to supply chain constraints relating to the availability of the minipigs required for the 90-day dermal minipig study.
Once we are able to hold an End of Phase 2 meeting with the FDA and we receive approval to proceed with Phase 3, we plan to initiate a pivotal Phase 3 program for DMT310 for the treatment of moderate-to-severe acne. We expect this Phase 3 program to include two, multi-center, placebo-controlled trials with identical clinical endpoints to our recently successful Phase 2b clinical trial of DMT310 for the treatment of moderate-to-severe acne. We expect to report top-line data from this program in 2024 and, assuming positive results, file a new drug application, or NDA, with the FDA shortly thereafter. This Phase 3 program is intended to be designed to demonstrate the safety and efficacy of the treatment of DMT310 relative to placebo for the treatment of moderate-to-severe acne. Prior to, or in parallel with our planned Phase 3 program, we intend to continue conducting and complete the additional non-clinical studies necessary to file the NDA. We also intend to conduct a long-term safety study. If DMT310 is approved for the treatment of acne, we believe DMT310 can eventually be an attractive prescription to over-the-counter switch, or Rx-to-OTC, target, which could provide a substantially larger sales opportunity. There can be no assurance that DMT310 will receive FDA approval for the treatment of acne.
| 95 |
| Table of Contents |
DMT310 Phase 1a Clinical Results for Psoriasis
We recently completed a Phase 1a POC trial of DMT310 for the treatment of mild-to-moderate psoriasis. This was an open-label, multi-center, 12-week study in 30 mild-to-moderate psoriasis patients with psoriatic lesions coving between 2-30% of body surface area. The trial aimed at evaluating the tolerability, safety, and efficacy of once-weekly treatments of DMT310, which consists of 2 grams of Spongilla powder mixed with 6 mL of 3% H2O2. One mild or moderate lesion was selected, and patients were required to apply DMT310 to the entire lesion, once-weekly for 12 weeks with the first two weeks of treatment applied in office under the supervision of trained staff, then the remaining 10 weekly treatments were applied at home by the patient.
The endpoints in this study included:
| • | Physicians Global Assessment (PGA) of disease severity with success defined as absent or very mild disease, a score of 0 or 1 respectively on a 5-point scale, at the target lesion site. |
|
|
|
| • | Psoriasis Area Severity Index (PASI) at the target lesion site defined as absent or mild, a score of 0 or 1 respectively, on a 6-point scale. This required each psoriatic sign of scaling, erythema, and plaque elevation to have a score of 0 or 1 for the subject to be considered a responder. |
|
|
|
| • | Pruritus Visual Analog Scale (VAS) looking at the mean change and percent change from baseline in pruritis (itch). |
All efficacy analyses were performed using the As-Treated population, which consists of all enrolled patients who received at least one dose of study medication and the per protocol population, which consists of all enrolled patients with no significant protocol violations during the study that would affect the efficacy analyses.
The trial was completed in August 2021 and showed an acceptable safety and tolerability profile that we believe is clinically meaningful and warrants further investigation of DMT310 as a potential treatment for mild-to-moderate psoriasis. DMT310 was able to achieve a PGA score of 0 or 1 for the target lesion in 29.6% of patients at week 8. DMT310 also demonstrated a total PASI score of 0 or 1 for the target lesion in 25.9% of patients at week 8. Notably, DMT310 demonstrated a 19.6% reduction from baseline in pruritus (itch) at week 8 with a peek reduction of 22.5%. We believe these findings from a POC trial are encouraging for the potential use of DMT310 as an easy to apply topical treatment for mild-to-moderate psoriasis with an acceptable safety and tolerability profile.
No reported drug-related severe adverse events were reported in the trial and only two treatment emergent adverse events were reported, both being application site pruritus. Additionally, the drug also appeared to be tolerated by a majority of patients. Of those patients who did report tolerability issues, they also reported that the issues were usually transient and resolved quickly without intervention.
| 96 |
| Table of Contents |
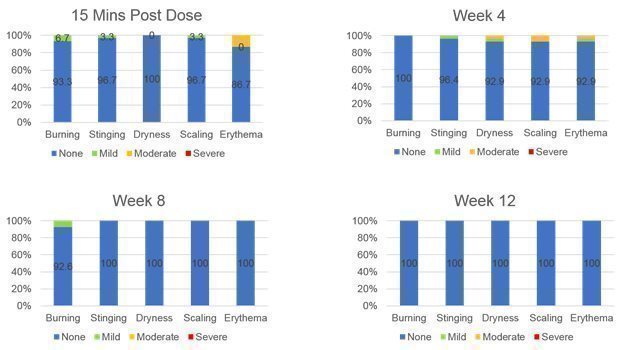
Image 9: Local Tolerability
DMT310 next steps for psoriasis
We are pleased with data already seen in our Phase 1 POC trial, especially seeing a reduction in itch as that is one of the main complaints of patients suffering from psoriasis. We are currently in the process of designing a Phase 2 study of DMT310 for the treatment of psoriasis. The Phase 2 study will be a larger randomized, double-blind, placebo control study of DMT310 for the treatment of psoriasis. Based on the data from the Phase 1b proof of concept study we are considering adding additional arms to examine once versus twice weekly treatment, potentially enhancing the treatment effect seen in our Phase 1b trial. Additionally, due to the unique nature of psoriasis and the general thickness of psoriatic plaques, we may also examine increasing the application pressure and the length of application. We believe that the thicker psoriatic plaques may require a more intensive treatment compared to the application regimen for acne where there is no thickened skin. We believe DMT310 could be a first in class treatment option for psoriasis patients and plan to initiate the Phase 2 trial in the first half of 2022 with top-line results expected in the first half of 2023. Assuming this trial is successful, we plan on requesting an End of Phase 2 meeting with the FDA in the second half of 2023 and initiating a Phase 3 program in the first half of 2024. However, the foregoing timeline is subject to potential delay pending the resolution of our request to the FDA for the waiver of the requirement to conduct a 90-day dermal minipig study, as discussed above with respect to DMT310 for the treatment of acne. There can be no assurance that DMT310 will receive FDA approval for the treatment of psoriasis.
DMT410 Phase 1b-Primary Axillary Hyperhidrosis
In the first quarter of 2019, we completed a Phase 1b, open-label, POC trial of DMT410 for the treatment of moderate-to-severe primary axillary hyperhidrosis. In this study we treated ten (10) patients with one application of our proprietary sponge powder followed by one topical application of BOTOX, reconstituted per label, to each axillae. Patients were followed for 4 weeks after which time their sweat production was measured and compared with baseline measurements.
The endpoints for this trial included:
| • | percent of patients with less than 50% reduction in gravimetrically measured sweat production from baseline, |
|
|
|
| • | percent of patients with gravimetric sweat production of greater than 50 mg, and |
|
|
|
| • | percentage change in gravimetric sweat production. |
| 97 |
| Table of Contents |
After 4 weeks, 80% of patients saw a decrease in gravimetric sweat production greater than 50%, 85% of patients had gravimetric sweat production of less than 50mg, and patients had a 75% decrease in gravimetric sweat product from baseline. Based on this clinical data, we believe that we were able to deliver botulinum toxin into the dermis through topical application for the treatment of primary axillary hyperhidrosis. Treatment with DMT410 also had an acceptable safety and tolerability profile. We plan to partner with a botulinum toxin in order to run a larger Phase 2 study of DMT410 for axillary hyperhidrosis and possibly for palmar (hand) or plantar (foot) hyperhidrosis where there is currently no approved product. There can be no assurances that DMT410 will receive FDA approval for the treatment of hyperhidrosis.
DMT410 Phase 1b- Upper Facial Lines
In November 2020, we enrolled our first patient in a Phase 1b open-label, POC trial of DMT410 for the treatment of upper facial lines along with multiple other aesthetic skin conditions that are affected by delivery of toxin to the dermis such as pore size, sebum production, brightness, luminosity, fine lines, and Global Aesthetic Improvement. Due to the fact that we do not own rights to a botulinum toxin product, we were required by the FDA to conduct this trial using an approved indication for BOTOX, upper facial lines, an approved dose for this indication (64 units of botulinum toxin) and an approved route of administration, which is typically injections into the muscle rather than the dermis. However, we were interested in studying the clinical effect of DMT410 for the aesthetic skin conditions that require delivery of botulinum toxin to the dermis rather than the muscle, because of our belief that DMT410 only delivers botulinum toxin to the dermis and not the muscle. With these constraints, we initiated the POC trial by enrolling ten (10) female patients, age 18 or older, each receiving one treatment of DMT410, consisting of one topical application of our proprietary sponge powder followed by one topical application for BOTOX, reconstituted per label, to the upper face. Patients were followed for sixteen (16) weeks to determine the achievement of our endpoints along with the duration of effect. We also collected safety and tolerability data. We received top-line data from this study in the November 2021, and believe that we achieved results in multiple aesthetic endpoints sufficient to warrant further investigation of DM410 for the treatment of various aesthetic skin conditions.
The endpoints for this trial were:
| • | Portion of patients achieving a grade of none or mild on the investigator’s assessment of lateral canthal, forehead, and glabellar lines based on the Facial Wrinkle Scale (FWS), which consists of a 5-point scale with 0 being none and 1 being almost none. To be considered a responder, both the patient and physician had to agree on the score. |
|
|
|
| • | Portion of patients achieving improvement on the physician’s assessment of pore size improvement, based on the Global Aesthetic Improvement Scale (GAIS), which consists of a 5-point scale with 0 being no improvement, 1 being less than or equal to 25% improvement, 2 being a 26-50% improvement, 3 being a 51-75% improvement, and 4 being a 76-100% improvement. |
|
|
|
| • | Mean and percent change from baseline in brightness based on the 10-point visual analog scale. |
|
|
|
| • | Mean and percent change from baseline in luminosity based on the 10-point visual analog scale. |
|
|
|
| • | Portion of patients achieving a two- grade improvement on the physician’s assessment of FWS of fine lines under the eye. |
The efficacy analysis was conducted on the ITT and Per Protocol (PP), meaning all patients completing the study without a major protocol violation, populations. In addition to the physician measured endpoints listed above, we also implemented 2-dimensional VISIA and 3-dimensional PRIMOS imaging technology from Canfield Scientific to provide objective analysis of many of the aesthetic endpoints.
Based on the assessment of forehead, lateral canthal, and glabellar lines, no patient was considered a responder in this study, while some patients did achieve at least a one-grade change throughout the course of the study. This result was not unexpected as BOTOX is only approved for injections into the muscle and we did not believe that we would see any potential distant spread of toxin outside of the dermis. Seeing no potential distant spread of toxin was encouraging for us as it provides important safety data that while DMT410 can be applied over a larger treatment area than injections, it does not appear to travel beyond the dermis which may cause unwanted facial effects.
We believe the real potential of DMT410 lies in the aesthetic endpoints that can be affected by delivering BOTOX to the dermis rather than the muscles. These clinical endpoints include pore size, global aesthetic improvement, brightness, luminosity, and fine lines. The following table (Image 10) shows the improvements in pore size, or a decrease in overall pore size, with patients achieving at least a 25% improvement in pore size, for assessment of GAI, or the overall improvement in skin quality, the with patients achieving at least a 25% improvement in GAI, for assessment of brightness, or the skin’s combined uniformity of color and texture, with patients having at least a 1-point improvement in brightness, and for assessment of luminosity, or the intensity of light area reflected off the face, with patients having at least a 1-point improvement in brightness.
| 98 |
| Table of Contents |
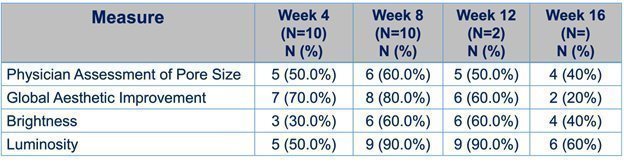
Image 10: Aesthetic Endpoints
In addition to the physician measured endpoints, we also utilized Canfield Scientific’s 2-dimensional VISIA and 3-dimensional PRIMOS imaging technologies to gather additional objective data on some of the key endpoints. Based on the VISIA system patients saw a 14.1% reduction in pore count and a 14.3% reduction in pore area at week 4. Patients also saw a 16.5% reduction in wrinkle count and a 11.5% reduction in wrinkle area at week 4. Using the PRIMOS image analysis patients saw a percent decrease in mean line, which consist of the deeper facial lines, of 12.1% and a percent change in mean roughness, which consists of the superficial fine lines, of 6.5%. We believe the subjective effects seen by the treating physicians were further validated by the objective measures using Canfield camera systems.
No reported drug-related adverse events were reported in the trial. The drug appeared to be well tolerated by patients, with only mild stinging and erythema reported 15 minutes after treatment and no tolerability issues reported at week 4, 8, 12 or 16.
DMT410 next steps for aesthetics
We are very encouraged by the results from our Phase 1b POC trial of DMT410 for the treatment of multiple aesthetic skin conditions. This was designed to be a signal detection trial of DMT410 for the treatment of a variety of aesthetic skin conditions based on the clinical trial design constraints, including the limitation on the quantity of BOTOX that we were able to apply, the clinical endpoints that needed to be included, and the area of the face that could be treated. Even with these limitations we believe that we achieved results sufficient to warrant the continued development of this program as there remains no approved botulinum toxin, whether via injection or topical, to treat many of the endpoints in which we saw a treatment effect. We believe this is further supported by the fact that many of the endpoints saw an improvement by week eight (8) or twelve (12) and started to return towards baseline at week 16. This is consistent with the knowledge that BOTOX lasts for about three (3) months before the effect begins to fade. We believe that if we can conduct a larger Phase 2 clinical trial that consists of multiple doses of botulinum toxin, we will be able to find the optimal dose for the treatment of a variety of aesthetic skin conditions including pore size, sebum production, fine lines, luminosity, brightness, and overall aesthetic improvement. We know that botulinum toxin has shown efficacy of these endpoints but there has been very little research conducted on the optimal dose or administration procedure, likely due to challenges with intradermal injections and the lack of topical applications that can effectively deliver botulinum toxin to a large enough treatment area as required to treat many of these aesthetic skin conditions. We believe DMT410 can meet this need as shown by our Phase 1b data, so we are actively seeking a partnership with a botulinum toxin company that is interesting in helping us further develop our DMT410 program for multiple aesthetic skin conditions. There can be no assurances that we are able to successfully negotiate a partnership with a botulinum toxin company or that DMT410 will receive FDA approval for the treatment any aesthetic skin conditions.
| 99 |
| Table of Contents |
Manufacturing
We do not currently own or operate any manufacturing facilities and do not plan to own any in the near future. We have been relying on our third-party partners for the manufacture of our products used in pre-clinical studies and clinical trials, and will likely continue to rely on these partners in the near term for the commercial manufacturing of our drug substance and drug product, if our drug product candidates are approved. Manufacturing of the active pharmaceutical ingredient, or API, for our product candidates requires a raw material that is derived from a natural source.
To date, we have obtained naturally sourced Spongilla raw material directly from our supplier based in Russia. In February 2020, we signed an exclusive supply agreement with this supplier of Spongilla raw material. Our supplier has over 18 years of experience collecting and processing Spongilla and has the capacity to collect and process large quantities of Spongilla per year. We believe our supplier is able to harvest sufficient quantities of raw material to fulfill our development and potential commercial needs, if a product candidate is approved using this raw material. However, we will explore alternative manufacturing sources, in order to ensure that we have access to sufficient manufacturing capacity to meet potential demand for any of our product candidates in a cost-efficient manner. See “Business-Material Agreements- Supply Agreement between Dermata Therapeutics LLC and Reka-Farm LLC” for more information regarding our supply of Spongilla.
Development and commercial quantities of any drug product candidates that we may develop will need to be harvested, manufactured in facilities, and processed in compliance with the requirements of the FDA and the regulatory agencies of other jurisdictions in which we are seeking approval. We currently employ internal resources to manage our manufacturing contractors. The relevant manufacturers of our drug product candidates have advised us that they are in compliance with both current Good Laboratory Practices, or cGLP, and cGMP.
We have relied upon our complete supply chain while supporting both our Phase 1 and Phase 2 clinical supply requirements and we are confident that our manufacturers have the ability to scale our processes to support our Phase 3 clinical studies and future commercial commitments. Our suppliers and manufactures were specifically selected based on the capabilities of their organization, their compliance to regulations, their personnel and the type and capabilities of their equipment. Testing methods for each stage of the manufacturing process form acquisition of raw materials through production of finished drug product have been developed and satisfactorily qualified per the FDA’s phase appropriate regulations relating to clinical materials for human use. Analytical methods and operational procedures related to each stage of our production operations including product release will continue to evolve and be validated as part of our overall development plan for Phase 3 clinical supplies and commercial production.
Commercialization
Given our stage of development, we do not currently have any internal sales, marketing, or distribution infrastructure or capabilities. If approved, we intend to commercialize DMT310, or any other product candidates that we may successfully develop, in the United States by building a specialized sales organization focused on dermatologists. We believe a scientifically oriented, customer-focused team of approximately 50-60 sales representatives would allow us to reach our targeted dermatologists in the U.S. with the highest potential for prescribing DMT310. In the future, we may develop and commercialize DMT310 for additional geographic regions, independently or with a strategic partner. If DMT310 is approved, and we are able to successfully commercialize it, we believe DMT310 can eventually become an attractive Rx-to-OTC switch target, which could provide a substantially larger addressable market and an expanded sales opportunity.
Competition
The medical and aesthetic pharmaceutical industries in which we plan to operate are competitive and subject to changes in practice. While we believe that our unique natural technology, knowledge, experience and resources provide us with competitive advantages, we may face competition from many different sources with respect to our current programs or any other product candidates that we may seek to develop or commercialize in the future. Possible competitors may include pharmaceutical companies, academic and medical institutions, governmental agencies and public and private research institutions. These prospective competitors have the ability to effectively commercialize, market and promote approved products, including communicating the effectiveness, safety and value of products to actual and prospective customers.
| 100 |
| Table of Contents |
Many of our prospective competitors have substantially greater manufacturing, financial, research and development, personnel and marketing resources than we do. Our prospective competitors may also have more experience and expertise in obtaining marketing approvals from the FDA and foreign regulatory authorities. In addition to product development, testing, approval and promotion, other competitive factors in the pharmaceutical industry include industry consolidation, product quality and price, product technology, reputation, customer service and access to technical information. As a result, our prospective competitors may be able to develop competing or superior products, and compete more aggressively and sustain their competitive advantage over a longer period of time than us. Our products may be rendered obsolete or may lack economic viability in the face of competition.
The key competitive factors affecting the success of DMT310, if approved, will likely be its efficacy, safety, convenience of administration and delivery, price and the availability of reimbursement from government and other third-party payors. With respect to DMT310 for the treatment of moderate-to-severe acne, if approved, we will primarily be competing with therapies such as other topical products, oral products, in-office procedures, such as laser surgery, off-label drugs, over the counter medication and homeopathic remedies. With regards to DMT310 for the treatment of mild-to-moderate psoriasis, if approved, we will face competition from topical therapies, oral therapies, systemic therapies, photo therapies and homeopathic treatments. However, based on our clinical trials, we believe that DMT310 has multiple competitive advantages over current treatment alternatives with significantly less adverse side effects. Our main competition in these indications will be with products from Vyne Therapeutics, Inc, Sol-Gel Technologies Ltd., Arcutis Biotherapeutics, Inc., Almirall S.A., Galderma S.A., Pfizer Inc. See “Business - Clinical Progress of our Lead Product Candidates” for the results of our completed and ongoing clinical trials. While we are unaware of any potential similar competitive topical products to DMT310 for the treatment of acne, psoriasis and rosacea, it is possible that such potentially similar competitive products are currently being developed.
We are also in early stages of clinical development for DMT410 for treating various medical and aesthetic skin conditions, and if we obtain marketing approval in the future, we would compete with traditional therapies, such as topical products, oral products, in-office procedures, such as botulinum toxin injections, off-label drugs, over the counter medication and homeopathic remedies, as well as additional new entrants to the applicable markets.
We also expect to face competition in our efforts to identify appropriate collaborators or partners to help commercialize our product candidate portfolio in our target commercial markets.
Intellectual Property
Overview
Our commercial success depends in part on our ability to obtain and maintain proprietary protection for DMT310, DMT410 and any of our future product candidates, medical devices, methodologies, assays, drug development technologies, harvesting procedures, know-how; to operate without infringing on or otherwise violating the proprietary rights of others; and to prevent others from infringing or otherwise violating our proprietary rights. Our strategy is to protect our proprietary position by, among other things, filing U.S. and foreign patent applications related to our product candidate and other proprietary technologies, inventions and improvements that are important to the development and implementation of our business. We also rely on trade secrets, trademarks, know-how, continuing technological innovations, exclusivity agreements, nondisclosure and confidentiality agreements, license agreements, assignment of inventions and potential in-licensing opportunities to develop and maintain our proprietary position.
Patent Portfolio
Our patent estate consists of in-licensed and solely owned patent applications. Typically, we initially file U.S. provisional patent applications and then file applications directly or under the Patent Cooperation Treaty, or PCT, which is an international patent law treaty that provides a unified procedure for filing a single initial patent application to seek patent protection from an invention simultaneously in any one of the designated member jurisdictions and states, including in the U.S. Although a PCT application does not issue as a patent, it allows the applicant to seek protection in any of the member states through national-phase applications filed at a later date. We currently have multiple patent or patent applications in our patent portfolio and continue to pursue and seek additional patent coverage of all our product candidates.
| 101 |
| Table of Contents |
DMT310
Our DMT310 portfolio includes two families, one in-licensed and one owned by Dermata. The in-licensed family includes patents and patent applications in-licensed from Villani, Inc. related to therapeutic compositions and methods for treating skin conditions. The in-licensed portfolio consists of one pending non-provisional U.S. patent application, two granted U.S. patents, and granted foreign patents in Australia, Brazil, Canada, France, Germany, Ireland, Italy, Mexico, Russia, Singapore, South Korea, Spain, Switzerland and the United Kingdom. Additionally, the issued foreign patent in Japan, has lapsed and is no longer in force or valid. These patents are expected to expire between 2022 and 2023, absent any patent term adjustments or extensions. Based on the anticipated timing of any potential FDA approval of DMT310 for acne, the patents that expire in 2022 and 2023 are not material to our business, as we do not expect these patents to provide any protection for our product candidates. We expect our intellectual property portfolio to be protected by any potential NCE exclusivity for DMT310 and our other product candidates, the maintaining of our exclusive supply agreement for our raw material requirements, and our continued efforts to protect our proprietary information. We also have an additional Dermata owned family related to DMT310, with applications pending in the U.S., Australia, and Canada. This family refers to specific attributes of the DMT310 API and drug product as well as treatment related attributes for the treatment of acne based on the data received prior to its filing. Patents in this patent family, if granted, are expected to expire in 2039, absent any patent term adjustments or extensions.
DMT410
Our DMT410 portfolio includes two families owned by Dermata. The first family consists of one pending non-provisional U.S. patent application and six pending foreign patent applications in Australia, Canada, China, the European Patent Office, Japan, and South Korea. These patent applications relate to compositions for the treatment of skin diseases using our proprietary sponge powder in combination with multiple types of botulinum toxin for both medical and aesthetic skin conditions and diseases. Patents in this patent family, if granted, are expected to expire in 2039, absent any patent term adjustments or extensions. The second family is related to certain of our clinical methods related to sponge powder and botulinum toxin. This second family consists of a US non-provisional application and a PCT application. Patents in this patent family, if granted are expected to expire in 2041, absent any patent term adjustments or extensions.
DMT400
Our DMT400 portfolio includes two families owned by Dermata. The first family consists of pending applications in the U.S., Canada, and Japan covering our sponge powder in combination with many approved and development stage monoclonal antibodies for the treatment of skin diseases. Patents in this patent family, if granted, are expected to expire in 2039, absent any patent term adjustments or extensions. The second family consists of a pending applications in the U.S., Australia, Japan, and South Korea, covering compositions for the treatment of conditions by dermal fillers in combination with our proprietary sponge powder. Patents in this patent family, if granted, are expected to expire in 2040, absent any patent term adjustments or extensions.
Although we believe our patent portfolio offers significant protection for DMT310, DMT410 and DMT400 and additional combination regimens, the protection offered by our patents may be, to some extent, more limited than the protection provided by patents which claim chemical structures which were previously unknown. Accordingly, other parties may compete with us, for example, by independently developing or obtaining competing topical formulations that design around our patent claims, but which may contain the same or similar active ingredients, or by seeking to invalidate our patents.
| 102 |
| Table of Contents |
The term of individual patents depends upon the laws of the countries in which they are obtained. In most countries in which we file, the patent term is 20 years from the earliest priority date of filing of a non-provisional patent application. However, the term of United States patents may be extended for delays incurred due to compliance with the FDA, requirements or by delays encountered during prosecution that are caused by the United States Patent and Trademark Office, or the USPTO. For example, the Hatch-Waxman Act permits a patent term extension for FDA-approved drugs of up to five years beyond the expiration of the patent. The length of the patent term extension is related to the length of time the drug is under regulatory review. Patent extension cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval, and only one patent applicable to an approved drug may be extended. Similar provisions are available in Europe and other jurisdictions to extend the term of a patent that covers an approved drug. In the future, if and when our product candidates receive FDA approval, we expect to apply for patent term extensions on patents covering those product candidates. We intend to seek patent term extensions in any jurisdiction where these are available and where we also have a patent that may be eligible; however there is no guarantee that the applicable authorities, including the United State Patent and Trademark Office and United States FDA, will agree with our assessment of whether such extensions should be granted, and even if granted, the length of such extensions.
Other Intellectual Property
In addition to patent protection, we also rely heavily on trade secrets, including unpatented know-how, technology innovation, technical specifications and assays and other proprietary information in attempting to develop and maintain our competitive advantage. We believe our ability to protect our unpatented know-how and trade secrets are as important if not more important than our patent portfolio due the complex nature and lack of expiration associated with such information.
We seek trademark protection in the United States and in certain other jurisdictions where available and when we deem appropriate. We currently have registrations for Dermata in the United States and multiple other jurisdictions. We intend to file applications for trademark registrations in connection with our therapeutic candidates in various jurisdictions, including the United States.
Material Agreements
License Agreement between Dermata Therapeutics, LLC and Villani, Inc.
On March 31, 2017, we entered into a License Agreement (or, the License Agreement) with Villani, Inc. (or, Villani), whereby Villani has granted us an exclusive, sub-licensable, royalty-bearing license (or, the License) under the Licensed Patents (as defined in the License Agreement), to formulate, develop, seek regulatory approval for, make or sell products that contain Spongilla lacustris (alone or in combination with other active or inactive ingredients) for the treatment of diseases, disorders and conditions of the skin, including but not limited to acne, rosacea, psoriasis, atopic dermatitis, seborrheic dermatitis, actinic keratosis and eczema that were developed using certain licensed know-how (or, the Licensed Products). We are responsible for the development (including manufacturing, packaging, non-clinical studies, clinical trials and obtaining regulatory approval) and commercialization (including marketing, promotion, distribution, etc.) for all Licensed Products.
In partial consideration of the License, we forgave our outstanding loan to Villani in the amount of $400,000, and agreed to make future milestone payments to Villani. The milestone payments due to Villani under the License Agreement have been amended to the amounts described below in connection with our entry into the Second License Amendment (as defined below).
The License Agreement may be terminated (i) by either party for material breach with 90 days written notice, or 30 days’ notice if for material payment breach, if such material breach is not cured within such notice period, (ii) immediately upon written notice to either party if either party initiates a voluntary bankruptcy proceeding, dissolves or winds-up its business, (iii) immediately upon written notice to either party if either party becomes subject to involuntary bankruptcy proceedings, if such proceedings are not dismissed or stayed within 90 days.
The License Agreement includes customary terms relating to, among others, indemnification, intellectual property protection, confidentiality, remedies and warranties.
| 103 |
| Table of Contents |
On June 4, 2019, we entered into a License Amendment and Settlement Agreement (or the First License Amendment) with Villani. Pursuant to the First License Amendment, we made milestone payments to Villani in an aggregate amount of $750,000, and in exchange for certain know-how, we issued to Villani 5,221,156 units of our Series 1c Preferred Units (which units were converted into shares of our Series 1c Preferred Stock in connection with our conversion into a Delaware corporation, which was equal to 5% of our total capitalization (on a fully-diluted basis) at the time of issuance). At the time of issuance, these units were valued at $730,962. We subsequently cancelled the shares of Series 1c Preferred Stock issued to Villani pursuant to the Second License Amendment, as described below. Pursuant to the First License Amendment, we also agreed to make certain milestone payments to Villani, which rates were subsequently amended pursuant to the Second License Amendment, as discussed below. To date, the $750,000 milestone payment made in connection with the First License Amendment is the total amount paid to Villani in connection with the License.
On July 30, 2021, we entered into a Second Amendment to the License and Settlement Agreement (or, the Second License Amendment), whereby, for the settlement of certain disputes arising under the First License Amendment, we agreed to exchange the shares of Series 1c Preferred Stock owned by Villani for an increase of milestone payments and royalty rates due to Villani under the License Agreement. The resulting royalty rates payable pursuant to the Second License Amendment are equal to single-digit percentages of net sales of Licensed Products and HMW Combination Products (as defined in the License Agreement), subject to certain adjustments as set forth in the Second License Amendment. Royalties are payable on a country-by-country and Licensed Product-by-Licensed Product basis, for the period of time from the effective date of the License Agreement until the later of (i) the expiration of the last to expire valid claim in such country (which is set to expire in 2023), (ii) the expiration of regulatory exclusivity for such Licensed Product in such country, and (iii) 15 years from the date of the first commercial sale of the Licensed Product in such country. Pursuant to the Second License Amendment, if we sublicense the License, we are obligated to pay to Villani a sublicense fee of between 10% and 30% of Sublicense Revenues (as defined in the License Agreement). Such future milestone payments due to Villani (all payable to Villani in cash or in equity, at the option of Villani) are in aggregate amounts of up to $3.5 million in development milestones and $37.0 million in sales milestones. We paid to Villani $1.0 million upon the closing of our initial public offering.
Supply Agreement between Dermata Therapeutics LLC and Reka-Farm LLC
On February 27, 2020, we entered into an exclusive Supply Agreement (or, the Supply Agreement) with Reka-Farm, LLC (or, Reka-Farm), whereby Reka-Farm will supply us with the Spongilla raw materials necessary for use in the development of our product candidates. The Supply Agreement has an indefinite term unless and until terminated. For the term of the Supply Agreement, Reka-Farm is prohibited from supplying Spongilla for development and sale of any other product outside of the Russian Federation, other than Cosmetic Products (as defined in the Supply Agreement).
Pursuant to the Supply Agreement, we shall provide Reka-Farm with two-year rolling forecasts of our Spongilla raw material requirements, and such forecasts shall be provided to Reka-Farm on a semi-annual basis, beginning on January 1, 2021 (each, a Forecast). Pursuant to the Supply Agreement, Reka-Farm has guaranteed its ability to supply us with the required amounts of Spongilla as specified in each Forecast for the first 12 months of each Forecast. All Forecasts are non-binding on us. If Reka-Farm is unable to supply us with Spongilla raw material in accordance with a Forecast, all available quantities of Spongilla then available to Reka-Farm shall be made available to us on a first priority basis until all amounts of Spongilla set forth in the Forecast are supplied.
Pursuant to the Supply Agreement, we pay a pre-negotiated price per kilogram for Spongilla supplied by Reka-Farm, and we are required to pay to Reka-Farm a royalty payments of less than one percent of the Net Sales (as defined in the Supply Agreement) of any products we develop containing Spongilla raw material supplied by Reka-Farm.
The Supply Agreement may be terminated (i) by either party for material breach with 90 days written notice, if such material breach is not cured within such notice period and (ii) by us for any reason or no reason upon 90 days written notice to Reka-Farm.
The Supply Agreement includes customary terms relating to, among others, indemnification, intellectual property protection, confidentiality, remedies, warranties, as well as certain quality requirements.
| 104 |
| Table of Contents |
Employees
As of the date of this prospectus, we have eight full time employees, with three employees working in the general and administrative department, two engaged in non-clinical and clinical development, two working in the chemistry, manufacturing and controls department, and one employee working in the regulatory affairs and quality control department.
Legal Proceedings
We are not currently subject to any material legal proceedings. However, we may from time to time become a party to various legal proceedings arising in the ordinary course of our business.
MANAGEMENT
Executive Officers and Directors
The following table provides information regarding our executive officers, key employees and directors as of the date of this prospectus:
Name |
| Age |
| Position |
Executive Officers |
|
| ||
Gerald T. Proehl |
| 62 |
| President, Chief Executive Officer and Chairman of the Board |
Kyri K. Van Hoose |
| 43 |
| Senior Vice President and Chief Financial Officer |
Christopher J. Nardo, M.P.H., Ph.D. |
| 57 |
| Senior Vice President, Development |
Maria Bedoya Toro Munera, Ph.D. |
| 68 |
| Senior Vice President, Regulatory Affairs & Quality Assurance |
Non-Employee Directors |
|
| ||
David Hale (2)(3) |
| 72 |
| Lead Director |
Wendell Wierenga, Ph.D. (2) |
| 75 |
| Director |
Kathleen Scott (1)(4) |
| 52 |
| Director |
Stephen J. Mento, Ph.D. (3) |
| 69 |
| Director |
Mary Fisher (1) |
| 59 |
| Director |
Andrew Sandler, M.D. (2)(3) |
| 56 |
| Director |
Brittany Bradrick (1) |
| 52 |
| Director |
(1) | Member of the Audit Committee. |
(2) | Member of the Compensation Committee. |
(3) | Member of the Nominating and Governance Committee. |
Management
Gerald T. Proehl became a director and our President and Chief Executive Officer in December 2014 and became our Chairman in April 2021. Mr. Proehl has more than 30 years of experience within the pharmaceutical industry. From January 2002 until January 2014, Mr. Proehl was President and CEO of Santarus, Inc., where he led the sale of Santarus, Inc. to Salix Pharmaceuticals, Inc. for $2.6 billion. Prior to Santarus, Inc., Mr. Proehl worked for Hoechst Marion Roussel, Inc. for 14 years, where he served in various capacities, including VP of Global Marketing. While at Hoechst, he was responsible for marketing products in multiple therapeutic areas, including cardiology, allergy/respiratory, immunology, and neurology. Mr. Proehl holds a B.S. in Education from State University of New York at Cortland, an M.A. in Exercise Physiology from Wake Forest University, and an M.B.A. from Rockhurst University. Mr. Proehl currently serves on the board of one public company, Tenax Therapeutics, Inc. (NYSE: TENX). Mr. Proehl was selected as an officer and director due to his leadership experience at other companies and his history of founding and operating specialty pharmaceutical companies.
| 105 |
| Table of Contents |
Kyri K. Van Hoose became our Senior Vice President and Chief Financial Officer in September 2021. Ms. Van Hoose is a seasoned and collaborative finance and accounting professional with over 20 years of experience, including more than 15 years of accounting and finance experience in the life science industry. Prior to joining Dermata, from September 2020 to April 2021, Ms. Van Hoose served as Chief Financial Officer of TEGA Therapeutics, Inc., a private biotechnology company. Prior to TEGA, from November 2019 to April 2020, Ms. Van Hoose served as the head of finance for Curzion Pharmaceuticals, Inc., a private, rare disease company, until its acquisition by Horizon Therapeutics plc in April 2020. Ms. Van Hoose also served at Avelas Biosciences, Inc., a clinical-stage biotechnology company from December 2017 to July 2019. From September 2005 to February 2016, Ms. Van Hoose held leadership positions of increasing responsibilities at Acadia Pharmaceuticals, Inc., including Senior Director of Finance and Corporate Controller. Ms. Van Hoose began her career at Deloitte and is a licensed Certified Public Accountant (California active). Ms. Van Hoose earned her B.S. in Accounting at the University of Southern California and M.B.A. in Finance at University of California, Irvine.
Christopher J. Nardo, M.P.H., Ph.D. became our Senior Vice President of Development in June 2015. Dr. Nardo has more than 15 years of experience in the pharmaceutical industry. From September 2010 to May 2015, Dr. Nardo served in positions of increasing responsibility at Allergan, PLC, initially leading clinical development of BOTOX for the treatment of Over Active Bladder from Phase 3 to product approval and launch, and ultimately leading clinical development for the aesthetic group within the Dermatology Therapeutic Area, as well as serving as Global Development Team Leader for the Allergan/Medytox collaboration including serving as the Chair of the AGN/MTX Joint Development Committee. From 2006 to 2010, Dr. Nardo served as Vice President, Clinical Operations and Biostatistics at Spectrum Pharmaceuticals, Inc. (NASDAQ: SPPI). Prior to that, Dr. Nardo held various clinical development leadership positions at CancerVax Corporation, The Immune Response Corporation, and Procter and Gamble Company (NYSE: PG). Dr. Nardo obtained a Ph.D. in Epidemiology from the Gillings School of Public Health at the University of North Carolina at Chapel Hill, his M.P.H. in Epidemiology and Biostatistics from the School of Public Health at San Diego State University, and his B.S. in Biology at Loyola Marymount University.
Maria Bedoya Toro Munera, Ph.D. became our Senior Vice President of Regulatory Affairs and Quality Assurance in January 2016. Dr. Bedoya Toro Munera has more than 30 years of experience in regulatory compliance, quality control and quality assurance within the pharmaceutical industry. From 2014 until its sale to Celgene in 2015, Dr. Bedoya Toro Munera served as Senior Vice President, Regulatory Affairs and Quality Assurance at Receptos Inc. Prior to Receptos, Inc., Dr. Bedoya Toro Munera served as Senior Vice President of Regulatory Affairs and Quality Assurance at Santarus, Inc. from June 2007 to January 2014. She previously served as Senior Director Regulatory Affairs at Eisai Medical Research Inc., from November 2006 to May 2007, moving to Eisai from Ligand Pharmaceuticals, Inc. when Ligand divested their oncology products to Eisai in November 2006. Dr. Bedoya Toro Munera worked as Senior Director Global Regulatory Affairs and Compliance at Ligand from 2003 to 2006. From 2000 to 2003, she served as Director Global Regulatory Affairs at Baxter Hyland Immuno. From 1998 to 2000, Dr. Bedoya Toro Munera worked at BASF Bioresearch Corporation as Director, Regulatory Affairs/Quality, and from 1996 to 1998, she worked as Director, Quality Assurance and Regulatory Compliance at Amylin Pharmaceuticals. From 1988 to 1996, Dr. Bedoya Toro Munera worked at Rhone-Poulenc Rorer in a number of increasingly responsible positions in regulatory compliance, quality assurance, quality control and compliance. Dr. Bedoya Toro Munera holds an M.B.A. from the University of Chicago, and a Ph.D. in bio-analytical chemistry from Ohio University. In addition, she has a M.A. in bio-analytical chemistry and a B.S. in chemistry from Western Michigan University.
Non-Employee Directors
David Hale is our co-founder and has served as a member of our board of directors since December 2014, and as Lead Director since April 2021. Mr. Hale is Chairman and CEO of Hale BioPharma Ventures, LLC a private company focused on the formation and development of biotechnology, specialty pharma, diagnostic and medical device companies. Mr. Hale is a serial entrepreneur who has been involved in the formation and development of a number of successful biomedical companies. He served as the Chairman of Santarus, Inc., a specialty biopharmaceutical company, since 2004 and a member of Santarus’ board since 2000, prior to its acquisition by Salix Pharmaceuticals, Ltd. in 2014, and as Chairman of SkinMedica, Inc., prior to its sale to Allergan in 2012, Micromet, Inc., prior to its sale to Amgen Inc. in 2012, Somaxon Pharmaceuticals, Inc., prior to its sale to Pernix Therapeutics Holdings Inc. in 2013, Crisi Medical Systems, Inc., prior to its sale to Becton Dickinson & Company in 2015, and Agility Clinical, Inc. prior to its sale to Precision Medicine Inc. in 2017.
| 106 |
| Table of Contents |
Mr. Hale currently sits on the board of directors of two public companies, including Oncternal Therapeutics, Inc. (NASDAQ: ONCT), since 2019, and BIOCEPT, Inc. (NASDAQ: BIOC), since 2011.
Mr. Hale is a co-founder and Chairman of Adigica Health, Inc., Zerigo Health, Inc., Oncternal Therapeutics, Inc., Neurana Pharmaceuticals, Inc. He is also co-founder and Director of Neurelis Pharmaceuticals, Inc. and Recros Medica, Inc., a co-founder of Cadence Inc., Evoke Pharma Inc., Elevation Pharmaceutical Inc. and Zogenix Inc.
Mr. Hale is a co-founder and serves on the Board of Directors of BIOCOM and CONNECT and is a former member of the Board of the Biotechnology Industry Organization (BIO), and the Biotechnology Institute. He has served on the Board of Rady Children’s Hospital since 1986, including Chairman of the Board from 2011 to 2015, and is founder and Chairman of the Rady Children’s Institute of Genomic Medicine. He is a member of the UCSD Rady School of Management Dean’s Advisory Council and a Director of the San Diego Economic Development Corporation. Mr. Hale was selected as a director due to his industry and executive business experience.
Wendell Wierenga, Ph.D. became a director in September 2016. From June 2011 to January 2014, Dr. Wierenga served as Executive Vice President, Research and Development at Santarus, Inc., a public biopharmaceutical company that was acquired by Salix Pharmaceuticals, Inc. in January 2014. From July 2004 to May 2011, Dr. Wierenga served as Executive Vice President, Research and Development at Ambit Biosciences Corporation and Neurocrine Biosciences, Inc. (NASDAQ: NBIX). Prior to Neurocrine, from August 1999 to June 2004 he served as the Chief Executive Officer for Syrrx, Inc. where he built an early stage biotech company which was acquired by Takeda Pharmaceutical Company Limited in 2005. From 1990 to 2000, Dr. He also was Sr. VP of Research at Parke Davis/Warner Lambert, when it was acquired by Pfizer Inc. and prior to that held various positions in research at Upjohn Pharmaceuticals from 1974-1990. Dr. Wierenga earned his Ph.D. in chemistry from Stanford University and his B.A. from Hope College in Holland, Michigan. He is currently the chair of the Board of Directors of Crinetics (NASDAQ: CRNX) and is also a member of the Board of Directors of Cytokinetics, Inc. (NASDAQ: CYTK). Most recently was on the Board of Directors for Anacor Pharmaceuticals Inc. and XenoPort, Inc. prior to their sales to Pfizer Inc. and Arbor Pharmaceuticals, LLC, respectively. Dr. Wierenga was selected as a director due to his industry and executive business experience.
Kathleen Scott became a director upon the effectiveness of our initial public offering in August 2021. Ms. Scott is currently the Chief Financial Officer of Neurana Pharmaceuticals, Inc., a privately held biotech company. Prior to Neurana, Ms. Scott was the Chief Financial Officer of Recros Medica from August 2014 to April 2021, Adigica Health from February 2016 to March 2021, Clarify Medical from August 2014 to December 2016, Oncternal Therapeutics from March 2016 to May 2016, MDRejuvena from August 2014 to August 2016, and BioSurplus from March 2010 to November 2014. Prior to BioSurplus, Ms. Scott was a Partner at RA Capital Advisors, a San Diego private investment bank providing financial advisory services. Ms. Scott spent over 15 years with RA Capital Advisors, from December 1994 to July 2010, completing billions of dollars of mergers, acquisitions, divestitures, and restructurings for a broad range of corporate clients. Ms. Scott started her career as an auditor in Arthur Andersen’s San Diego office, focusing on both public and private clients. Ms. Scott is immediate past board chair of the YMCA of San Diego County and is a CPA and CFA charter holder. Ms. Scott graduated magna cum laude from UCLA with a B.S. in economics/business. Ms. Scott was selected as a director due to her extensive industry and financial experience.
Steven J. Mento, Ph.D. became a director upon the effectiveness of our initial public offering in August 2021. Since November 2021, Dr. Mento has served as Executive Chairman and Interim President and CEO of Histogen, Inc. Since July 2005, Dr. Mento has served as a director on the board of directors of Conatus Pharmaceuticals, Inc. (now Histogen, Inc.) and from July 2005 to December 2012, Dr. Mento served as chairman of Conatus’ board of directors. Dr. Mento was a co-founder of Conatus and served as its President and Chief Executive Officer from July 2005 until its merger with Histogen Inc. (NASDAQ: HSTO) in May 2020. Dr. Mento has over 35 years of combined experience in the biotechnology and pharmaceutical industries. From 1997 to 2005, Dr. Mento was President, Chief Executive Officer and a member of the board of directors of Idun Pharmaceuticals, Inc. Dr. Mento guided Idun during its transition from a discovery focused organization to a drug development company with multiple products in or near human clinical testing. In April 2005, Idun was sold to Pfizer Inc. Previously, Dr. Mento served as President of Chiron Viagene, Inc. (subsequently Chiron Technologies, Center for Gene Therapy) from 1995 to 1997, and Vice President of Chiron Corporation from 1995 to 1997. Dr. Mento was Vice President of research and development at Viagene from 1992 to 1995. Prior to Viagene, Dr. Mento held various positions at American Cyanamid Company from 1982 to 1992, including as Director of Viral Vaccine Research and Development at Lederle-Praxis Biologicals, a business unit of American Cyanamid. Dr. Mento currently serves on the board of directors of Histogen, and BIOCOM California. He previously served on the boards of Biotechnology Innovation Organization, BIO Emerging Companies Section Governing Board, BIO Health Section Governing Board, and Sangamo Biosciences, Inc. Dr. Mento holds a Ph.D. and M.S., both in Microbiology, from Rutgers University, and a B.A. in Microbiology from Rutgers College. Dr. Mento was selected as a director due to his experience in the biotechnology and pharmaceutical industries, including executive leadership experience at several pharmaceutical companies.
| 107 |
| Table of Contents |
Mary Fisher became a director upon the effectiveness of our initial public offering in August 2021. Ms. Fisher currently serves as Chief Executive Officer, Chair and a Director at Colorescience Inc., a science-based skincare company and former division of SkinMedica, Inc. While at SkinMedica, Ms. Fisher served as Chief Executive Officer from April 2008 to December 2012, where she led the successful sale of the company to Allergan, Inc. for $350 million, with the potential for another $25 million contingent on certain sales milestones. Prior to joining SkinMedica, from June 2000 to July 2007, Ms. Fisher served as the Chief Operating Officer of Acorda Therapeutics, Inc. (NASDAQ: ACOR). She previously held management and leadership positions at Cephalon, Inc. from March 1994 to March 1999, Immunex Corp. from November 1990 to March 1994, and Boehringer Ingelheim from 1981 to 1990. She previously served on the Board of Directors at ZELTIQ Aesthetics, Inc. from September 2012 to April 2017, and Ovascience from June 2013 to August 2018. Ms. Fisher currently sits on the Board of Sientra since January 2019. Ms. Fisher was selected as a director due to her extensive business and professional experience.
Andrew Sandler, M.D. became a director upon the effectiveness of our initial public offering in August 2021. Since September 2017, Dr. Sandler has served as Chief Medical Officer at Kiadis Pharma N.V. Prior to Kiadis, Dr. Sandler was Senior Vice President, Medical Affairs at Medivation (acquired by Pfizer) from January 2016 to June 2017. Dr. Sandler held various additional roles including Chief Medical Officer and Seattle Site Head at Dendreon Pharmaceuticals from October 2010 to April 2015. Prior to Dendreon, Dr. Sandler was Chief Medical Officer at Spectrum Pharmaceuticals from September 2008 to April 2010, and Vice President, Head of Global Medical Affairs, Oncology for Bayer Healthcare Pharmaceuticals from February 2008 to February 2010. Dr. Sandler also held various positions at Berlex Oncology/Schering AG from October 2003 to August 2008, and Seagen, Inc. from October 1999 to June 2003. Dr. Sandler was a Fellow in Hematology/Medical Oncology at the University of California, San Francisco (UCSF) from July 1994 to June 1996. He did his Internship, Residency, and Chief Residency at Mt. Sinai Hospital in New York, NY from July 1990 to June 1994. Dr. Sandler attended and received his MD degree from Mount Sinai School of Medicine (Icahn School of Medicine at Mt. Sinai) from July 1986 to June 1990. In addition, he graduated from the University of Rochester with a B.S. degree in Neuroscience in 1986. Dr. Sandler was selected as a director due to his experience in the biotechnology and pharmaceutical industries as well as his leadership experience.
Brittany Bradrick became a director in January 2022. Ms. Bradrick has served as the Chief Financial Officer of Neurelis, Inc. since October 2021. Prior to joining Neurelis, Ms. Bradrick was Chief Operating Officer and Chief Financial Officer at ViaCyte Inc. from June 2020 to September 2021. Prior to Viacyte, Ms. Bradrick served in strategy and corporate development positions at Insulet Corporation as Vice President, Strategy & Corporate Development from 2016 to 2020 and as Director, Business Development & Alliance Management at Abbot Laboratories (NYSE: ABT). Prior to these positions, Ms. Bradrick was an investment banker for the life science industry at Piper Jaffray, Credit Suisse, and Chase Securities from 1997 to 2007. Ms. Bradrick began her career as a Federal Reserve Bank Examiner. Ms. Bradrick holds an M.B.A. from the Johnson Graduate School of Management at Cornell University and a B.S. in Business Administration from the University of Missouri. Ms. Bradrick was selected as a director due to her extensive industry and financial experience.
Family Relationships
There are no family relationships among any of the directors or executive officers.
Committees of the Board of Directors
Our board of directors has established an Audit Committee, a Compensation Committee, and a Nominating and Corporate Governance Committee. Our board of directors may establish other committees to facilitate the management of our business. The composition and functions of each committee are described below. Members serve on these committees until their resignation or until otherwise determined by our board of directors. Each of these committees operate under a charter that has been approved by our board of directors, which are available on our website.
| 108 |
| Table of Contents |
Audit Committee. Our Audit Committee consists of Kathleen Scott, Mary Fisher, and Brittany Bradrick, with Ms. Scott serving as the Chairwoman of the Audit Committee. Our board of directors has determined that the three directors currently serving on our Audit Committee are independent within the meaning of the Nasdaq Marketplace Rules and Rule 10A-3 under the Exchange Act. In addition, our board of directors has determined that Kathleen Scott qualifies as an audit committee financial expert within the meaning of SEC regulations and The Nasdaq Marketplace Rules.
The Audit Committee oversees and monitors our financial reporting process and internal control system, reviews and evaluates the audit performed by our registered independent public accountants and reports to the board of directors any substantive issues found during the audit. The Audit Committee is directly responsible for the appointment, compensation and oversight of the work of our registered independent public accountants. The Audit Committee reviews and approves all transactions with affiliated parties.
Compensation Committee. Our Compensation Committee consists of Wendell Wierenga, Ph.D., David Hale and Andrew Sandler, with Dr. Wierenga serving as the Chairman of the Compensation Committee. Our board of directors has determined that the three directors currently serving on our Compensation Committee are independent under the listing standards, are “non-employee directors” as defined in rule 16b-3 promulgated under the Exchange Act and are “outside directors” as that term is defined in Section 162(m) of the Internal Revenue Code of 1986, as amended.
The Compensation Committee provides advice and makes recommendations to the board of directors in the areas of employee salaries, benefit programs and director compensation. The Compensation Committee also reviews and approves corporate goals and objectives relevant to the compensation of our President, Chief Executive Officer, and other officers and makes recommendations in that regard to the board of directors as a whole.
Nominating and Corporate Governance Committee. Our Nominating and Corporate Governance Committee consists of David Hale, Steven Mento and Andrew Sandler, M.D., with Mr. Hale serving as the Chairman of the Nominating and Corporate Governance Committee. The Nominating and Corporate Governance Committee nominates individuals to be elected to the board of directors by our stockholders. The Nominating and Corporate Governance Committee considers recommendations from stockholders if submitted in a timely manner in accordance with the procedures set forth in our bylaws and will apply the same criteria to all persons being considered. All members of the Nominating and Corporate Governance Committee are independent directors as defined under the Nasdaq listing standards.
Board Composition
Our board of directors consists of seven members. Our directors will hold office until their successors have been elected and qualified or until the earlier of their resignation or removal.
In accordance with the terms of our restated certificate of incorporation and bylaws, our board of directors is divided into three classes, class I, class II and class III, with each class serving staggered three-year terms. Upon the expiration of the term of a class of directors, directors in that class will be eligible to be elected for a new three-year term at the annual meeting of stockholders in the year in which their term expires. Our directors are divided among the three classes as follows:
| • |
| The Class I directors are Andrew Sandler, M.D. and Mary Fisher; their terms will expire at the 2022 Annual Meeting. |
| • |
| The Class II directors are David Hale, Steven J. Mento and Brittany Bradrick; their terms will expire at the 2023 Annual Meeting. |
| • |
| The Class III directors are Gerald T. Proehl, Wendell Wierenga, Ph.D. and Kathleen Scott; their terms will expire at the 2024 Annual Meeting. |
| 109 |
| Table of Contents |
We expect that any additional directorships resulting from an increase in the number of directors will be distributed among the three classes so that, as nearly as possible, each class will consist of one-third of the directors. The division of our board of directors into three classes with staggered three-year terms may delay or prevent a change of our management or a change in control.
Our amended and restated certificate of incorporation and amended and restated bylaws provide that the authorized number of directors may be changed only by resolution of our board of directors. Our amended and restated certificate of incorporation and amended and restated bylaws also provide that our directors may be removed only for cause, and that any vacancy on our board of directors, including a vacancy resulting from an enlargement of our board of directors, may be filled only by vote of a majority of our directors then in office.
We have no formal policy regarding board diversity. Our priority in selection of board members is identification of members who will further the interests of our stockholders through his or her established record of professional accomplishment, the ability to contribute positively to the collaborative culture among board members, knowledge of our business and understanding of the competitive landscape.
Director Independence
Our common stock is listed on the Nasdaq Capital Market (or Nasdaq). Under the listing requirements and rules of Nasdaq, independent directors must constitute a majority of a listed company’s board of directors. Under the rules of Nasdaq, a director will only qualify as an “independent director” if, in the opinion of that company’s board of directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. In order to be considered independent for purposes of Rule 10A-3 of the Exchange Act, a member of an audit committee of a listed company may not, other than in his or her capacity as a member of the audit committee, the board of directors, or any other board committee, accept, directly or indirectly, any consulting, advisory, or other compensatory fee from the listed company or any of its subsidiaries or otherwise be an affiliated person of the listed company or any of its subsidiaries.
Our board of directors has determined that David Hale, Wendell Wierenga, Ph.D., Andrew Sandler, M.D., Mary Fisher, Steven J. Mento Ph.D., Brittany Bradrick and Kathleen Scott are “independent directors” as such term is defined by Nasdaq Marketplace Rule 5605(a)(2). We have established an Audit Committee, a Compensation Committee and a Nominating and Corporate Governance Committee. Our board of directors has determined that Kathleen Scott is an “audit committee financial expert,” as defined under the applicable rules of the SEC, and that all members of the Audit Committee are “independent” within the meaning of the applicable Nasdaq listing standards and the independence standards of Rule 10A-3 of the Securities Exchange Act of 1934. Each of the members of the Audit Committee meets the requirements for financial literacy under the applicable rules and regulations of the SEC and the Nasdaq Stock Market.
Role of Board in Risk Oversight Process
The chief executive officer and Chairman positions are held by Gerald T. Proehl. Mr. Proehl currently beneficially owns approximately 41% of the voting power of our common stock (including shares beneficially owned by Proehl Investment Ventures LLC), or 28% after the closing of this offering. Periodically, our Board of directors assesses these roles and the board of directors leadership structure to ensure the interests of Dermata and our stockholders are best served. Our board of directors has determined that its current leadership structure is appropriate. Gerald T. Proehl, as one of our founders and as our chief executive officer and Chairman, has extensive knowledge of all aspects of Dermata, our business and risks.
While management is responsible for assessing and managing risks to Dermata, our board of directors is responsible for overseeing management’s efforts to assess and manage risk. This oversight is conducted primarily by our full board of directors, which has responsibility for general oversight of risks, and standing committees of our board of directors. Our board of directors satisfies this responsibility through full reports by each committee chair regarding the committee’s considerations and actions, as well as through regular reports directly from officers responsible for oversight of particular risks within our company. Our board of directors believes that full and open communication between management and the board of directors is essential for effective risk management and oversight.
| 110 |
| Table of Contents |
Compensation Committee Interlocks and Insider Participation
None of our executive officers serves, or in the past has served, as a member of the board of directors or compensation committee, or other committee serving an equivalent function, of any entity that has one or more executive officers who serve as members of our board of directors or our compensation committee. None of the members of our compensation committee is, or has ever been, an officer or employee of our company.
Code of Business Conduct and Ethics
We have adopted a written code of business conduct and ethics that applies to our employees, officers and directors. A current copy of the code is posted on the Corporate Governance section of our website, which is located at www.dermatarx.com. We intend to disclose future amendments to certain provisions of our code of business conduct and ethics, or waivers of such provisions applicable to any principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions, and our directors, on our website identified above or in filings with the SEC.
Limitation of Directors Liability and Indemnification
The Delaware General Corporation Law authorizes corporations to limit or eliminate, subject to certain conditions, the personal liability of directors to corporations and their stockholders for monetary damages for breach of their fiduciary duties. Our amended and restated certificate of incorporation limits the liability of our directors to the fullest extent permitted by Delaware law. In addition, we have entered into indemnification agreements with all of our directors and named executive officers whereby we have agreed to indemnify those directors and officers to the fullest extent permitted by law, including indemnification against expenses and liabilities incurred in legal proceedings to which the director or officer was, or is threatened to be made, a party by reason of the fact that such director or officer is or was a director, officer, employee or agent of ours, provided that such director or officer acted in good faith and in a manner that the director or officer reasonably believed to be in, or not opposed to, our best interests.
We have director and officer liability insurance to cover liabilities our directors and officers may incur in connection with their services to us, including matters arising under the Securities Act. Our certificate of incorporation and bylaws also provide that we will indemnify our directors and officers who, by reason of the fact that he or she is or was one of our officers or directors of our Company, is involved in any action, suit or proceeding, whether civil, criminal, administrative or investigative related to their board role with us.
There is no pending litigation or proceeding involving any of our directors, officers, employees or agents in which indemnification will be required or permitted. We are not aware of any threatened litigation or proceeding that may result in a claim for such indemnification.
Indemnification Agreements
We have entered into Indemnification Agreements with each of our current directors and executive officers. The Indemnification Agreements provide for indemnification against expenses, judgments, fines and penalties actually and reasonably incurred by an indemnitee in connection with threatened, pending or completed actions, suits or other proceedings, subject to certain limitations. The Indemnification Agreements also provide for the advancement of expenses in connection with a proceeding prior to a final, non-appealable judgment or other adjudication, provided that the indemnitee provides an undertaking to repay to us any amounts advanced if the indemnitee is ultimately found not to be entitled to indemnification by us. The Indemnification Agreements set forth procedures for making and responding to a request for indemnification or advancement of expenses, as well as dispute resolution procedures that will apply to any dispute between us and an indemnitee arising under the Indemnification Agreements.
| 111 |
| Table of Contents |
EXECUTIVE COMPENSATION
The following tables and accompanying narrative disclosure set forth information about the compensation earned by our named executive officers during the year ended December 31, 2021. Our named executive officers, who are our principal executive officer and the two most highly compensated executive officers (other than our principal executive officer) serving as executive officers as of December 31, 2021, were:
| • |
| Gerald T. Proehl, our President and Chief Executive Officer; |
|
|
|
|
| • |
| Kyri K. Van Hoose, our Senior Vice President and Chief Financial Officer; and |
| • |
| Christopher J. Nardo, Ph.D., our Senior Vice President, Development. |
Summary Compensation Table
The following table sets forth information concerning the compensation of our Named Executive Officers for the year ended December 31, 2021 and 2020:
Name and Principal Position |
| Year |
| Salary |
|
| Bonus (1) |
|
| Stock Option Awards (2) |
|
| Total |
| ||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
Gerald T. Proehl |
| 2021 |
| $ | 135,907 |
|
| $ | - |
|
| $ | 1,162,365 |
|
| $ | 1,298,272 |
|
President and Chief Executive Officer |
| 2020 |
|
| 27,040 |
|
|
| - |
|
|
| - |
|
|
| 27,040 |
|
Kyri K. Van Hoose |
| 2021 |
|
| 78,750 |
|
|
| 28,800 |
|
|
| 354,900 |
|
|
| 462,450 |
|
Senior Vice President, Chief Financial Officer |
| (3) |
|
| - |
|
|
| - |
|
|
| - |
|
|
| - |
|
Christopher J. Nardo, M.P.H, Ph.D. |
| 2021 |
|
| 270,000 |
|
|
| 86,400 |
|
|
| 322,504 |
|
|
| 678,904 |
|
Senior Vice President, Development |
| 2020 |
|
| 270,000 |
|
|
| - |
|
|
| - |
|
|
| 270,000 |
|
(1) | Mr. Proehl’s bonus for 2021 was remunerated in fully vested stock options. All stock options are granted pursuant to our 2021 Plan. Ms. Van Hoose and Dr. Nardo’s bonuses for 2021 were paid in cash. No bonuses were paid for 2020. |
(2) | In accordance with SEC rules, this column reflects the aggregate grant date fair value of the stock option awards granted during 2021. These amounts have been computed in accordance with FASB ASC Topic 718. Assumptions used in the calculation of this amount are described in the notes to our financial statements included elsewhere in this document. This amount does not reflect the actual economic value that will be realized by the executives upon the vesting of the stock options, the exercise of the stock options, or the sale of the common stock underlying such stock options. |
(3) | Ms. Van Hoose joined Dermata in September 2021. |
Employment Arrangements with our Named Executive Officers
We are party to employment agreements with each of our Named Executive Officers listed below. Each of these Named Executive Officers are currently party to customary confidentiality and intellectual property assignment agreements with us.
Gerald T. Proehl
On December 6, 2021, we entered into an employment agreement with Mr. Proehl (the Proehl Agreement). Under the terms of the Proehl Agreement, Mr. Proehl holds the position of President and Chief Executive Officer and receives a base salary of $350,000 annually. In addition, Mr. Proehl is eligible to receive an annual bonus, with a target amount equal to 50% of Mr. Proehl’s base salary. The actual amount of each annual bonus will be based upon the level of achievement of certain of our corporate objectives and Mr. Proehl’s individual objectives, in each case, as established by the Company and Mr. Proehl for the calendar year with respect to which the annual bonus relates. The determination of the level of achievement of the corporate objectives and Mr. Proehl’s individual performance objectives for a year shall be made by us in our reasonable discretion. In addition, Mr. Proehl is eligible to receive, from time to time, equity awards under our existing equity incentive plan, or any other equity incentive plan we may adopt in the future, and the terms and conditions of such awards, if any, will be determined our board of directors or the Compensation Committee, in their discretion. Mr. Proehl will also be eligible to participate in any executive benefit plan or program we adopt.
We may terminate Mr. Proehl’s employment at any time without Cause (as that term is defined in the Proehl Agreement) upon four weeks prior written notice to Mr. Proehl. Mr. Proehl may terminate his employment for Good Reason (as that term is defined in the Proehl Agreement) upon 60 days written notice.
| 112 |
| Table of Contents |
If Mr. Proehl’s employment is terminated without Cause or for Good Reason, Mr. Proehl will be entitled to receive (i) his earned but unpaid base salary through the final day of his employment, (ii) expenses reimbursable under the Proehl Agreement incurred on or prior to the last day of his employment, (iii) any amounts or benefits that are vested amounts or benefits that Mr. Proehl is entitled to receive under any of our equity compensation plans (clauses (i) through (iii) collectively, the Accrued Obligations), (iv) severance payments equal to 12 months of Mr. Proehl’s base salary (to be paid in a lump sum on the next regular payroll date within 60 days of Mr. Proehl’s termination), (v) a pro-rated payment equal to the annual bonus the Board determines is due, and (vi) if elected, we will reimburse Mr. Proehl for certain COBRA health benefits for 12 months.
Notwithstanding the above, if Mr. Proehl’s employment is terminated without Cause or he resigns for Good Reason either within three months immediately preceding or within one year after a Change of Control (as defined in the 2021 Plan), Mr. Proehl will receive (i) the Accrued Obligations, (ii) severance payments equal to 18 months of Mr. Proehl’s base salary (to be paid in a lump sum on the next regular payroll date within 60 days of Mr. Proehl’s termination), (iii) the targeted annual bonus amount the Board determines is due to Mr. Proehl, (iv) if elected, we will reimburse Mr. Proehl for certain COBRA health benefits for 18 months, and (v) Mr. Proehl will be deemed to be fully vested in all of his outstanding equity awards as of the date of his termination.
If Mr. Proehl’s employment is terminated with Cause or without Good Reason, he will be entitled to receive (i) his earned but unpaid base salary through the final day of his employment, (ii) expenses reimbursable under the employment agreement incurred on or prior to the last day of his employment, and (iii) any amounts or benefits that are vested amounts or benefits that Mr. Proehl is entitled to receive under any of our equity compensation plans.
We may terminate Mr. Proehl’s employment at any time for Cause upon written notice to Mr. Proehl. Mr. Proehl may voluntarily terminate his employment at any time without Good Reason upon four weeks prior written notice.
Kyri K. Van Hoose
On November 19, 2021, we entered into an employment agreement with Ms. Van Hoose, which was subsequently amended on January 1, 2022 (as amended, the Van Hoose Agreement). Under the terms of the Van Hoose Agreement, she holds the position of Senior Vice President and Chief Financial Officer and receives a base salary of $300,000 annually. In addition, Ms. Van Hoose is eligible to receive an annual bonus, with a target amount equal to forty percent (40%) of Ms. Van Hoose’s base salary. The actual amount of each annual bonus will be based upon the level of achievement of our corporate objectives and Ms. Van Hoose’s individual objectives, in each case, as established by us and Ms. Van Hoose for the calendar year with respect to which the annual bonus relates. The determination of the level of achievement of the corporate objectives and the Ms. Van Hoose’s individual performance objectives for a year shall be made by us in our reasonable discretion. In addition, pursuant to the terms of her employment agreement, Ms. Van Hoose is eligible to receive, from time to time, equity awards under our existing equity incentive plan, or any other equity incentive plan we may adopt in the future, and the terms and conditions of such awards, if any, will be determined by our board of directors or Compensation Committee, in their discretion. Ms. Van Hoose is also eligible to participate in any executive benefit plan or program we adopt.
We may terminate Ms. Van Hoose’s employment at any time without Cause (as that term is defined in Ms. Van Hoose’s employment agreement) upon two weeks prior written notice to Ms. Van Hoose. Ms. Van Hoose may terminate her employment for Good Reason (as that term is defined in Ms. Van Hoose’s employment agreement) upon 60 days written notice to us, upon which notice we have 30 days to cure the conditions that Ms. Van Hoose considers to be Good Reason, subject to certain conditions set forth in her employment agreement.
If Ms. Van Hoose’s employment is terminated without Cause or for Good Reason, Ms. Van Hoose will be entitled to receive (i) the Accrued Obligations, (iv) severance payments equal to nine months of Ms. Van Hoose’s base salary (to be paid in a lump sum on the next regular payroll date within 60 days of Ms. Van Hoose’s termination), (v) the targeted annual bonus amount the Board determines is due to Ms. Van Hoose, and (vi) if elected, the Company will reimburse Ms. Van Hoose for certain COBRA health benefits for nine months.
Notwithstanding the above, if Ms. Van Hoose’s employment is terminated without Cause or she resigns for Good Reason either within three months immediately preceding or within one year after a Change of Control (as defined in the 2021 Plan), Ms. Van Hoose will receive (i) the Accrued Obligations, (ii) severance payments equal to 12 months of Ms. Van Hoose’s base salary (to be paid in a lump sum on the next regular payroll date within 60 days of Ms. Van Hoose’s termination), (iii) the targeted annual bonus amount the Board determines is due to Ms. Van Hoose, (iv) if elected, the Company will reimburse Ms. Van Hoose for certain COBRA health benefits for 12 months, and (v) Ms. Van Hoose will be deemed to be fully vested in all of her outstanding equity awards as of the date of her termination.
| 113 |
| Table of Contents |
If Ms. Van Hoose’s employment is terminated with Cause or without Good Reason, she is entitled to receive (i) her earned but unpaid base salary through the final day of her employment, (ii) expenses reimbursable under the employment agreement incurred on or prior to the last day of her employment, and (iii) any amounts or benefits that are vested amounts or benefits that Ms. Van Hoose is entitled to receive under any of our equity compensation plans.
We may terminate Ms. Van Hoose’s employment at any time for Cause upon written notice to Ms. Van Hoose. Ms. Van Hoose may voluntarily terminate her employment at any time without Good Reason upon two weeks prior written notice to us.
Christopher J. Nardo, M.P.H., Ph.D.
On August 17, 2021, we entered into an employment agreement with Dr. Nardo, which was subsequently amended on December 6, 2021 and January 1, 2022 (as amended, the Nardo Agreement). Under the terms of the Nardo Agreement, he holds the position of Senior Vice President, Development and receives a base salary of $280,000 annually. In addition, Dr. Nardo is eligible to receive an annual bonus, with a target amount equal to forty percent (40%) of Dr. Nardo’s base salary. The actual amount of each annual bonus will be based upon the level of achievement of our corporate objectives and Dr. Nardo’s individual objectives, in each case, as established by us and Dr. Nardo for the calendar year with respect to which the annual bonus relates. The determination of the level of achievement of the corporate objectives and the Dr. Nardo’s individual performance objectives for a year shall be made by us in our reasonable discretion. In addition, pursuant to the terms of his employment agreement, Dr. Nardo is eligible to receive, from time to time, equity awards under our existing equity incentive plan, or any other equity incentive plan we may adopt in the future, and the terms and conditions of such awards, if any, will be determined by our board of directors or Compensation Committee, in their discretion. Dr. Nardo is also eligible to participate in any executive benefit plan or program we adopt.
We may terminate Dr. Nardo’s employment at any time without Cause (as that term is defined in Dr. Nardo’s employment agreement) upon two weeks prior written notice to Dr. Nardo. Dr. Nardo may terminate his employment for Good Reason (as that term is defined in Dr. Nardo’s employment agreement) upon 60 days written notice to us, upon which notice we have 30 days to cure the conditions that Dr. Nardo considers to be Good Reason, subject to certain conditions set forth in his employment agreement.
If Dr. Nardo’s employment is terminated without Cause or for Good Reason, Dr. Nardo will be entitled to receive (i) the Accrued Obligations, (iv) severance payments equal to nine months of Dr. Nardo’s base salary (to be paid in a lump sum on the next regular payroll date within 60 days of Dr. Nardo’s termination), (v) the targeted annual bonus amount the Board determines is due to Dr. Nardo, and (vi) if elected, the Company will reimburse Dr. Nardo for certain COBRA health benefits for nine months.
Notwithstanding the above, if Dr. Nardo’s employment is terminated without Cause or he resigns for Good Reason either within three months immediately preceding or within one year after a Change of Control (as defined in the 2021 Plan), Dr. Nardo will receive (i) the Accrued Obligations, (ii) severance payments equal to 12 months of Dr. Nardo’s base salary (to be paid in a lump sum on the next regular payroll date within 60 days of Dr. Nardo’s termination), (iii) the targeted annual bonus amount the Board determines is due to Dr. Nardo, (iv) if elected, the Company will reimburse Dr. Nardo for certain COBRA health benefits for 12 months, and (v) Dr. Nardo will be deemed to be fully vested in all of his outstanding equity awards as of the date of his termination.
If Dr. Nardo’s employment is terminated with Cause or without Good Reason, he is entitled to receive (i) his earned but unpaid base salary through the final day of his employment, (ii) expenses reimbursable under the employment agreement incurred on or prior to the last day of his employment, and (iii) any amounts or benefits that are vested amounts or benefits that Dr. Nardo is entitled to receive under any of our equity compensation plans.
We may terminate Dr. Nardo’s employment at any time for Cause upon written notice to Dr. Nardo. Dr. Nardo may voluntarily terminate his employment at any time without Good Reason upon two weeks prior written notice to us.
| 114 |
| Table of Contents |
Outstanding Equity Awards at Fiscal Year-End
The following table summarizes, for each of our Named Executive Officers, the number of shares of our Common Stock underlying outstanding stock options held as of December 31, 2021:
|
| Number of Securities Underlying Unexercised Options |
|
| Option Exercise |
|
| Option Expiration |
| Vesting |
| |||||||
Name |
| Exercisable |
|
| Unexercisable |
|
| Price |
|
| Date |
| Schedule |
| ||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
Gerald T. Proehl |
|
| 3,434 |
|
|
| 7,493 |
|
| $ | 6.31 |
|
| 03/31/26 |
|
| (1) | |
|
|
| 11,178 |
|
|
| 37,602 |
|
|
| 6.31 |
|
| 03/31/26 |
|
| (2) | |
|
|
| 9,552 |
|
|
| 204 |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (3) | |
|
|
| 7,317 |
|
|
| 2,439 |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (4) | |
|
|
| 18,926 |
|
|
| 18,926 |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (5) | |
|
|
| 49,462 |
|
|
| - |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (6) | |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Kyri K. Van Hoose |
|
| 6,250 |
|
|
| 68,750 |
|
|
| 5.39 |
|
| 08/31/31 |
|
| (7) | |
Christopher J. Nardo, M.P.H., Ph.D. |
|
| 2,439 |
|
|
| - |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (8) | |
|
|
| 5,969 |
|
|
| 128 |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (3) | |
|
|
| 4,572 |
|
|
| 1,525 |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (4) | |
|
|
| 12,195 |
|
|
| 12,195 |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (5) | |
|
|
| 1,585 |
|
|
| - |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (6) | |
|
|
| 5,589 |
|
|
| 18,801 |
|
|
| 5.74 |
|
| 03/31/31 |
|
| (2) | |
_____________
(1) | This stock option award was granted March 31, 2021. The stock option vests in 35 equal monthly installments commencing January 11, 2021. |
(2) | This stock option award was granted March 31, 2021. The stock option vests in 48 equal monthly installments commencing January 1, 2021. |
(3) | This stock option award was granted March 31, 2021. The stock option vests in 48 equal monthly installments commencing January 1, 2018. |
(4) | This stock option award was granted March 31, 2021. The stock option vests in 48 equal monthly installments commencing December 19, 2018. |
(5) | This stock option award was granted March 31, 2021. The stock option vests in 48 equal monthly installments commencing December 11, 2019 |
(6) | This stock option award was granted March 31, 2021 and was fully vested at issuance. |
(7) | This stock option award was granted September 1, 2021. The stock option vests in 48 equal monthly installments commencing September 1, 2021. |
(8) | This stock option award was granted on March 31, 2021 and is fully vested. The stock option vested in 48 equal monthly installments commencing September 1, 2017. |
We did not engage in any repricings or other modifications or cancellations to any of our named executive officers' outstanding option awards during the year ended December 31, 2021.
Employee benefits plans
We currently provide broad-based health and welfare benefits that are available to all of our employees, including our Named Executive Officers, including medical, dental, and vision insurance.
401(k) Plan
We sponsor a 401(k) savings plan (or, the 401(k) Plan) for all eligible employees. Under the 401(k) Plan, we may make discretionary matching contributions to the 401(k) Plan to be allocated to employee accounts based upon employee deferrals and compensation. To date, we have not made any matching contributions into the 401(k) Plan.
Limitation of Directors Liability and Indemnification
The Delaware General Corporation Law authorizes corporations to limit or eliminate, subject to certain conditions, the personal liability of directors to corporations and their stockholder for monetary damages for breach of their fiduciary duties. Our amended and restated certificate of incorporation limits the liability of our directors to the fullest extent permitted by Delaware law. In addition, we have entered into indemnification agreements with all of our directors and named executive officers whereby we have agreed to indemnify those directors and officers to the fullest extent permitted by law, including indemnification against expenses and liabilities incurred in legal proceedings to which the director or officer was, or is threatened to be made, a party by reason of the fact that such director or officer is or was a director, officer, employee or agent of ours, provided that such director or officer acted in good faith and in a manner that the director or officer reasonably believed to be in, or not opposed to, our best interests.
| 115 |
| Table of Contents |
We have director and officer liability insurance to cover liabilities our directors and officers may incur in connection with their services to us, including matters arising under the Securities Act. Our amended and restated certificate of incorporation and amended and restated bylaws also provide that we will indemnify our directors and officers who, by reason of the fact that he or she is or was one of our officers or directors of our Company, is involved in any action, suit or proceeding, whether civil, criminal, administrative or investigative related to their board role with us.
There is no pending litigation or proceeding involving any of our directors, officers, employees or agents in which indemnification will be required or permitted. We are not aware of any threatened litigation or proceeding that may result in a claim for such indemnification.
Indemnification Agreements
We have entered into Indemnification Agreements with each of our current directors and executive officers. The Indemnification Agreements provide for indemnification against expenses, judgments, fines and penalties actually and reasonably incurred by an indemnitee in connection with threatened, pending or completed actions, suits or other proceedings, subject to certain limitations. The Indemnification Agreements also provide for the advancement of expenses in connection with a proceeding prior to a final, nonappealable judgment or other adjudication, provided that the indemnitee provides an undertaking to repay to us any amounts advanced if the indemnitee is ultimately found not to be entitled to indemnification by us. The Indemnification Agreement sets forth procedures for making and responding to a request for indemnification or advancement of expenses, as well as dispute resolution procedures that will apply to any dispute between us and an indemnitee arising under the Indemnification Agreements.
2021 Equity Incentive Plan
General
On March 24, 2021, our board of directors and stockholders adopted the 2021 Plan which provides for the grant of incentive stock options and non-qualified stock options to purchase shares of our common stock and other types of awards. On June 29, 2021, our board of directors and stockholders approved an amendment to the 2021 Plan to increase the aggregate number of shares of common stock available for issuance in connection with options and other awards granted under the 2021 Plan. The general purpose of the 2021 Plan is to provide a means whereby eligible employees, officers, non-employee directors and other individual service providers develop a sense of proprietorship and personal involvement in our development and financial success, and to encourage them to devote their best efforts to our business, thereby advancing our interests and the interests of our stockholders. By means of the 2021 Plan, we seek to retain the services of such eligible persons and to provide incentives for such persons to exert maximum efforts for our success and the success of our subsidiaries.
Description of the 2021 Omnibus Equity Incentive Plan
The following description of the principal terms of the 2021 Plan is a summary and is qualified in its entirety by the full text of the 2021 Plan.
Administration. In general, the 2021 Plan will be administered by the Compensation Committee of the board of directors. The Compensation Committee will determine the persons to whom options to purchase shares of common stock, stock appreciation rights (or SARs), restricted stock units, restricted or unrestricted shares of common stock, performance shares, performance units, incentive bonus awards, other stock-based awards and other cash-based awards may be granted. The Compensation Committee may also establish rules and regulations for the administration of the 2021 Plan and amendments or modifications of outstanding awards. The Compensation Committee may delegate authority to the chief executive officer, other executive officers, 10% or greater stockholders and/or employees of the Company or our subsidiaries to grant options and other awards to employees (other than themselves), subject to applicable law and the 2021 Plan. No options, stock purchase rights or awards may be made under the 2021 Plan on or after March 31, 2031 (or, the expiration date), but the 2021 Plan will continue thereafter while previously granted options, SARs or other awards remain outstanding.
| 116 |
| Table of Contents |
Eligibility. Persons eligible to receive options, SARs or other awards under the 2021 Plan are those employees, officers, directors, consultants, advisors and other individual service providers of our Company and our subsidiaries who, in the opinion of the Compensation Committee, are in a position to contribute to our success, or any person who is determined by the Compensation Committee to be a prospective employee, officer, director, consultant, advisor or other individual service provider of the Company or any subsidiary. As the date of this prospectus, we had eight full-time employees, of which four are executive officers. As awards under the 2021 Plan are within the discretion of the Compensation Committee, we cannot determine how many individuals in each of the categories described above will receive awards.
Shares Subject to the 2021 Plan. The aggregate number of shares of common stock available for issuance in connection with options and other awards granted under the 2021 Plan is 1,731,499.
The number of shares of common stock available for issuance under the 2021 Plan will automatically increase on January 1st of each year, commencing with January 1, 2022, and on each January 1 thereafter until the expiration date, in an amount equal to one percent (1%) of the total number of shares of our common stock outstanding on December 31st of the preceding calendar year, unless the board of directors takes action prior thereto to provide that there will not be an increase in the share reserve for such year or that the increase in the share reserve for such year will be of a lesser number of shares of common stock than would otherwise occur.
“Incentive stock options”, or ISOs, that are intended to meet the requirements of Section 422 of the Internal Revenue Code of 1986, as amended (or, the Code) may be granted under the 2021 Plan with respect to all of the 1,731,499 shares of common stock authorized for issuance under the 2021 Plan. None of the additional shares of common stock available for issuance pursuant to the previous paragraph may be subject to ISOs.
If any option or SAR granted under the 2021 Plan terminates without having been exercised in full or if any award is forfeited, or if shares of common stock are withheld to cover withholding taxes on options or other awards or applied to the payment of the exercise price of an option or purchase price of an award, the number of shares of common stock as to which such option or award was forfeited, withheld or paid, will be available for future grants under the 2021 Plan. Awards settled in cash will not count against the number of shares available for issuance under the 2021 Plan.
No non-employee director may receive awards in any calendar year having an accounting value in excess of $500,000 (inclusive of any cash awards to the non-employee director for such year that are not made pursuant to the 2021 Plan); provided that in the case of a new non-employee director, such amount is increased to $750,000 for the initial year of the non-employee director’s term.
The number of shares authorized for issuance under the 2021 Plan and the foregoing share limitations are subject to customary adjustments for stock splits, stock dividends, similar transactions or any other change affecting our common stock.
Terms and Conditions of Options. Options granted under the 2021 Plan may be either ISOs or “nonstatutory stock options” that do not meet the requirements of Section 422 of the Code. The Compensation Committee will determine the exercise price of options granted under the 2021 Plan. The exercise price of stock options may not be less than the fair market value per share of our common stock on the date of grant (or 110% of fair market value in the case of ISOs granted to a ten-percent stockholder).
If on the date of grant the common stock is listed on a stock exchange or is quoted on the automated quotation system of the Nasdaq Stock Market, the fair market value will generally be the closing sale price on the date of grant (or the last trading day before the date of grant if no trades occurred on the date of grant). If no such prices are available, the fair market value will be determined in good faith by the Compensation Committee based on the reasonable application of a reasonable valuation method.
No option may be exercisable for more than ten years (five years in the case of an ISO granted to a ten-percent stockholder) from the date of grant. Options granted under the 2021 Plan will be exercisable at such time or times as the Compensation Committee prescribes at the time of grant. No employee may receive ISOs that first become exercisable in any calendar year in an amount exceeding $100,000. The Compensation Committee may, in its discretion, permit a holder of an option to exercise the option before it has otherwise become exercisable, in which case the shares of our common stock issued to the recipient will continue to be subject to the vesting requirements that applied to the option before exercise.
| 117 |
| Table of Contents |
Generally, the option price may be paid in cash or by certified check, bank draft or money order. The Compensation Committee may permit other methods of payment, including (a) through delivery of shares of our common stock having a fair market value equal to the purchase price, (b) by a full recourse, interest bearing promissory note having such terms as the Compensation Committee may permit, or (c) a combination of these methods, as set forth in an award agreement or as otherwise determined by the Compensation Committee. The Compensation Committee is authorized to establish a cashless exercise program and to permit the exercise price (or tax withholding obligations) to be satisfied by reducing from the shares otherwise issuable upon exercise a number of shares having a fair market value equal to the exercise price.
No option may be transferred other than by will or by the laws of descent and distribution, and during a recipient’s lifetime an option may be exercised only by the recipient. However, the Compensation Committee may permit the holder of an option, SAR or other award to transfer the option, right or other award to immediate family members, a family trust for estate planning purposes or by gift to charitable institutions. The Compensation Committee will determine the extent to which a holder of a stock option may exercise the option following termination of service with us.
Stock Appreciation Rights. The Compensation Committee may grant SARs under the 2021 Plan. The Compensation Committee will determine the other terms applicable to SARs. The exercise price per share of a SAR will not be less than 100% of the fair market value of a share of our common stock on the date of grant, as determined by the Compensation Committee. The maximum term of any SAR granted under the 2021 Plan is ten years from the date of grant. Generally, each SAR will entitle a participant upon exercise to an amount equal to:
| • |
| the excess of the fair market value on the exercise date of one share of our common stock over the exercise price, multiplied by |
| • |
| the number of shares of common stock covered by the SAR. |
Payment may be made in shares of our common stock, in cash, or partly in common stock and partly in cash, all as determined by the Compensation Committee.
Restricted Stock and Restricted Stock Units. The Compensation Committee may award restricted common stock and/or restricted stock units under the 2021 Plan. Restricted stock awards consist of shares of stock that are transferred to a participant subject to restrictions that may result in forfeiture if specified conditions are not satisfied. Restricted stock units confer the right to receive shares of our common stock, cash, or a combination of shares and cash, at a future date upon or following the attainment of certain conditions specified by the Compensation Committee. The restrictions and conditions applicable to each award of restricted stock or restricted stock units may include performance-based conditions. Dividends or distributions with respect to restricted stock may be paid to the holder of the shares as and when dividends are paid to stockholders or at the time that the restricted stock vests, as determined by the Compensation Committee. If any dividends or distributions are paid in stock before the restricted stock vests they will be subject to the same restrictions. Dividend equivalent amounts may be paid with respect to restricted stock units either when cash dividends are paid to stockholders or when the units vest. Unless the Compensation Committee determines otherwise, holders of restricted stock will have the right to vote the shares.
Performance Shares and Performance Units. The Compensation Committee may award performance shares and/or performance units under the 2021 Plan. Performance shares and performance units are awards, denominated in either shares or U.S. dollars, which are earned during a specified performance period subject to the attainment of performance criteria, as established by the Compensation Committee. The Compensation Committee will determine the restrictions and conditions applicable to each award of performance shares and performance units.
Incentive Bonuses. The Compensation Committee may grant incentive bonus awards under the 2021 Plan from time to time. The terms of incentive bonus awards will be set forth in award agreements. Each award agreement will have such terms and conditions as the Compensation Committee determines, including performance goals and amount of payment based on achievement of such goals. Incentive bonus awards are payable in cash and/or shares of our common stock.
| 118 |
| Table of Contents |
Other Stock-Based and Cash-Based Awards. The Compensation Committee may award other types of equity-based or cash-based awards under the 2021 Plan, including the grant or offer for sale of shares of our common stock that do not have vesting requirements and the right to receive one or more cash payments subject to satisfaction of such conditions as the Compensation Committee may impose.
Effect of Certain Corporate Transactions. The Compensation Committee may, at the time of the grant of an award provide for the effect of a change in control (as defined in the 2021 Plan) on any award, including (i) accelerating or extending the time periods for exercising, vesting in, or realizing gain from any award, (ii) eliminating or modifying the performance or other conditions of an award, or (iii) providing for the cash settlement of an award for an equivalent cash value, as determined by the Compensation Committee. The Compensation Committee may, in its discretion and without the need for the consent of any recipient of an award, also take one or more of the following actions contingent upon the occurrence of a change in control: (a) cause any or all outstanding options and SARs to become immediately exercisable, in whole or in part; (b) cause any other awards to become non-forfeitable, in whole or in part; (c) cancel any option or SAR in exchange for a substitute option; (d) cancel any award of restricted stock, restricted stock units, performance shares or performance units in exchange for a similar award of the capital stock of any successor corporation; (e) redeem any restricted stock for cash and/or other substitute consideration; (f) cancel or terminate any award for cash and/or other substitute consideration in exchange for an amount of cash and/or property equal to the amount, if any, that would have been attained upon the exercise of such award or realization of the participant’s rights as of the date of the occurrence of the change in control, but if the change in control consideration with respect to any option or SAR does not exceed its exercise price, the option or SAR may be canceled without payment of any consideration; or (g) make such other modifications, adjustments or amendments to outstanding awards as the Compensation Committee deems necessary or appropriate.
Amendment, Termination. The board of directors may at any time amend the 2021 Plan for the purpose of satisfying the requirements of the Code, or other applicable law or regulation or for any other legal purpose, provided that, without the consent of our stockholders, the board of directors may not (a) increase the number of shares of common stock available under the 2021 Plan, (b) change the group of individuals eligible to receive options, SARs and/or other awards, or (c) extend the term of the 2021 Plan.
U.S. Federal Income Tax Consequences
Following is a summary of the U.S. federal income tax consequences of option and other grants under the 2021 Plan. Optionees and recipients of other rights and awards granted under the 2021 Plan are advised to consult their personal tax advisors before exercising an option or SAR or disposing of any stock received pursuant to the exercise of an option or SAR or following the vesting and payment of any award. In addition, the following summary is based upon an analysis of the Code as currently in effect, existing laws, judicial decisions, administrative rulings, regulations and proposed regulations, all of which are subject to change and does not address state, local, foreign or other tax laws.
Treatment of Options
The Code treats incentive stock options and nonstatutory stock options differently. However, as to both types of options, no income will be recognized to the optionee at the time of the grant of the options under the 2021 Plan, nor will our Company be entitled to a tax deduction at that time.
Generally, upon exercise of a nonstatutory stock option (including an option intended to be an incentive stock option but which has not continued to so qualify at the time of exercise), an optionee will recognize ordinary income tax on the excess of the fair market value of the stock on the exercise date over the option price. Our Company will be entitled to a tax deduction in an amount equal to the ordinary income recognized by the optionee in the fiscal year which includes the end of the optionee’s taxable year. We will be required to satisfy applicable withholding requirements in order to be entitled to a tax deduction. In general, if an optionee, in exercising a nonstatutory stock option, tenders shares of our common stock in partial or full payment of the option price, no gain or loss will be recognized on the tender. However, if the tendered shares were previously acquired upon the exercise of an incentive stock option and the tender is within two years from the date of grant or one year after the date of exercise of the incentive stock option, the tender will be a disqualifying disposition of the shares acquired upon exercise of the incentive stock option.
| 119 |
| Table of Contents |
For incentive stock options, there is no taxable income to an optionee at the time of exercise. However, the excess of the fair market value of the stock on the date of exercise over the exercise price will be taken into account in determining whether the “alternative minimum tax” will apply for the year of exercise. If the shares acquired upon exercise are held until at least two years from the date of grant and more than one year from the date of exercise, any gain or loss upon the sale of such shares, if held as capital assets, will be long-term capital gain or loss (measured by the difference between the sales price of the stock and the exercise price). Under current federal income tax law, a long-term capital gain will be taxed at a rate which is less than the maximum rate of tax on ordinary income. If the two-year and one year holding period requirements are not met (a “disqualifying disposition”), an optionee will recognize ordinary income in the year of disposition in an amount equal to the lesser of (i) the fair market value of the stock on the date of exercise minus the exercise price or (ii) the amount realized on disposition minus the exercise price. The remainder of the gain will be treated as long-term capital gain, depending upon whether the stock has been held for more than a year. If an optionee makes a disqualifying disposition, our Company will be entitled to a tax deduction equal to the amount of ordinary income recognized by the optionee.
In general, if an optionee, in exercising an incentive stock option, tenders shares of common stock in partial or full payment of the option price, no gain or loss will be recognized on the tender. However, if the tendered shares were previously acquired upon the exercise of another incentive stock option and the tender is within two years from the date of grant or one year after the date of exercise of the other option, the tender will be a disqualifying disposition of the shares acquired upon exercise of the other option.
As noted above, the exercise of an incentive stock option could subject an optionee to the alternative minimum tax. The application of the alternative minimum tax to any particular optionee depends upon the particular facts and circumstances which exist with respect to the optionee in the year of exercise. However, as a general rule, the amount by which the fair market value of the common stock on the date of exercise of an option exceeds the exercise price of the option will constitute an item of “adjustment” for purposes of determining the alternative minimum taxable income on which the alternative tax may be imposed. As such, this item will enter into the tax base on which the alternative minimum tax is computed and may therefore cause the alternative minimum tax to become applicable in any given year.
Treatment of Stock Appreciation Rights
Generally, the recipient of a SAR will not recognize any income upon grant of the SAR, nor will our Company be entitled to a deduction at that time. Upon exercise of a SAR, the holder will recognize ordinary income, and our Company generally will be entitled to a corresponding deduction, equal to the excess of fair market value of our common stock at that time over the exercise price.
Treatment of Stock Awards
Generally, absent an election to be taxed currently under Section 83(b) of the Code (or, a Section 83(b) Election), there will be no federal income tax consequences to either the recipient or our Company upon the grant of a restricted stock award or award of performance shares. At the expiration of the restriction period and the satisfaction of any other restrictions applicable to the restricted shares, the recipient will recognize ordinary income and our Company generally will be entitled to a corresponding deduction equal to the fair market value of the common stock at that time. If a Section 83(b) Election is made within 30 days after the date the restricted stock award is granted, the recipient will recognize an amount of ordinary income at the time of the receipt of the restricted shares, and our Company generally will be entitled to a corresponding deduction, equal to the fair market value (determined without regard to applicable restrictions) of the shares at such time, less any amount paid by the recipient for the shares. If a Section 83(b) Election is made, no additional income will be recognized by the recipient upon the lapse of restrictions on the shares (and prior to the sale of such shares), but, if the shares are subsequently forfeited, the recipient may not deduct the income that was recognized pursuant to the Section 83(b) Election at the time of the receipt of the shares.
| 120 |
| Table of Contents |
The recipient of an unrestricted stock award, including a performance unit award, will recognize ordinary income, and our Company generally will be entitled to a corresponding deduction, equal to the fair market value of our common stock that is the subject of the award when the Award is made.
The recipient of a restricted stock unit generally will recognize ordinary income as and when the units vest and are settled. The amount of the income will be equal to the fair market value of the shares of our common stock issued at that time, and our Company will be entitled to a corresponding deduction. The recipient of a restricted stock unit will not be permitted to make a Section 83(b) Election with respect to such award.
Treatment of Incentive Bonus Awards and Other Stock or Cash Based Awards
Generally, the recipient of an incentive bonus or other stock or cash based award will not recognize any income upon grant of the award, nor will our Company be entitled to a deduction at that time. Upon payment with respect to such an award, the recipient will recognize ordinary income, and our Company generally will be entitled to a corresponding deduction, equal to the amount of cash paid and/or the fair market value of our common stock issued at that time.
Potential Limitation on Company Deductions
Section 162(m) of the Code generally disallows a tax deduction for compensation in excess of $1 million paid in a taxable year by a publicly held corporation to its chief executive officer and certain other “covered employees.” Our Board and the Compensation Committee intend to consider the potential impact of Section 162(m) on grants made under the 2021 Plan, but reserve the right to approve grants of options and other awards for an executive officer that exceed the deduction limit of Section 162(m).
Tax Withholding
As and when appropriate, we shall have the right to require each optionee purchasing shares of common stock and each grantee receiving an award of shares of common stock under the 2021 Plan to pay any federal, state or local taxes required by law to be withheld.
Director compensation
The following table shows for the fiscal year ended December 31, 2021, certain information with respect to the compensation of all non-employee directors of the Company:
Name | Fees Earned or Paid in Cash (1) | Stock Option Awards (2) | Total | |||||||
David Hale | $ | 27,188 | $ | 81,126 | (3) | $ | 108,314 | |||
Wendell Wierenga Ph.D. |
|
| 18,750 |
|
| 81,126 | (3) |
| 99,876 | |
Kathleen Scott | 20,625 | 42,280 | (4) | 62,905 | ||||||
Steven J. Mento Ph.D. |
|
| 19,219 |
|
| 42,280 | (4) |
| 61,499 | |
Mary Fisher | 17,813 | 42,280 | (4) | 60,093 | ||||||
Andrew Sandler M.D. |
|
| 18,281 |
|
| 42,280 | (4) |
| 60,561 | |
Brittany Bradrick | - | - | - | |||||||
_____________
(1) | Board of Director fees earned or paid in cash were for calendar year 2021, representing fees earned by our Directors since our IPO in August 2021. |
(2) | In accordance with SEC rules, this column reflects the aggregate grant date fair value of the stock option awards granted during 2021. These amounts have been computed in accordance with FASB ASC Topic 718. Assumptions used in the calculation of this amount are described in the notes to our financial statements included elsewhere in this document. This amount does not reflect the actual economic value that will be realized by the Directors upon the vesting of the stock options, the exercise of the stock options, or the sale of the common stock underlying such stock options. |
(3) | Mr. Hale and Dr. Wierenga served as Directors prior to our IPO in August 2021. As part of their compensation for prior Board service, Mr. Hale and Dr. Wierenga received stock option grants on March 31, 2021. |
(4) | These stock option awards were granted on August 17, 2021 with a vesting schedule of 1/12th vesting monthly. |
We have adopted a compensation policy pursuant to which our board members receive $40,000 per year ($60,000 for Lead Director), each member of the Audit Committee receives $7,500 per year ($15,000 for the Chair), each member of the Compensation Committee receives $5,000 per year ($10,000 for the Chair), and each member of the Nominating and Corporate Governance Committee receives $4,000 per year ($8,000 for the Chair). Any compensation to be paid under this policy may be made in cash or restricted stock units at the election of each board member which must be made in the prior calendar year.
We have also adopted an equity compensation policy pursuant to which board members shall automatically be granted stock options to purchase 10,000 shares of our common stock upon joining the board of directors, and on January 1 of each year, each then serving non-employee director shall be automatically granted stock options to purchase 5,000 shares of our common stock. These stock options shall have a term of ten years and shall have an exercise price equal to 100% of the fair market value of a share of common stock on the date of grant. All options to be granted under this policy will be granted pursuant to our 2021 Plan.
| 121 |
| Table of Contents |
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
The following is a description of transactions since January 1, 2020 to which we have been a participant in which the amount involved exceeded or will exceed $120,000, and in which any of our directors, executive officers or holders of more than 5% of our voting securities, or any members of their immediate family, had or will have a direct or indirect material interest, other than compensation arrangements which are described under “Executive Compensation.”
Sean Proehl
Sean Proehl, the son of our Chief Executive Officer, is currently employed as our Senior Director, Legal & Business Development. Mr. Sean Proehl receives a salary of $160,000 a year. In addition, Mr. Sean Proehl is eligible to receive, from time to time, equity awards under our existing equity incentive plan, or any other equity incentive plan we may adopt in the future, and the terms and conditions of such awards, if any, will be determined by our board of directors or Compensation Committee, in their discretion.
Sales of Preferred Units
In March 2021, we issued an aggregate of 686,742 Series 1d Preferred Units at $0.83 per unit for gross proceeds of approximately $570,000. Proehl Investment Ventures, LLC, Allison Taylor Proehl 2020 Irrevocable Trust, Meghan Proehl Wilder 2020 Irrevocable Trust and Sean Michael Proehl Irrevocable Trusts Dated December 18, 2020 purchased 307,228, 120,481, 60,240 and 120,481 Series 1d Preferred Units, respectively. Mr. Proehl, our President and Chief Executive Officer and the Chairman of the board of directors, is the managing member of Proehl Investment Ventures, LLC and the trustee of Allison Taylor Proehl 2020 Irrevocable Trust, Meghan Proehl Wilder 2020 Irrevocable Trust and Sean Michael Proehl Irrevocable Trusts Dated December 18, 2020. The shares of Series 1d Preferred Stock held by Mr. Proehl (in the aggregate), automatically converted into 90,203 shares of common stock upon the completion of our initial public offering, at a conversion price equal to $5.60 (80% of the initial offering price).
LLC Conversion
In March 2021, in connection with the conversion of Dermata Therapeutics, LLC into a Delaware corporation, Proehl Investment Ventures, LLC received an aggregate of 35,791,829 shares of our preferred stock, warrants exercisable for 573,750 shares of our preferred stock with an exercise price of $1.00 per share, 917,317 shares of our common stock, and Hale BioPharma Ventures, LLC received an aggregate of 5,089,162 shares of our then-outstanding preferred stock, warrants exercisable for 62,500 shares of our then-outstanding preferred stock with an exercise price of $1.00 per share and 254,634 shares of our common stock. Mr. Hale, a member of the board of directors, is the managing member of Hale BioPharma Ventures, LLC.
Stockholders’ Agreement
On March 24, 2021, in connection with the conversion of Dermata Therapeutics, LLC into a Delaware corporation, we entered into a Stockholders’ Agreement (as amended, the Stockholders’ Agreement) with all of our then-existing stockholders, including Proehl Investment Ventures, LLC and Hale BioPharma Ventures, LLC. The Stockholders’ Agreement among other things, provided for certain restrictions on transfer of our shares of capital stock, set forth agreements and understandings with respect to how shares of our capital stock held by the stockholders party thereto would be voted on, or tendered in connection with, an acquisition of the Company and to provided for certain voting rights with respect to the election of directors. The Stockholders’ Agreement automatically terminated upon the completion of our initial public offering.
| 122 |
| Table of Contents |
Convertible Note Financing
In July 2020, we held the first closing of the Notes for an aggregate principal amount of $2,330,000, including $500,000 from Proehl Investment Ventures, LLC. In October 2020, we held the second closing of the Notes for an aggregate principal amount of $670,000, including $420,000 from Proehl Investment Ventures, LLC. In February 2021, we held the third closing of the Notes for an aggregate principal amount of $1,556,000, including $825,000 from Proehl Investment Ventures, LLC, $100,000 from the Proehl Family Trust and $250,000 Hale BioPharma Ventures, LLC.
On March 15, 2021, we completed the conversion of $4,391,000 of Notes into 5,379,247 Series 1d Preferred Units. At that time Proehl Investment Ventures, LLC, Sean Michael Proehl Irrevocable Trusts Dated December 18, 2020 and Hale BioPharma Ventures, LLC held an aggregate principal amount of $1,745,000, $100,000 and $250,000 of Notes, respectively. Mr. Proehl, our President and Chief Executive Officer and a member of the board of directors, is the managing member of Proehl Investment Ventures, LLC and the trustee of Sean Michael Proehl Irrevocable Trusts Dated December 18, 2020. Mr. Hale, a member of the board of directors, is the managing member of Hale BioPharma Ventures, LLC. In addition, Wendell Wierenga, a member of our board of directors, held $45,000 principal amount of the Notes. Each of Messrs. Proehl, Hale and Wierenga converted their aggregate principal amounts of Notes into Series 1d Preferred Units on March 15, 2021, which units were subsequently converted into shares of our Series 1d Preferred Stock in connection with our conversion to a Delaware corporation. The shares of Series 1d Preferred Stock that were converted from the Notes held by Messrs. Proehl, Hale and Wierenga were automatically converted into 333,494, 44,777 and 8,136 shares of common stock, respectively, upon the completion of our initial public offering, at a conversion price equal to $5.60 (80% of the initial offering price).
The Notes had an interest rate of 4.0 % per annum, were unsecured, had a maturity date of December 31, 2021 and provided for conversion into our common stock upon the earlier of (i) qualified Series A Financing (as defined in the Notes) which resulted in aggregate gross proceeds to the Company of at least ten million dollars ($10,000,000), or (ii) the closing of our initial public offering. Upon the completion of our initial public offering, the aggregate principal amount and all accrued but unpaid interest on the Notes automatically converted into an aggregate of 32,219 shares of our common stock at a conversion price of $5.60 per share (which was 80% of the initial offering price).
Indemnification of Officers and Directors
We have entered into indemnification agreements with each of our current directors and executive officers. These agreements require us to indemnify these individuals to the fullest extent permitted under Delaware law against liabilities that may arise by reason of their service to us, and to advance expenses incurred as a result of any proceeding against them as to which they could be indemnified. We also intend to enter into indemnification agreements with our future directors and executive officers.
Policies and Procedures for Related Party Transactions
We have adopted a policy that our executive officers, directors, nominees for election as a director, beneficial owners of more than 5% of any class of our common stock, any members of the immediate family of any of the foregoing persons and any firms, corporations or other entities in which any of the foregoing persons is employed or is a partner or principal or in a similar position or in which such person has a 5% or greater beneficial ownership interest, or related parties, are not permitted to enter into a transaction with us without the prior consent of our board of directors acting through the audit committee or, in certain circumstances, the chairman of the audit committee. Any request for us to enter into a transaction with a related party, in which the amount involved exceeds $100,000 and such related party would have a direct or indirect interest must first be presented to our audit committee, or in certain circumstances the chairman of our audit committee, for review, consideration and approval. In approving or rejecting any such proposal, our audit committee is to consider the material facts of the transaction, including, but not limited to, whether the transaction is on terms no less favorable than terms generally available to an unaffiliated third party under the same or similar circumstances, the extent of the benefits to us, the availability of other sources of comparable products or services and the extent of the related person’s interest in the transaction.
| 123 |
| Table of Contents |
PRINCIPAL STOCKHOLDERS
The following table sets forth information regarding beneficial ownership of our common stock as of the date of this prospectus by:
| · | each person, or group of affiliated persons, known by us to beneficially own more than 5% of our common stock; |
|
|
|
| · | each of our directors; |
|
|
|
| · | each of our named executive officers; and |
|
|
|
| · | all of our current executive officers and directors as a group. |
We have determined beneficial ownership in accordance with the rules of the SEC. Under these rules, beneficial ownership includes any shares of common stock as to which the individual or entity has sole or shared voting power or investment power. Applicable percentage ownership is based on 8,328,629 shares of common stock outstanding as of the date of this prospectus. The percentage of beneficial ownership after this offering assumes the sale and issuance of shares of common stock in this offering (based upon the assumed sale of 4,444,444 Units in this offering at an assumed public offering price of $2.25 per Unit, the last reported sale price of our common stock on Nasdaq on January 31, 2022) and assuming no exercise of the Warrants issued as part of the Units, no sale of Units including a Pre-Funded Warrant, and no exercise by the underwriter of its option to purchase additional shares of common stock and/or Warrants.
In computing the number of shares beneficially owned by an individual or entity and the percentage ownership of that person, shares of common stock subject to options held by such person that are currently exercisable or will become exercisable within 60 days of the date of this prospectus are considered outstanding, although these shares are not considered outstanding for purposes of computing the percentage ownership of any other person.
Each of the stockholders listed has sole voting and investment power with respect to the shares beneficially owned by the stockholder unless noted otherwise, subject to community property laws where applicable.
|
| Number of Shares |
|
| Percentage of Shares |
| ||||||
|
| Beneficially Owned |
|
| Beneficially Owned |
| ||||||
| Name of Beneficial Owner (1) |
| Prior to Offering (2) |
|
| Prior to Offering |
|
| After Offering |
| |||
|
|
|
|
|
|
|
|
|
| |||
| 5% of Greater Shareholders |
|
|
|
|
|
|
|
|
| |||
| Proehl Investment Ventures LLC |
|
| 2,933,531 | (3)(4) |
|
| 34.5 | % |
|
| 22.7 | % |
| Hale BioPharma Ventures LLC |
|
| 535,973 | (9)(10) |
|
| 6.4 | % |
|
| 4.2 | % |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Named Executive Officers and Directors other than |
|
|
|
|
|
|
|
|
|
|
|
|
| 5% or Greater Shareholders |
|
|
|
|
|
|
|
|
|
|
|
|
| Gerald T. Proehl |
|
| 3,651,484 | (3)(4)(5) |
|
| 41.4 | % |
|
| 27.5 | % |
| Christopher J. Nardo, M.P.H., Ph.D. |
|
| 63,974 | (6) |
| * |
|
| * |
| ||
| Maria Bedoya Toro Munera, Ph.D., M.B.A. |
|
| 146,242 | (7) |
|
| 1.7 | % |
|
| 1.1 | % |
| Kyri K. Van Hoose |
|
| 10,937 | (8) |
| * |
|
| * |
| ||
| David Hale |
|
| 641,576 | (9)(10)(11) |
|
| 7.7 | % |
|
| 5.0 | % |
| Wendell Wierenga, Ph.D. |
|
| 31,363 | (12) |
| * |
|
| * |
| ||
| Kathleen Scott |
|
| 9,522 | (13) |
| * |
|
| * |
| ||
| Steven J. Mento, Ph.D. |
|
| 7,083 | (14) |
| * |
|
| * |
| ||
| Mary Fisher |
|
| 7,083 | (15) |
| * |
|
| * |
| ||
| Andrew Sandler, M.D. |
|
| 7,083 | (16) |
| * |
|
| * |
| ||
| Brittany Bradrick, M.B.A. |
|
| 1,666 | (17) |
| * |
|
| * |
| ||
| All Directors and Officers as a Group |
|
| 4,578,013 |
|
|
| 50.9 | % |
|
| 34.1 | % |
| (11 persons) |
|
|
|
|
|
|
|
|
|
|
|
|
___________
* | Less than 1%. |
|
|
(1) | Unless noted otherwise, the address of all listed stockholder is 3525 Del Mar Heights Rd., #322 San Diego, CA 92130. Each of the stockholder listed has sole voting and investment power with respect to the shares beneficially owned by the stockholder unless noted otherwise, subject to community property laws where applicable. |
|
|
(2) | We have determined beneficial ownership in accordance with Rule 13d-3 under the Securities Exchange Act of 1934, as amended, which is generally determined by voting power and/or dispositive power with respect to securities. Unless otherwise noted, the shares of common stock listed above are owned as of the date of this prospectus, and are owned of record by each individual named as beneficial owner and such individual has sole voting and dispositive power with respect to the shares of common stock owned by each of them. |
|
|
(3) | Gerald T. Proehl, our Chairman and Chief Executive Officer is the Chairman and Chief Executive Officer of Proehl Investment Ventures LLC. Due to Mr. Proehl s ownership of Proehl Investment Ventures LLC, he may be deemed to have sole voting and dispositive control over the shares of our common stock held by Proehl Investment Ventures LLC. As a result, Mr. Proehl may be deemed to beneficially own the shares of our common stock held by Proehl Investment Ventures LLC. |
| 124 |
| Table of Contents |
(4) | Includes (i) 2,905,544 shares of common stock held by Proehl Investment Ventures LLC, (ii) 27,987 shares of common stock issuable upon exercise of warrants held by Proehl Investment Ventures LLC that are exercisable within 60 days of the date of this prospectus. |
|
|
(5) | Includes (i) 59,512 shares of common stock held by Mr. Proehl, (ii) 310,175 shares of common stock issuable upon exercise of stock options held by Mr. Proehl exercisable within 60 days of the date of this prospectus, (iii) 8,928 shares of common stock held by Mr. Proehl as Trustee of the Megan Proehl Wilder 2020 Irrevocable Trust, (iv) 17,857 shares of common stock held by Mr. Proehl as Trustee of the Allison Taylor Proehl 2020 Irrevocable Trust, and (v) 35,767 shares of common stock held by Mr. Proehl as Trustee of the Sean Michael Proehl Irrevocable Trust Dated December 18, 2020, and (vi) 142,857 shares of common stock and warrants to purchase up to 142,857 shares of Common Stock held by Mr. Proehl as Trustee of the Proehl Family Trust. Does not include 158,485 shares of common stock issuable upon exercise of stock options held by Mr. Proehl that are not exercisable within 60 days of the date of this prospectus. |
|
|
(6) | Includes (i) 25,121 shares of common stock held by Dr. Nardo, (ii) 36,414 shares of common stock issuable upon exercise of stock options held by Dr. Nardo exercisable within 60 days of the date of this prospectus, and (iii) 2,439 shares of common stock held by Dr. Nardo as Co-Trustee of the Nardo Family Trust Dated October 3, 2001. Does not include 58,584 shares of common stock issuable upon exercise of stock options held by Dr. Nardo that are not exercisable within 60 days of the date of this prospectus. |
|
|
(7) | Includes (i) 30,487 shares of common stock held by Dr. Munera, (ii) 26,734 shares of common stock issuable upon exercise of stock options held by Dr. Munera exercisable within 60 days of the date of this prospectus, (iii) 84,143 shares of common stock held by Dr. Munera as Co-Trustee of the Munera Family Trust, and (iv) 4,878 shares of common stock issuable upon exercise of warrants held by Dr. Munera as Co-Trustee of the Munera Family Trust that are exercisable within 60 days of the date of this prospectus. Does not include 20,947 shares of common stock issuable upon exercise of stock options held by Dr. Munera that are not exercisable within 60 days of the date of this prospectus. |
|
|
(8) | Includes 10,937 shares of common stock issuable upon exercise of stock options held by Ms. Van Hoose exercisable within 60 days of the date of this prospectus. Does not include 94,063 shares of common stock issuable upon exercise of stock options held by Ms. Van Hoose that are not exercisable within 60 days of the date of this prospectus. |
|
|
(9) | David Hale, our Lead Director, is the Chairman and Chief Executive Officer of Hale BioPharma Ventures LLC. Due to Mr. Hale s control of Hale BioPharma Ventures LLC, he may be deemed to have sole voting and dispositive control over the shares of our common stock held by Hale BioPharma Ventures LLC. As a result, Mr. Hale may be deemed to beneficially own the shares of our common stock held by Hale BioPharma Ventures LLC. |
|
|
(10) | Includes (i) 532,925 shares of common stock held by Hale BioPharma Ventures LLC, and (ii) 3,048 shares of common stock held by Hale BioPharma Ventures LLC issuable upon exercise of warrants exercisable within 60 days of the date of this prospectus. |
|
|
(11) | Includes (i) 3,658 shares of common stock held by Mr. Hale, (ii) 18,322 shares of common stock issuable upon exercise of stock options held by Mr. Hale exercisable within 60 days of the date of this prospectus, and (iii) 12,195 shares of common stock held by a limited partnership of which Mr. Hale serves as the General Partner and as such, has voting and dispositive control over the shares of common stock, and (iv) 35,714 shares of common stock and warrants to purchase up to 35,714 shares of Common Stock held by Mr. Hale as Trustee of the Hale Family Trust. Does not include 3,750 shares of common stock issuable upon exercise of stock options held by Mr. Hale that are not exercisable within 60 days of the date this prospectus. |
|
|
(12) | Includes (i) 13,041 shares of common stock held by Dr. Wierenga, and (ii) 18,322 shares of common stock issuable upon exercise of stock options held by Dr. Wierenga exercisable within 60 days of this prospectus. Does not include 3,750 shares of common stock issuable upon exercise of stock options that are not exercisable within 60 days of the date of this prospectus. |
|
|
(13) | Includes (i) 2,439 shares of common stock held by Ms. Scott as Trustee of the Scott 2008 Trust dated 3/28/08, and (ii) 7,083 shares of common stock issuable upon exercise of stock options held by Ms. Scott exercisable within 60 days of this prospectus. Does not include 7,917 shares of common stock issuable upon exercise of stock options that are not exercisable within 60 days of the date of this prospectus. |
|
|
(14) | Includes 7,083 shares of common stock issuable upon exercise of stock options held by Dr. Mento exercisable within 60 days of the date of this prospectus. Does not include 7,917 shares of common stock issuable upon exercise of stock options held by Dr. Mento that are not exercisable within 60 days of the date of this prospectus. |
|
|
(15) | Includes 7,083 shares of common stock issuable upon exercise of stock options held by Ms. Fisher exercisable within 60 days of the date of this prospectus. Does not include 7,917 shares of common stock issuable upon exercise of stock options held by Ms. Fisher that are not exercisable within 60 days of the date of this prospectus. |
|
|
(16) | Includes 7,083 shares of common stock issuable upon exercise of stock options held by Dr. Sandler exercisable within 60 days of the date of this prospectus. Does not include 7,917 shares of common stock issuable upon exercise of stock options held by Dr. Sandler that are not exercisable within 60 days of the date of this prospectus. |
|
|
(17) | Includes 1,666 shares of common stock issuable upon exercise of stock options held by Ms. Bradrick exercisable within 60 days of the date of this prospectus. Does not include 8,334 shares of common stock issuable upon exercise of stock options held by Ms. Bradrick that are not exercisable within 60 days of the date of this prospectus. |
| 125 |
| Table of Contents |
MARKET PRICE OF OUR COMMON STOCK AND RELATED STOCKHOLDER MATTERS
Our common stock and warrants trade on Nasdaq under the symbols “DRMA” and “DRMAW,” respectively. We do not intend to apply for the listing of the Warrants or the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system, including Nasdaq.
As of January 28, 2022, there were approximately 87 holders of record of our common stock and one holder of record of our warrants. This number does not include beneficial owners whose shares were held in street name. The actual number of holders of our common stock and warrants is greater than this number of record holders and includes stockholders and warrant holders who are beneficial owners, but whose shares or warrants are held in street name by brokers or held by other nominees.
DESCRIPTION OF SECURITIES
The following description summarizes the most important terms of our capital stock. Because it is only a summary, it does not contain all the information that may be important to you. For a complete description, you should refer to our restated certificate of incorporation and restated bylaws, which are included as exhibits to the registration statement of which this prospectus forms a part, and to the applicable provisions of Delaware law.
The following is a summary of all material characteristics of our capital stock as set forth in our amended and restated certificate of incorporation and amended and restated bylaws. The summary does not purport to be complete and is qualified in its entirety by reference to our amended and restated certificate of incorporation and amended and restated bylaws, all of which are incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and the applicable provisions of Delaware law.
Authorized Capitalization
We have 100,000,000 shares of capital stock authorized under our amended and restated certificate of incorporation, consisting of 90,000,000 shares of common stock with a par value of $0.0001 per share and 10,000,000 shares of preferred stock with a par value of $0.0001 per share. As of the date of this prospectus, there were 8,328,629 shares of common stock outstanding. In addition, as of the date of this prospectus, we had outstanding options to purchase an aggregate of 523,199 shares of our common stock under the 2021 Plan, at a weighted average exercise price equal to $5.84 per share. Our authorized but unissued shares of common stock and preferred stock are available for issuance without further action by our stockholders, unless such action is required by applicable law or the rules of any stock exchange or automated quotation system on which our securities may be listed or traded in the future.
Common Stock
Holders of our common stock are entitled to such dividends as may be declared by our board of directors out of funds legally available for such purpose. The shares of common stock are neither redeemable nor convertible. Holders of common stock have no preemptive or subscription rights to purchase any of our securities.
Each holder of our common stock is entitled to one vote for each such share outstanding in the holder’s name. No holder of common stock is entitled to cumulate votes in voting for directors.
In the event of our liquidation, dissolution or winding up, the holders of our common stock are entitled to receive pro rata our assets, which are legally available for distribution, after payments of all debts and other liabilities. All of the outstanding shares of our common stock are fully paid and non-assessable. The shares of common stock offered by this prospectus will also be fully paid and non-assessable.
| 126 |
| Table of Contents |
Preferred Stock
Our board of directors has the authority, without further action by our stockholders, to issue up to 10,000,000 shares of preferred stock in one or more classes or series and to fix the designations, rights, preferences, privileges and restrictions thereof, without further vote or action by the stockholders. These rights, preferences and privileges could include dividend rights, conversion rights, voting rights, terms of redemption, liquidation preferences, sinking fund terms and the number of shares constituting, or the designation of, such class or series, any or all of which may be greater than the rights of common stock. The issuance of our preferred stock could adversely affect the voting power of holders of common stock and the likelihood that such holders will receive dividend payments and payments upon our liquidation. In addition, the issuance of preferred stock could have the effect of delaying, deferring or preventing a change in control of our company or other corporate action. Immediately after completion of this offering, no shares of preferred stock will be outstanding, and we have no present plan to issue any shares of preferred stock.
Stock Options
As of the date of this prospectus, we had reserved the following shares of common stock for issuance pursuant to stock options under the 2021 Plan described below:
| · | 1,000,326 shares of our common stock reserved for issuance under stock option agreements issued pursuant to the 2021 Plan with a weighted-average exercise price of $3.96 per share; and |
|
|
|
| · | 731,173 shares of our common stock reserved for future issuance under the 2021 Plan. |
Warrants Offered in this Offering
The following summary of certain terms and provisions of the Warrants offered hereby is not complete and is subject to, and qualified in its entirety by the provisions of the form of Warrant, which is filed as an exhibit to the registration statement of which this prospectus is a part. Prospective investors should carefully review the terms and provisions set forth in the form of Warrant.
The Warrants issued in this offering entitle the registered holders to purchase common stock at a price equal to 100% of the offering price of the Unit sold in this offering, subject to adjustment as discussed below, immediately following the issuance of such Warrants and terminating at 5:00 p.m., New York City time, five years after the closing of this offering.
The exercise price and number of shares of common stock issuable upon exercise of the Warrants may be adjusted in certain circumstances, including in the event of a stock dividend or recapitalization, reorganization, merger or consolidation. However, the Warrants will not be adjusted for issuances of shares of common stock at prices below its exercise price.
Exercisability. The Warrants are exercisable immediately upon issuance and at any time up to the date that is five years from the date of issuance. The Warrants will be exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed exercise notice accompanied by payment in full for the number of shares of common stock purchased upon such exercise. Each Warrant entitles the holder thereof to purchase one share of common stock. Warrants are not exercisable for a fraction of a share and may only be exercised into whole numbers of shares. In lieu of fractional shares, we will, pay the holder an amount in cash equal to the fractional amount multiplied by the exercise price and round down to the nearest whole share. Unless otherwise specified in the Warrant, the holder will not have the right to exercise the Warrants, in whole or in part, if the holder (together with its affiliates) would beneficially own in excess of 4.99% (or 9.99% at the holder’s election) of the number of our shares of common stock outstanding immediately after giving effect to the exercise, as such percentage is determined in accordance with the terms of the Warrant. However, any holder may increase or decrease such percentage to any other percentage not in excess of 9.99% upon at least 61 days’ prior notice from the holder to us.
Exercise Price. The exercise price per share of common stock purchasable upon exercise of the Warrants is no less than 100% of the public offering price per Unit, and is subject to adjustments for stock splits, reclassifications, subdivisions, and other similar transactions. In addition to the exercise price per share of common stock, and other applicable charges and taxes are due and payable upon exercise.
Warrant Agent; Global Certificate. The Warrants will be issued in registered form under a warrant agency agreement between a warrant agent and us. The Warrants will initially be represented only by one or more global warrants deposited with the warrant agent, as custodian on behalf of The Depository Trust Company, or DTC, and registered in the name of Cede & Co., a nominee of DTC, or as otherwise directed by DTC.
| 127 |
| Table of Contents |
Transferability. Subject to applicable laws, the Warrants may be transferred at the option of the holders upon surrender of the Warrants to the warrant agent, together with the appropriate instruments of transfer.
Exchange Listing. There is no established public trading market for the Warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply to list the Warrants on any national securities exchange or other nationally recognized trading system, including the Nasdaq Capital Market. Without an active trading market, the liquidity of Warrants will be limited.
Adjustments; Fundamental Transaction. The exercise price and the number of shares underlying the Warrants are subject to appropriate adjustment in the event of stock splits, stock dividends on our common shares, stock combinations or similar events affecting our common shares. In addition, in the event we consummate a merger or consolidation with or into another person or other reorganization event in which our common shares are converted or exchanged for securities, cash or other property, or we sell, lease, license, assign, transfer, convey or otherwise dispose of all or substantially all of our assets or we or another person acquire 50% or more of our outstanding common shares (each, a Fundamental Transaction), then following such Fundamental Transaction the holders of the Warrants will be entitled to receive upon exercise of the Warrants the same kind and amount of securities, cash or property which the holders would have received had they exercised the Warrants immediately prior to such Fundamental Transaction. Any successor to us or surviving entity will assume the obligations under the warrants. Additionally, as more fully described in the Warrant, in the event of certain Fundamental Transactions, the holders of the Warrants will be entitled to receive consideration in an amount equal to the Black Scholes value of the Warrants on the date of consummation of such transaction.
Rights as a Shareholder. Except by virtue of such holder’s ownership of our common stock, the holder of a Warrant does not have rights or privileges of a shareholder, including any voting rights, until the holder exercises such Warrant.
Pre-Funded Warrants Offered in this Offering
The following summary of certain terms and provisions of the Pre-Funded Warrants offered hereby is not complete and is subject to, and qualified in its entirety by the provisions of the form of Pre-Funded Warrant, which is filed as an exhibit to the registration statement of which this prospectus is a part. Prospective investors should carefully review the terms and provisions set forth in the form of Pre-Funded Warrant.
The term “pre-funded” refers to the fact that the purchase price of our common stock in this offering includes almost the entire exercise price that will be paid under the Pre-Funded Warrants, except for a nominal remaining exercise price of $0.0001. The purpose of the Pre-Funded Warrants is to enable investors that may have restrictions on their ability to beneficially own more than 4.99% (or, upon election of the holder, 9.99%) of our outstanding common stock following the consummation of this offering the opportunity to invest capital into our Company without triggering their ownership restrictions, by receiving Pre-Funded Warrants in lieu of our common stock which would result in such ownership of more than 4.99% (or 9.99%), and receive the ability to exercise their option to purchase the shares underlying the Pre-Funded Warrants at such nominal price at a later date.
Duration. The Pre-Funded Warrants offered hereby will entitle the holders thereof to purchase shares of our common stock at a nominal exercise price of $0.0001 per share, at any time after its original issuance until exercised in full.
Exercise Limitation. A holder will not have the right to exercise any portion of the Pre-Funded Warrant if the holder (together with its affiliates and certain related parties) would beneficially own in excess of 4.99% (or, upon election of the holder, 9.99%) of the number of shares of our common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the Pre-Funded Warrants. However, any holder may increase, but not in excess of 9.99%, or decrease such percentage, provided that any increase will not be effective until the 61st day after such election.
Exercise Price. The Pre-Funded Warrants will have an exercise price of $0.0001 per share. The exercise price is subject to appropriate adjustment in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting our common stock and also upon any distributions of assets, including cash, stock or other property to our stockholders.
| 128 |
| Table of Contents |
Transferability. Subject to applicable laws, the Pre-Funded Warrants may be offered for sale, sold, transferred or assigned without our consent.
Exchange Listing. There is no established public trading market for the Pre-Funded Warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply to list the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system, including the Nasdaq Capital Market. Without an active trading market, the liquidity of Pre-Funded Warrants will be limited.
Fundamental Transactions. If a fundamental transaction occurs, then the successor entity will succeed to, and be substituted for us, and may exercise every right and power that we may exercise and will assume all of our obligations under the Pre-Funded Warrants with the same effect as if such successor entity had been named in the Pre-Funded Warrant itself. If holders of our common stock are given a choice as to the securities, cash or property to be received in a fundamental transaction, then the holder shall be given the same choice as to the consideration it receives upon any exercise of the Pre-Funded Warrant following such fundamental transaction.
Rights as a Stockholder. Except as otherwise provided in the Pre-Funded Warrants or by virtue of such holder’s ownership of shares of our common stock, the holder of a Pre-Funded Warrant does not have the rights or privileges of a holder of our common stock, including any voting rights, until the holder exercises the Pre-Funded Warrant.
Warrants Issued in IPO
In connection with our IPO, we sold warrants to purchase up to 2,571,428 shares of our common stock. Other than the exercise price of the Warrants sold in this offering, terms of the warrants sold in our IPO are identical to the Warrants being issued in this offering, as described above. The warrants are currently listed on Nasdaq and trade under the symbol “DRMAW.”
Underwriters’ Warrants
In connection with our IPO, we agreed to sell to the representative of the underwriters, for nominal consideration, warrants to purchase 128,571 shares of our common stock as additional consideration to the underwriters. The underwriters’ warrants have an exercise price of $8.05 per share and are exercisable for a period of five years and contain customary “cashless” exercise and registration rights provisions. The warrant issued to the underwriters are not be exercisable until February 12, 2022.
Anti-Takeover Effects of Delaware law and Our Certificate of Incorporation and Bylaws
The provisions of Delaware law, our amended and restated certificate of incorporation and our amended and restated bylaws described below may have the effect of delaying, deferring or discouraging another party from acquiring control of us.
Section 203 of the Delaware General Corporation Law
We are subject to Section 203 of the Delaware General Corporation Law, which prohibits a Delaware corporation from engaging in any business combination with any interested stockholder for a period of three years after the date that such stockholder became an interested stockholder, with the following exceptions:
| · | before such date, the board of directors of the corporation approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder; |
|
|
|
| · | upon completion of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction began, excluding for purposes of determining the voting stock outstanding (but not the outstanding voting stock owned by the interested stockholder) those shares owned (i) by persons who are directors and also officers and (ii) employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or |
|
|
|
| · | on or after such date, the business combination is approved by the board of directors and authorized at an annual or special meeting of the stockholders, and not by written consent, by the affirmative vote of at least 66 2/3% of the outstanding voting stock that is not owned by the interested stockholder. |
| 129 |
| Table of Contents |
In general, Section 203 defines business combination to include the following:
| · | any merger or consolidation involving the corporation and the interested stockholder; |
|
|
|
| · | any sale, transfer, pledge or other disposition of 10% or more of the assets of the corporation involving the interested stockholder; |
|
|
|
| · | subject to certain exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder; |
|
|
|
| · | any transaction involving the corporation that has the effect of increasing the proportionate share of the stock or any class or series of the corporation beneficially owned by the interested stockholder; or |
|
|
|
| · | the receipt by the interested stockholder of the benefit of any loss, advances, guarantees, pledges or other financial benefits by or through the corporation. |
In general, Section 203 defines an “interested stockholder” as an entity or person who, together with the person’s affiliates and associates, beneficially owns, or within three years prior to the time of determination of interested stockholder status did own, 15% or more of the outstanding voting stock of the corporation.
Certificate of Incorporation and Bylaws
Our amended and restated certificate of incorporation and amended and restated bylaws provide for:
| · | classifying our board of directors into three classes; |
|
|
|
| · | authorizing the issuance of “blank check” preferred stock, the terms of which may be established and shares of which may be issued without stockholder approval; |
|
|
|
| · | limiting the removal of directors by the stockholders; |
|
|
|
| · | requiring a supermajority vote of stockholders to amend our bylaws or certain provisions our certificate of incorporation; |
|
|
|
| · | prohibiting stockholder action by written consent, thereby requiring all stockholder actions to be taken at a meeting of our stockholders; |
|
|
|
| · | eliminating the ability of stockholders to call a special meeting of stockholders; |
|
|
|
| · | establishing advance notice requirements for nominations for election to the board of directors or for proposing matters that can be acted upon at stockholder meetings; and |
|
|
|
| · | establishing Delaware as the exclusive jurisdiction for certain stockholder litigation against us. |
| 130 |
| Table of Contents |
Potential Effects of Authorized but Unissued Stock
Pursuant to our amended and restated certificate of incorporation, we have shares of common stock and preferred stock available for future issuance without stockholder approval. We may utilize these additional shares for a variety of corporate purposes, including future public offerings to raise additional capital, to facilitate corporate acquisitions or payment as a dividend on the capital stock.
The existence of unissued and unreserved common stock and preferred stock may enable our board of directors to issue shares to persons friendly to current management or to issue preferred stock with terms that could render more difficult or discourage a third-party attempt to obtain control of us by means of a merger, tender offer, proxy contest or otherwise, thereby protecting the continuity of our management. In addition, the board of directors has the discretion to determine designations, rights, preferences, privileges and restrictions, including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences of each series of preferred stock, all to the fullest extent permissible under the Delaware General Corporation Law and subject to any limitations set forth in our certificate of incorporation. The purpose of authorizing the board of directors to issue preferred stock and to determine the rights and preferences applicable to such preferred stock is to eliminate delays associated with a stockholder vote on specific issuances. The issuance of preferred stock, while providing desirable flexibility in connection with possible financings, acquisitions and other corporate purposes, could have the effect of making it more difficult for a third-party to acquire, or could discourage a third-party from acquiring, a majority of our outstanding voting stock.
Choice of Forum
Unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware shall be the sole and exclusive forum for any stockholder to bring (i) any derivative action or proceeding brought on behalf of the Company, (ii) any action asserting a claim of breach of fiduciary duty owed by any director, officer or other employee of the Company or the Company’s stockholders, (iii) any action asserting a claim against the Company or any director or officer of the Company arising pursuant to, or a claim against the Company or any director or officer of the Company, with respect to the interpretation or application of any provision of, the DGCL, our certificate of incorporation or bylaws, or (iv) any action asserting a claim governed by the internal affairs doctrine, except for, in each of the aforementioned actions, any claims to which the Court of Chancery of the State of Delaware determines it lacks jurisdiction. This provision will not apply to claims arising under the Exchange Act, or for any other federal securities laws which provide for exclusive federal jurisdiction. However, the exclusive forum provision provides that unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States of America will be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act. Therefore, this provision could apply to a suit that falls within one or more of the categories enumerated in the exclusive forum provision and that asserts claims under the Securities Act, inasmuch as Section 22 of the Securities Act creates concurrent jurisdiction for federal and state courts over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder. There is uncertainty as to whether a court would enforce such an exclusive forum provision with respect to claims under the Securities Act.
We note that there is uncertainty as to whether a court would enforce the provision and that investors cannot waive compliance with the federal securities laws and the rules and regulations thereunder. Although we believe this provision benefits us by providing increased consistency in the application of Delaware law in the types of lawsuits to which it applies, the provision may have the effect of discouraging lawsuits against our directors and officers.
Transfer Agent
The name, address and telephone number of our stock transfer agent is Direct Transfer, LLC, 500 Perimeter Park Dr., Suite D, Morrisville, NC 27560, (919) 744-2722.
Listing
Our common stock and certain of our warrants are listed on Nasdaq under the symbols “DRMA” and “DRMAW,” respectively. We do not intend to apply for the listing of the Warrants or the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system, including Nasdaq.
Lock-Up Agreements
Pursuant to certain “lock-up” agreements, we and our executive officers and directors have agreed not to, without the prior written consent of the representative of the underwriters, offer, sell, assign, transfer, pledge, contract to sell, or otherwise dispose of or announce the intention to otherwise dispose of, or enter into any swap, hedge or similar agreement or arrangement that transfers, in whole or in part, the economic risk of ownership of, directly or indirectly, engage in any short selling of any common stock or securities convertible into or exchangeable or exercisable for any common stock, whether currently owned or subsequently acquired, for a period of 90 days from the date of this prospectus.
| 131 |
| Table of Contents |
MATERIAL U.S. FEDERAL INCOME TAX CONSEQUENCES
The following discussion is a summary of the material U.S. federal income tax consequences of the purchase, ownership and disposition of the shares of common stock, Pre-Funded Warrants and Warrants acquired pursuant to this offering but does not purport to be a complete analysis of all potential tax effects. The effects of other U.S. federal tax laws, such as estate and gift tax laws, and any applicable state, local or foreign tax laws are not discussed. This discussion is based on the Code, Treasury Regulations promulgated thereunder, judicial decisions, and published rulings and administrative pronouncements of the IRS, in effect as of the date of this offering. These authorities may change or be subject to differing interpretations. Any such change or differing interpretation may be applied retroactively in a manner that could adversely affect a U.S. holder of our common stock, Pre-Funded Warrants and Warrants. We have not sought and will not seek any rulings from the IRS regarding the matters discussed below. There can be no assurance the IRS or a court will not take a contrary position regarding the tax consequences of the purchase, ownership and disposition of our common stock, Pre-Funded Warrants and Warrants.
We assume in this discussion that each holder holds shares of our common stock, Pre-Funded Warrants and Warrants as “capital assets” within the meaning of Section 1221 of the Code (generally, property held for investment). This discussion does not address all U.S. federal income tax consequences that may be relevant to a particular holder’s individual circumstances, including the impact of the alternative minimum tax or the unearned income Medicare contribution tax. In addition, it does not address consequences relevant to holders subject to particular rules, including, without limitation:
| · | U.S. expatriates and certain former citizens or long-term residents of the United States; |
|
|
|
| · | persons holding our common stock, Pre-Funded Warrants or Warrants as part of a hedge, straddle or other risk reduction strategy or as part of a conversion transaction or other integrated investment; |
|
|
|
| · | banks, insurance companies, and other financial institutions; |
|
|
|
| · | regulated investment companies or real estate investment trusts; |
|
|
|
| · | brokers, dealers or traders in securities or currencies; |
|
|
|
| · | controlled foreign corporations, “passive foreign investment companies,” and corporations that accumulate earnings to avoid U.S. federal income tax; |
|
|
|
| · | partnerships or other entities or arrangements treated as partnerships for U.S. federal income tax purposes (and investors therein); |
|
|
|
| · | tax-exempt organizations or governmental organizations; |
|
|
|
| · | persons deemed to sell our common stock, Pre-Funded Warrants or Warrants under the constructive sale provisions of the Code; |
|
|
|
| · | persons for whom our common stock or Pre-Funded Warrants constitutes “qualified small business stock” within the meaning of Section 1202 of the Code or as “Section 1244 stock” for purposes of Section 1244 of the Code; |
|
|
|
| · | persons subject to special tax accounting rules as a result of any item of gross income with respect to our common stock, Pre-Funded Warrants or Warrants being taken into account in an “applicable financial statement” (as defined in the Code); |
|
|
|
| · | persons who hold or receive our common stock, Pre-Funded Warrants or Warrants pursuant to the exercise of any employee stock option or otherwise as compensation; |
|
|
|
| · | tax-qualified retirement plans; and |
|
|
|
| · | “qualified foreign pension funds” as defined in Section 897(l)(2) of the Code and entities all of the interest of which are held by qualified foreign pension funds |
If a partnership (or other entity treated as a partnership for U.S. federal income tax purposes) holds our common stock, Pre-Funded Warrants or Warrants, the tax treatment of a partner in the partnership will depend on the status of the partner, the activities of the partnership and certain determinations made at the partner level. Accordingly, partnerships holding our common stock, Pre-Funded Warrants or Warrants, and the partners in such partnerships should consult their tax advisors regarding the U.S. federal income tax consequences to them.
| 132 |
| Table of Contents |
THIS DISCUSSION IS FOR INFORMATION PURPOSES ONLY AND IS NOT INTENDED AS LEGAL OR TAX ADVICE. INVESTORS SHOULD CONSULT THEIR TAX ADVISORS WITH RESPECT TO THE APPLICATION OF THE U.S. FEDERAL INCOME TAX LAWS TO THEIR PARTICULAR SITUATIONS AS WELL AS ANY TAX CONSEQUENCES OF THE PURCHASE, OWNERSHIP AND DISPOSITION OF OUR COMMON STOCK, PRE-FUNDED WARRANTS, AND WARRANTS ARISING UNDER THE U.S. FEDERAL ESTATE OR GIFT TAX LAWS OR UNDER THE LAWS OF ANY STATE, LOCAL OR NON-U.S. TAXING JURISDICTION OR UNDER ANY APPLICABLE INCOME TAX TREATY.
For purposes of this discussion, a “U.S. holder” is any beneficial owner of our common stock, Pre-Funded Warrants, or Warrants that, for U.S. federal income tax purposes, is:
| · | an individual who is a citizen or resident of the United States; |
|
|
|
| · | a corporation (or other entity treated as a corporation for U.S. federal income tax purposes) created or organized in or under the laws of the United States, any state thereof, or the District of Columbia; |
|
|
|
| · | an estate, the income of which is subject to U.S. federal income tax regardless of its source; or |
|
|
|
| · | a trust that (1) is subject to the primary supervision of a U.S. court and the control of one or more United States persons (within the meaning of Section 7701(a)(30) of the Code), or (2) has made a valid election under applicable Treasury Regulations to be treated as a United States person for U.S. federal income tax purposes. |
The term “non-U.S. holder” means any beneficial owner of our common stock, Pre-Funded Warrants or Warrants that is not a U.S. holder and is not a partnership or other entity properly classified as a partnership for U.S. federal income tax purposes. For the purposes of this discussion, U.S. holders and non-U.S. holders are referred to collectively as “holders.”
General Treatment of Pre-Funded Warrants
Although it is not entirely free from doubt, a Pre-Funded Warrant should be treated as a share of our common stock for U.S. federal income tax purposes and a holder of Pre-Funded Warrants should generally be taxed in the same manner as a holder of common stock as described below. Each holder should consult his, her or its own tax advisor regarding the risks associated with the acquisition of a Unit pursuant to this offering (including potential alternative characterizations). The balance of this discussion generally assumes that the characterization described above is respected for U.S. federal income tax purposes.
Allocation of Purchase Price Between Share of Common Stock and Accompanying Warrant to Purchase Our Common Stock
For U.S. federal income tax purposes, each Unit and Pre-Funded Unit should be treated as an “investment unit” consisting of one share of common stock or one Pre-Funded Warrant, as applicable, and a Warrant to acquire one share of our common stock. The purchase price for each investment unit will be allocated between these two components in proportion to their relative fair market values at the time the unit is purchased by the holder. This allocation of the purchase price for each investment unit will establish the holder’s initial tax basis for U.S. federal income tax purposes in the share of common stock or Pre-Funded Warrant, as applicable, and the Warrant included in each investment unit. The separation of the common stock or Pre-Funded Warrant, as applicable, and the Warrant included in each investment unit should not be a taxable event for U.S. federal income tax purposes. Each holder should consult his, her or its own tax advisor regarding the allocation of the purchase price for an investment unit.
| 133 |
| Table of Contents |
U.S. Holders
Exercise or Expiration of Warrants
In general, a U.S. holder will not recognize gain or loss for U.S. federal income tax purposes upon exercise of a Warrant, except to the extent the U.S. holder receives a cash payment for any fractional share of common stock that would otherwise have been issuable upon exercise of the Warrant, which will be treated as a sale subject to the rules described under “- Disposition of Our Common Stock, Pre-Funded Warrants or Warrants” below. The U.S. holder will take a tax basis in the shares acquired on the exercise of a Warrant equal to the exercise price of the Warrant, increased by the U.S. holder’s adjusted tax basis in the Warrant exercised (as determined pursuant to the rules discussed above) and decreased by the adjusted tax basis allocable to any fractional share that would otherwise have been issuable upon exercise of the Warrant. The U.S. holder’s holding period in the shares of our common stock acquired on exercise of the Warrant will begin on the date of exercise of the Warrant, and will not include any period for which the U.S. holder held the Warrant.
In certain limited circumstances, a U.S. holder may be permitted to undertake a cashless exercise of Warrants into our common stock. The U.S. federal income tax treatment of a cashless exercise of Warrants into our common stock is unclear, and the tax consequences of a cashless exercise could differ from the consequences upon the exercise of a Warrant described in the preceding paragraph. U.S. holders should consult their own tax advisors regarding the U.S. federal income tax consequences of a cashless exercise of Warrants.
The lapse or expiration of a Warrant will be treated as if the U.S. holder sold or exchanged the Warrant and recognized a capital loss equal to the U.S. holder’s tax basis in the Warrant. The deductibility of capital losses is subject to limitations.
Certain Adjustments to Warrants
Under Section 305 of the Code, an adjustment to the number of shares of common stock issued on the exercise of the Warrants, or an adjustment to the exercise price of the Warrants, may be treated as a constructive distribution to a U.S. holder of the Warrants if, and to the extent that, such adjustment has the effect of increasing such U.S. holder’s proportionate interest in our “earnings and profits” or assets, depending on the circumstances of such adjustment (for example, if such adjustment is to compensate for a distribution of cash or other property to our shareholders). An adjustment made pursuant to a bona fide reasonable adjustment formula that has the effect of preventing dilution should generally not be considered to result in a constructive distribution. Any such constructive distribution would be taxable whether or not there is an actual distribution of cash or other property to the holders of Warrants. In certain circumstances, if we were to make a distribution in cash or other property with respect to our common stock after the issuance of the Warrants, then we may make a corresponding distribution to a Warrant holder. The taxation of a distribution received with respect to a Warrant is unclear. It is possible such a distribution would be treated as a distribution (or constructive distribution), although other treatments are possible. For more information regarding the tax considerations related to distributions, see the discussion below regarding “Distributions.” U.S. holders should consult their tax advisors regarding the proper treatment of any adjustments to the Warrants and any distributions with respect to the Warrants.
Distributions
As described in the section entitled “Dividend Policy,” we do not anticipate declaring or paying dividends to holders of our common stock in the foreseeable future. However, if we do make distributions on our common stock or Pre-Funded Warrants to a U.S. holder, such distributions of cash or property generally will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. Distributions in excess of our current and accumulated earnings and profits will constitute a return of capital that is applied against and reduces, but not below zero, a U.S. holder’s adjusted tax basis in our common stock or Pre-Funded Warrant, as applicable. Any remaining excess will be treated as gain realized on the sale or exchange of our common stock or Pre-Funded Warrant as described below under the section titled “-Disposition of Our Common Stock, Pre-Funded Warrants or Warrants.”
| 134 |
| Table of Contents |
Disposition of Our Common Stock, Pre-Funded Warrants or Warrants
Upon a sale or other taxable disposition of our common stock, Pre-Funded Warrants or Warrants, a U.S. holder generally will recognize capital gain or loss in an amount equal to the difference between the amount realized and the U.S. holder’s adjusted tax basis in the common stock, Pre-Funded Warrants or Warrants. Capital gain or loss will constitute long-term capital gain or loss if the U.S. holder’s holding period for the common stock, Pre-Funded Warrants or Warrants exceeds one year. The deductibility of capital losses is subject to certain limitations. U.S. holders who recognize losses with respect to a disposition of our common stock, Pre-Funded Warrants or Warrants should consult their own tax advisors regarding the tax treatment of such losses.
Information Reporting and Backup Reporting
Information reporting requirements generally will apply to payments of dividends (including constructive dividends) on the common stock, Pre-Funded Warrants and Warrants and to the proceeds of a sale or other disposition of common stock, Pre-Funded Warrants and Warrants paid by us to a U.S. holder unless such U.S. holder is an exempt recipient, such as a corporation. Backup withholding will apply to those payments if the U.S. holder fails to provide the holder’s taxpayer identification number, or certification of exempt status, or if the holder otherwise fails to comply with applicable requirements to establish an exemption.
Backup withholding is not an additional tax. Rather, any amounts withheld under the backup withholding rules will be allowed as a refund or a credit against the U.S. holder’s U.S. federal income tax liability provided the required information is timely furnished to the IRS. U.S. holders should consult their own tax advisors regarding their qualification for exemption from information reporting and backup withholding and the procedure for obtaining such exemption.
Non-U.S. Holders
Exercise and Expiration of Warrants
In general, a non-U.S. holder will not recognize gain or loss for U.S. federal income tax purposes upon the exercise of Warrants into shares of common stock, except to the extent the non-U.S. holder receives a cash payment for any fractional share of common stock that would otherwise have been issuable upon exercise of the Warrant, which will be treated as a sale subject to the rules described under “-Disposition of Our Common Stock, Pre-Funded Warrants or Warrants” below. The U.S. federal income tax treatment of a cashless exercise of Warrants into our common stock is unclear. A non-U.S. holder should consult his, her, or its own tax advisor regarding the U.S. federal income tax consequences of a cashless exercise of Warrants.
The expiration of a Warrant will be treated as if the non-U.S. holder sold or exchanged the Warrant and recognized a capital loss equal to the non-U.S. holder’s tax basis in the Warrant. However, a non-U.S. holder will not be able to utilize a loss recognized upon expiration of a Warrant against the non-U.S. holder’s U.S. federal income tax liability unless the loss is effectively connected with the non-U.S. holder’s conduct of a trade or business within the United States (and, if an income tax treaty applies, is attributable to a permanent establishment or fixed base in the United States) or is treated as a U.S.-source loss and the non-U.S. holder is present 183 days or more in the taxable year of disposition and certain other conditions are met.
Certain Adjustments to Warrants
As described under “- U.S. Holders - Certain Adjustments to Warrants,” an adjustment to the Warrants could result in a constructive distribution to a non-U.S. holder, which would be treated as described under “Distributions” below. Any resulting withholding tax attributable to deemed dividends would be collected from other amounts payable or distributable to the non-U.S. holder. Non-U.S. holders should consult their tax advisors regarding the proper treatment of any adjustments to and distributions on the Warrants.
Distributions
As discussed above, we do not anticipate declaring or paying dividends in the foreseeable future. However, if we do make distributions on our common stock or Pre-Funded Warrants, such distributions of cash or property generally will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. Amounts not treated as dividends for U.S. federal income tax purposes will constitute a return of capital and first be applied against and reduce a non-U.S. holder’s adjusted tax basis in its common stock or Pre-Funded Warrants, but not below zero. Any excess will be treated as capital gain and will be treated as described below in the section relating to the sale or disposition of our common stock, Pre-Funded Warrants or Warrants. Because we may not know the extent to which a distribution is a dividend for U.S. federal income tax purposes at the time it is made, for purposes of the withholding rules discussed below we or the applicable withholding agent may treat the entire distribution as a dividend.
| 135 |
| Table of Contents |
Subject to the discussion below on backup withholding and the Foreign Account Tax Compliance Act, or FACTA,, dividends paid to a non-U.S. holder of our common stock or Pre-Funded Warrants that are not effectively connected with the non-U.S. holder’s conduct of a trade or business within the United States will be subject to U.S. federal withholding tax at a rate of 30% of the gross amount of the dividends (or such lower rate specified by an applicable income tax treaty). In order to receive a reduced treaty rate, you must provide us with an IRS Form W-8BEN, IRS Form W-8BEN-E or other appropriate version of IRS Form W-8 certifying qualification for the reduced rate.
If dividends paid to a non-U.S. holder are effectively connected with the non-U.S. holder’s conduct of a trade or business within the United States (and, if required by an applicable income tax treaty, the non-U.S. holder maintains a permanent establishment in the United States to which such dividends are attributable), then, although exempt from U.S. federal withholding tax (provided the non-U.S. holder provides appropriate certification, as described below), the non-U.S. holder will be subject to U.S. federal income tax on such dividends on a net income basis at the regular graduated rates. In order to obtain this exemption, you must provide us with an IRS Form W-8ECI or other applicable IRS Form W-8 properly certifying such exemption. In addition, a non-U.S. holder that is a corporation may be subject to a branch profits tax at a rate of 30% (or such lower rate specified by an applicable income tax treaty) on its effectively connected earnings and profits for the taxable year that are attributable to such dividends, as adjusted for certain items. Non-U.S. holders should consult their tax advisors regarding their entitlement to benefits under any applicable income tax treaty and regarding any applicable treaties that may provide for different rules.
If you hold our common stock, Pre-Funded Warrants or Warrants through a financial institution or other agent acting on your behalf, you will be required to provide appropriate documentation to the agent, which then will be required to provide certification to us or our paying agent, either directly or through other intermediaries. You may be eligible to obtain a refund of any excess amounts withheld by timely filing an appropriate claim for refund with the IRS.
Disposition of Our Common Stock, Pre-Funded Warrants or Warrants
In general, subject to the discussions below on backup withholding, information reporting and foreign accounts, a non-U.S. holder will not be subject to U.S. federal income tax on any gain realized upon the sale or other taxable disposition of our common stock, Pre-Funded Warrants or Warrants unless:
| · | the gain is effectively connected with the non-U.S. holder’s conduct of a trade or business within the United States (and, if required by an applicable income tax treaty, the non-U.S. holder maintains a permanent establishment or fixed base in the United States to which such gain is attributable); |
|
|
|
| · | the non-U.S. holder is a nonresident alien individual present in the United States for 183 days or more during the taxable year of the disposition and certain other requirements are met; or |
|
|
|
| · | our common stock or Pre-Funded Warrants constitutes U.S. real property interests, or USRPIs, by reason of our status as a U.S. real property holding corporation, or USRPHC, for U.S. federal income tax purposes at any time within the shorter of the five-year period preceding the non-U.S. holder’s disposition of, or their holding period for, our common stock, Pre-Funded Warrants or Warrants. |
Gain described in the first bullet point above will generally be subject to U.S. federal income tax on a net income basis at the regular rates. A non-U.S. holder that is a corporation also may be subject to a branch profits tax at a rate of 30% (or such lower rate specified by an applicable income tax treaty) on such effectively connected gain, as adjusted for certain items.
| 136 |
| Table of Contents |
A non-U.S. holder described in the second bullet point above will be subject to U.S. federal income tax at a rate of 30% (or such lower rate specified by an applicable income tax treaty) on any gain derived from the disposition, which may be offset by certain U.S. source capital losses of the non-U.S. holder (even though the individual non-U.S. holder is not considered a resident of the United States) provided the non-U.S. holder has timely filed U.S. federal income tax returns with respect to such losses.
With respect to the third bullet point above, we believe we are not currently and do not anticipate becoming a USRPHC. However, because the determination of whether we are a USRPHC depends on the fair market value of our USRPIs relative to the fair market value of our non-U.S. real property interests and our other business assets, there can be no assurance we will not become a USRPHC in the future. Even if we are determined to be or were to become a USRPHC, gain arising from the sale or other taxable disposition by a non-U.S. holder of our common stock, Pre-Funded Warrants or Warrants will not be subject to U.S. federal income tax if our common stock is “regularly traded,” as defined by applicable Treasury Regulations, on an established securities market, and such non-U.S. holder owned, actually and constructively, 5% or less of our common stock throughout the shorter of the five-year period ending on the date of the sale or other taxable disposition or the non-U.S. holder’s holding period. Special rules may apply to the determination of the 5% threshold in the case of a holder of a Pre-Funded Warrant or Warrant. Non-U.S. holders are urged to consult their own tax advisors regarding the effect of holding our Pre-Funded Warrants or Warrants on the calculation of such 5% threshold. If we are a USRPHC and either our common stock is not regularly traded on an established securities market or a non-U.S. holder holds, or is treated as holding, more than 5% of our outstanding common stock, directly or indirectly, during the applicable testing period, such non-U.S. holder’s gain on the disposition of shares of our common stock, Pre-Funded or Warrants generally will be taxed in the same manner as gain that is effectively connected with the conduct of a U.S. trade or business, except that the branch profits tax generally will not apply. If we are a USRPHC and our common stock is not regularly traded on an established securities market, a non-U.S. holder’s proceeds received on the disposition of shares will also generally be subject to withholding at a rate of 15%. No assurance can be provided that our common stock will be regularly traded on an established securities market for purposes of the rules described above. Prospective investors are encouraged to consult their tax advisors regarding the possible consequences to them if we are, or were to become, a USRPHC.
Non-U.S. holders should consult their tax advisors regarding potentially applicable income tax treaties that may provide for different rules.
Information Reporting and Backup Withholding
Generally, we must report annually to the IRS the amount of distributions (including constructive distributions) on our common stock, Pre-Funded Warrants or Warrants paid to each non-U.S. holder, their name and address, and the amount of tax withheld, if any. Copies of information returns that are filed with the IRS may also be made available under the provisions of an applicable treaty or agreement to the tax authorities of the country in which the non-U.S. holder resides or is established.
Payments of dividends (including constructive dividends) or of proceeds on the disposition of our common stock, Pre-Funded Warrants or Warrants made to a non-U.S. holder may be subject to information reporting and backup withholding at a current rate of 24% unless the non-U.S. holder establishes an exemption, for example, by properly certifying their non-U.S. status on an IRS Form W-8BEN, IRS Form W-8BEN-E or another appropriate version of IRS Form W-8. Notwithstanding the foregoing, backup withholding and information reporting may apply if either we or our paying agent has actual knowledge, or reason to know, that a holder is a U.S. person.
Under current U.S. federal income tax law, U.S. information reporting and backup withholding requirements generally will apply to the proceeds of a disposition of our common stock, Pre-Funded Warrants or Warrants effected by or through a U.S. office of any broker, U.S. or foreign, except that information reporting and such requirements may be avoided if the holder provides a properly executed and appropriate IRS Form W-8 or otherwise meets documentary evidence requirements for establishing non- U.S. holder status or otherwise establishes an exemption. Generally, U.S. information reporting and backup withholding requirements will not apply to a payment of disposition proceeds to a non-U.S. holder where the transaction is effected outside the U.S. through a non-U.S. office of a non-U.S. broker. Information reporting and backup withholding requirements may, however, apply to a payment of disposition proceeds if the broker has actual knowledge, or reason to know, that the holder is, in fact, a U.S. person. For information reporting purposes, certain brokers with substantial U.S. ownership or operations will generally be treated in a manner similar to U.S. brokers.
Non-U.S. holders should consult their tax advisors regarding the application of the information reporting and backup withholding rules to them.
| 137 |
| Table of Contents |
Foreign Account Tax Compliance Act
The Foreign Account Tax Compliance Act and the rules and regulations promulgated thereunder, collectively FATCA, generally impose withholding tax at a rate of 30% on dividends (including constructive dividends) on our common stock, Pre-Funded Warrants or Warrants, and certain other withholding payments, if paid to a non-U.S. entity unless (i) if the non-U.S. entity is a “foreign financial institution,” the non-U.S. entity undertakes certain due diligence, reporting, withholding, and certification obligations, (ii) if the non-U.S. entity is not a “foreign financial institution,” the non-U.S. entity identifies certain of its U.S. investors, if any, or (iii) the non-U.S. entity is otherwise exempt under FATCA. While withholding under FATCA may apply to payments of gross proceeds from a sale or other disposition of our common stock, Pre-Funded Warrants or Warrants, under proposed U.S. Treasury Regulations withholding on payments of gross proceeds is not required. Although such regulations are not final, applicable withholding agents may rely on the proposed regulations until final regulations are issued.
The preceding discussion of material U.S. federal income tax considerations is for informational purposes only. It is not tax advice. Prospective investors should consult their own tax advisors regarding the particular U.S. federal, state, local and non-U.S. tax consequences of purchasing, holding and disposing of our common stock, Pre-Funded Warrants or Warrants, including the consequences of any proposed changes in applicable laws.
UNDERWRITING
We are offering our Units as described in this prospectus through the underwriters named below. Maxim Group LLC (or Maxim) is acting as the sole representative of the underwriters. We have entered into an underwriting agreement with the underwriters. Subject to the terms and conditions of the underwriting agreement, each of the underwriters has severally agreed to purchase, and we have agreed to sell to the underwriter, the number of Units listed next to its name in the following table.
Underwriters |
| Number of |
| |
Maxim Group LLC |
|
|
| |
|
|
|
|
|
Total |
|
|
| |
The underwriting agreement provides that the underwriter must buy all of the Units if they buy any of them. However, the underwriters are not required to take or pay for the shares of common stock, Pre-Funded Warrants and/or the Warrants covered by the underwriters’ option to purchase additional shares as described below.
Our Units are offered subject to a number of conditions, including:
| · | receipt and acceptance of our shares of common stock, Warrants and Pre-Funded Warrants by the underwriters; and |
|
|
|
| · | the underwriters’ right to reject orders in whole or in part. |
We have been advised by Maxim that the underwriters intend to make a market in our shares of common stock, Warrants and Pre-Funded Warrants but that they are not obligated to do so and may discontinue making a market at any time without notice.
In connection with this offering, certain of the underwriters or securities dealers may distribute prospectuses electronically.
| 138 |
| Table of Contents |
Option to Purchase Additional Shares
We have granted the underwriters an option to buy up to an aggregate of 666,666 additional shares of common stock at a price per share of $ and/or up to an additional 666,666 Warrants to purchase up to 666,666 shares of common stock at a price per Warrant of $0.01, less, in each case, discounts and commissions, to cover over-allotments, if any. The underwriters have 45 days from the date of this prospectus to exercise this option. If the underwriters exercise this option, they will each purchase additional shares of common stock and/or Warrants approximately in proportion to the amounts specified in the table above.
Underwriting Discount
Units sold by the underwriters to the public will initially be offered at the offering price set forth on the cover of this prospectus, except that Units sold with Pre-Funded Warrants will be priced at the offering price minus $0.0001. Any Units sold by the underwriter to securities dealers may be sold at a discount of up to $ per share from the public offering price. The underwriter may offer the Units through one or more of their affiliates or selling agents. If all the Units are not sold at the public offering price, Maxim may change the offering price and the other selling terms. Upon execution of the underwriting agreement, the underwriters will be obligated to purchase the Units at the prices and upon the terms stated therein.
The underwriting discount is equal to the public offering price per Unit, less the amount paid by the underwriter to us per Unit. The underwriting discount was determined through an arms’ length negotiation between us and the underwriters. We have agreed to sell the Units of common stock to the underwriters at the offering price of $ per share of common stock, and $0.01 per Warrant, which represents the public offering price of our Units set forth on the cover page of this prospectus less a 7.0% underwriting discount.
The following table shows the public offering price, underwriting discount and proceeds, before expenses, to us. The information assumes either no exercise or full exercise by the underwriters of their over-allotment option.
|
| No |
|
| Full |
| ||
Per Unit Public Offering Price (1) |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Underwriting Discounts and Commissions (2) |
|
|
|
|
|
|
|
|
Total |
| $ |
|
| $ |
| ||
____________
| (1) | Units sold with Pre-Funded Warrants will be priced at the offering price minus $0.0001. |
|
|
|
| (2) | See “Underwriting” for additional disclosure regarding underwriting discounts and commissions and reimbursement of expenses. |
We have agreed to pay Maxim’s out-of-pocket accountable expenses, including Maxim’s legal fees, up to a maximum amount of $75,000, irrespective of whether the offering is consummated.
We estimate that the total expenses of the offering payable by us, not including the underwriting discount, will be approximately $320,000.
Right of First Refusal
In connection with our initial public offering, we granted to Maxim, for the fifteen (15) month period following the commencement of sales of that offering, a right of first refusal to act as sole lead manager, underwriter and/or placement agent for any and all future public or private equity, equity-linked, convertible and debt offerings (excluding commercial bank debt) during such fifteen (15) month period by us, or any successor to or any subsidiary of our company subject to such procedures as agreed upon in the underwriting agreement entered into in connection with our initial public offering. The Company also granted to Maxim, upon the commencement of sales of our initial public offering, the right of first refusal to act as lead advisor with respect to transactions between the company and third parties, including, without limitation, any merger, acquisition or sale of stock or assets (in which the company may be the acquiring or the acquired entity), joint venture, strategic alliance or other similar transaction during such fifteen (15) month period. This fifteen month period remains in effect until November 2022.
| 139 |
| Table of Contents |
Lock-Up Agreements
We and our directors and officers shall enter into customary “lock-up” agreements in favor of Maxim pursuant to which such persons and entities shall agree, for a period of 90 days after the effective date of the registration statement related to this offering, that they shall neither offer, issue, sell, contract to sell, encumber, grant any option for the sale of or otherwise dispose of any of our securities without Maxim’s prior written consent, including the issuance of shares of common stock upon the exercise of currently outstanding convertible securities.
Indemnification
We have agreed to indemnify the several underwriters against certain liabilities, including certain liabilities under the Securities Act. If we are unable to provide this indemnification, we have agreed to contribute to payments the underwriters may be required to make in respect of those liabilities.
Other Relationships
Some of the underwriters and their affiliates have engaged in, and may in the future engage in, investment banking and other commercial dealings in the ordinary course of business with us or our affiliates. They have received, or may in the future receive, customary fees and commissions for these transactions.
Stock Exchange
Our common stock and certain of our warrants are listed on Nasdaq under the symbols “DRMA” and “DRMAW,” respectively We do not intend to apply for the listing of the Warrants or the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system, including Nasdaq.
Price Stabilization, Short Positions
In connection with this offering, the underwriters may engage in activities that stabilize, maintain or otherwise affect the price of our securities during and after this offering, including:
| · | stabilizing transactions; |
|
|
|
| · | short sales; |
|
|
|
| · | purchases to cover positions created by short sales; |
|
|
|
| · | imposition of penalty bids; and |
|
|
|
| · | syndicate covering transactions. |
Stabilizing transactions consist of bids or purchases made for the purpose of preventing or retarding a decline in the market price of our shares of common stock while this offering is in progress. Stabilization transactions permit bids to purchase the underlying security so long as the stabilizing bids do not exceed a specified maximum. These transactions may also include making short sales of our shares of common stock, which involve the sale by the underwriters of a greater number of shares of common stock than they are required to purchase in this offering and purchasing shares of common stock on the open market to cover short positions created by short sales. Short sales may be “covered short sales,” which are short positions in an amount not greater than the underwriters’ option to purchase additional shares referred to above, or may be “naked short sales,” which are short positions in excess of that amount.
| 140 |
| Table of Contents |
The underwriters may close out any covered short position by either exercising their option, in whole or in part, or by purchasing shares in the open market. In making this determination, the underwriters will consider, among other things, the price of shares available for purchase in the open market as compared to the price at which they may purchase shares through the over-allotment option.
Naked short sales are short sales made in excess of the over-allotment option. The underwriters must close out any naked short position by purchasing shares in the open market. A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of the shares of common stock in the open market that could adversely affect investors who purchased in this offering.
The underwriters also may impose a penalty bid. This occurs when a particular underwriter repays to the underwriters a portion of the underwriting discount received by it because Maxim has repurchased shares sold by or for the account of that underwriter in stabilizing or short covering transactions.
These stabilizing transactions, short sales, purchases to cover positions created by short sales, the imposition of penalty bids and syndicate covering transactions may have the effect of raising or maintaining the market price of our common stock or preventing or retarding a decline in the market price of our common stock. As a result of these activities, the price of our common stock may be higher than the price that otherwise might exist in the open market. The underwriters may carry out these transactions on the NASDAQ Capital Market, in the over-the-counter market or otherwise. Neither we nor the underwriters make any representation or prediction as to the effect that the transactions described above may have on the price of the shares. Neither we, nor any of the underwriters make any representation that the underwriters will engage in these stabilization transactions or that any transaction, once commenced, will not be discontinued without notice.
Affiliations
The underwriters and their respective affiliates are full service financial institutions engaged in various activities, which may include securities trading, commercial and investment banking, financial advisory, investment management, investment research, principal investment, hedging, financing and brokerage activities. The underwriters and their affiliates may from time to time in the future engage with us and perform services for us or in the ordinary course of their business for which they will receive customary fees and expenses. In the ordinary course of their various business activities, the underwriters and their respective affiliates may make or hold a broad array of investments and actively trade debt and equity securities (or related derivative securities) and financial instruments (including bank loans) for their own account and for the accounts of their customers, and such investment and securities activities may involve securities and/or instruments of us. The underwriters and their respective affiliates may also make investment recommendations and/or publish or express independent research views in respect of these securities or instruments and may at any time hold, or recommend to clients that they acquire, long and/or short positions in these securities and instruments.
Electronic Distribution
A prospectus in electronic format may be made available on the Internet sites or through other online services maintained by one or more of the underwriters participating in this offering, or by their affiliates. In those cases, prospective investors may view offering terms online and, depending upon the particular underwriter, prospective investors may be allowed to place orders online. The underwriters may agree with us to allocate a specific number of shares for sale to online brokerage account holders. Any such allocation for online distributions will be made by the underwriters on the same basis as other allocations. Other than the prospectus in electronic format, the information on any underwriter’s website and any information contained in any other website maintained by an underwriter is not part of the prospectus or the registration statement of which this prospectus forms a part, has not been approved and/or endorsed by us or any underwriter in its capacity as underwriter and should not be relied upon by investors.
| 141 |
| Table of Contents |
Selling Restrictions
Canada. The securities may be sold in Canada only to purchasers purchasing, or deemed to be purchasing, as principal that are accredited investors, as defined in National Instrument 45-106 Prospectus Exemptions or subsection 73.3(1) of the Securities Act (Ontario), and are permitted clients, as defined in National Instrument 31 103 Registration Requirements, Exemptions and Ongoing Registrant Obligations. Any resale of the securities must be made in accordance with an exemption from, or in a transaction not subject to, the prospectus requirements of applicable securities laws.
Securities legislation in certain provinces or territories of Canada may provide a purchaser with remedies for rescission or damages if this prospectus supplement (including any amendment thereto) contains a misrepresentation, provided that the remedies for rescission or damages are exercised by the purchaser within the time limit prescribed by the securities legislation of the purchaser’s province or territory. The purchaser should refer to any applicable provisions of the securities legislation of the purchaser’s province or territory for particulars of these rights or consult with a legal advisor.
Pursuant to section 3A.3 of National Instrument 33 105 Underwriting Conflicts (NI 33 105), the underwriters are not required to comply with the disclosure requirements of NI 33-105 regarding underwriters conflicts of interest in connection with this offering.
European Economic Area. In relation to each Member State of the European Economic Area which has implemented the Prospectus Directive (each, a “Relevant Member State”) an offer to the public of any securities may not be made in that Relevant Member State, except that an offer to the public in that Relevant Member State of any securities may be made at any time under the following exemptions under the Prospectus Directive, if they have been implemented in that Relevant Member State:
| · | to any legal entity which is a qualified investor as defined in the Prospectus Directive; |
|
|
|
| · | to fewer than 100 or, if the Relevant Member State has implemented the relevant provision of the 2010 PD Amending Directive, 150, natural or legal persons (other than qualified investors as defined in the Prospectus Directive), as permitted under the Prospectus Directive, subject to obtaining the prior consent of the representatives for any such offer; or |
|
|
|
| · | in any other circumstances falling within Article 3(2) of the Prospectus Directive, provided that no such offer of securities shall result in a requirement for the publication by us or any underwriters of a prospectus pursuant to Article 3 of the Prospectus Directive. |
For the purposes of this provision, the expression an “offer to the public” in relation to any securities in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and any securities to be offered so as to enable an investor to decide to purchase any securities, as the same may be varied in that Member State by any measure implementing the Prospectus Directive in that Member State, the expression “Prospectus Directive” means Directive 2003/71/EC (and amendments thereto, including the 2010 PD Amending Directive, to the extent implemented in the Relevant Member State), and includes any relevant implementing measure in the Relevant Member State, and the expression “2010 PD Amending Directive” means Directive 2010/73/EU.
United Kingdom. Each underwriter has represented and agreed that:
| · | it has only communicated or caused to be communicated and will only communicate or cause to be communicated an invitation or inducement to engage in investment activity (within the meaning of Section 21 of the Financial Services and Markets Act 2000 (the FSMA) received by it in connection with the issue or sale of the securities in circumstances in which Section 21(1) of the FSMA does not apply to us; and |
|
|
|
| · | it has complied and will comply with all applicable provisions of the FSMA with respect to anything done by it in relation to the securities in, from or otherwise involving the United Kingdom. |
| 142 |
| Table of Contents |
Switzerland. The securities may not be publicly offered in Switzerland and will not be listed on the SIX Swiss Exchange (the SIX) or on any other stock exchange or regulated trading facility in Switzerland. This document has been prepared without regard to the disclosure standards for issuance prospectuses under art. 652a or art. 1156 of the Swiss Code of Obligations or the disclosure standards for listing prospectuses under art. 27 ff. of the SIX Listing Rules or the listing rules of any other stock exchange or regulated trading facility in Switzerland. Neither this document nor any other offering or marketing material relating to the securities or the offering may be publicly distributed or otherwise made publicly available in Switzerland.
Neither this document nor any other offering or marketing material relating to the offering, or the securities have been or will be filed with or approved by any Swiss regulatory authority. In particular, this document will not be filed with, and the offer of securities will not be supervised by, the Swiss Financial Market Supervisory Authority FINMA, and the offer of securities has not been and will not be authorized under the Swiss Federal Act on Collective Investment Schemes (CISA). Accordingly, no public distribution, offering or advertising, as defined in CISA, its implementing ordinances and notices, and no distribution to any non-qualified investor, as defined in CISA, its implementing ordinances and notices, shall be undertaken in or from Switzerland, and the investor protection afforded to acquirers of interests in collective investment schemes under CISA does not extend to acquirers of securities.
Australia. No placement document, prospectus, product disclosure statement or other disclosure document has been lodged with the Australian Securities and Investments Commission (ASIC), in relation to the offering.
This prospectus does not constitute a prospectus, product disclosure statement or other disclosure document under the Corporations Act 2001 (the Corporations Act) and does not purport to include the information required for a prospectus, product disclosure statement or other disclosure document under the Corporations Act.
Any offer in Australia of the securities may only be made to persons (the Exempt Investors) who are “sophisticated investors” (within the meaning of section 708(8) of the Corporations Act), “professional investors” (within the meaning of section 708(11) of the Corporations Act) or otherwise pursuant to one or more exemptions contained in section 708 of the Corporations Act so that it is lawful to offer the securities without disclosure to investors under Chapter 6D of the Corporations Act.
The securities applied for by Exempt Investors in Australia must not be offered for sale in Australia in the period of 12 months after the date of allotment under the offering, except in circumstances where disclosure to investors under Chapter 6D of the Corporations Act would not be required pursuant to an exemption under section 708 of the Corporations Act or otherwise or where the offer is pursuant to a disclosure document which complies with Chapter 6D of the Corporations Act. Any person acquiring securities must observe such Australian on-sale restrictions.
This prospectus contains general information only and does not take account of the investment objectives, financial situation or particular needs of any particular person. It does not contain any securities recommendations or financial product advice. Before making an investment decision, investors need to consider whether the information in this prospectus is appropriate to their needs, objectives and circumstances, and, if necessary, seek expert advice on those matters.
Notice to Prospective Investors in the Cayman Islands. No invitation, whether directly or indirectly, may be made to the public in the Cayman Islands to subscribe for our securities.
Taiwan. The securities have not been and will not be registered with the Financial Supervisory Commission of Taiwan pursuant to relevant securities laws and regulations and may not be sold, issued or offered within Taiwan through a public offering or in circumstances which constitutes an offer within the meaning of the Securities and Exchange Act of Taiwan that requires a registration or approval of the Financial Supervisory Commission of Taiwan. No person or entity in Taiwan has been authorized to offer, sell, give advice regarding or otherwise intermediate the offering and sale of the securities in Taiwan.
| 143 |
| Table of Contents |
Notice to Prospective Investors in Hong Kong. The contents of this prospectus have not been reviewed by any regulatory authority in Hong Kong. You are advised to exercise caution in relation to the offer. If you are in any doubt about any of the contents of this prospectus, you should obtain independent professional advice. Please note that (i) our shares may not be offered or sold in Hong Kong, by means of this prospectus or any document other than to “professional investors” within the meaning of Part I of Schedule 1 of the Securities and Futures Ordinance (Cap.571, Laws of Hong Kong) (SFO) and any rules made thereunder, or in other circumstances which do not result in the document being a “prospectus” within the meaning of the Companies Ordinance (Cap.32, Laws of Hong Kong) (CO) or which do not constitute an offer or invitation to the public for the purpose of the CO or the SFO, and (ii) no advertisement, invitation or document relating to our shares may be issued or may be in the possession of any person for the purpose of issue (in each case whether in Hong Kong or elsewhere) which is directed at, or the contents of which are likely to be accessed or read by, the public in Hong Kong (except if permitted to do so under the securities laws of Hong Kong) other than with respect to the shares which are or are intended to be disposed of only to persons outside Hong Kong or only to “professional investors” within the meaning of the SFO and any rules made thereunder.
Notice to Prospective Investors in the People’s Republic of China. This prospectus may not be circulated or distributed in the PRC and the shares may not be offered or sold, and will not offer or sell to any person for re-offering or resale directly or indirectly to any resident of the PRC except pursuant to applicable laws, rules and regulations of the PRC. For the purpose of this paragraph only, the PRC does not include Taiwan and the special administrative regions of Hong Kong and Macau.
Israel. This document does not constitute a prospectus under the Israeli Securities Law, 5728-1968, or the Securities Law, and has not been filed with or approved by the Israel Securities Authority. In the State of Israel, this document is being distributed only to, and is directed only at, and any offer of the shares is directed only at, investors listed in the first addendum, or the Addendum, to the Israeli Securities Law, consisting primarily of joint investment in trust funds, provident funds, insurance companies, banks, portfolio managers, investment advisors, members of the Tel Aviv Stock Exchange, underwriters, venture capital funds, entities with equity in excess of NIS 50 million and “qualified individuals”, each as defined in the Addendum (as it may be amended from time to time), collectively referred to as qualified investors (in each case purchasing for their own account or, where permitted under the Addendum, for the accounts of their clients who are investors listed in the Addendum). Qualified investors will be required to submit written confirmation that they fall within the scope of the Addendum, are aware of the meaning of same and agree to it.
LEGAL MATTERS
The validity of the shares of common stock and Warrants offered by this prospectus will be passed upon for us by Lowenstein Sandler LLP. Certain legal matters relating to the offering will be passed upon for the underwriters by Ellenoff Grossman & Schole LLP.
| 144 |
| Table of Contents |
EXPERTS
Mayer Hoffman McCann P.C., independent registered public accounting firm, has audited our financial statements as of and for the years ended December 31, 2020 and 2019, as set forth in their report (which includes an explanatory paragraph regarding the existence of substantial doubt about the Company’s ability to continue as a going concern). We have included our financial statements in the prospectus and elsewhere in the registration statement in reliance on the report of Mayer Hoffman McCann P.C., given on their authority as experts in accounting and auditing in giving said reports.
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the Securities and Exchange Commission a registration statement on Form S-1 under the Securities Act with respect to the shares of common stock and Warrants offered hereby. This prospectus, which constitutes a part of the registration statement, does not contain all of the information set forth in the registration statement or the exhibits and schedules filed therewith. For further information about us and the common stock and Warrants offered hereby, we refer you to the registration statement and the exhibits and schedules filed thereto. Statements contained in this prospectus regarding the contents of any contract or any other document that is filed as an exhibit to the registration statement are not necessarily complete, and each such statement is qualified in all respects by reference to the full text of such contract or other document filed as an exhibit to the registration statement. We are required to file periodic reports, proxy statements, and other information with the Securities and Exchange Commission pursuant to the Securities Exchange Act of 1934. The Securities and Exchange Commission also maintains an Internet website that contains reports, proxy statements and other information about registrants, like us, that file electronically with the Securities and Exchange Commission. The address of that site is www.sec.gov.We also maintain a website at www.dermatarx.com. Upon completion of this offering, you may access these materials at our website free of charge as soon as reasonably practicable after they are electronically filed with, or furnished to, the SEC. Information contained in, or that can be accessed through, our website is not a part of, and is not incorporated into, this prospectus.
| 145 |
| Table of Contents |
INDEX TO FINANCIAL STATEMENTS
Audited Financial Statements |
| Page No. |
| |
|
| F-2 |
| |
|
| F-3 |
| |
Statements of Operations and Comprehensive Loss for the Years Ended December 31, 2019 and 2020 |
|
| F-4 |
|
Statement of Members’ Equity (Deficit) for the Years Ended December 31, 2019 and 2020 |
|
| F-5 |
|
Statements of Cash Flows for the Years Ended December 31, 2019 and 2020 |
|
| F-6 |
|
|
| F-7 |
| |
Interim Unaudited Financial Statements |
|
|
| |
Balance Sheets as of December 31, 2020 and September 30, 2021 (Unaudited) |
|
| F-18 |
|
Statements of Operations for the Three and Nine Months Ended September 30, 2020 and 2021 (Unaudited) |
|
| F-19 |
|
|
| F-20 |
| |
Statements of Cash Flows for the Three and Nine Months Ended September 30, 2020 and 2021 (Unaudited) |
|
| F-22 |
|
|
| F-23 |
| |
| F-1 |
| Table of Contents |
Report of Independent Registered Public Accounting Firm
To the Board of Directors and
Stockholders of Dermata Therapeutics, Inc.:
Opinion on the Financial Statements
We have audited the accompanying balance sheets of Dermata Therapeutics, Inc. (formerly Dermata Therapeutics, LLC) (“Company”) as of December 31, 2020 and 2019, and the related statements of operations and comprehensive loss, members’ equity (deficit), and cash flows for each of the two years in the period ended December 31, 2020, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2020 and 2019, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2020 in conformity with accounting principles generally accepted in the United States of America.
Going Concern Uncertainty
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the financial statements, the Company has incurred recurring losses and negative cash flows from operations and is dependent on additional financing to fund operations. These conditions raise substantial doubt about its ability to continue as a going concern. Management’s plans regarding these matters are also described in Note 2. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
We have served as the Company’s auditor since 2016.
/s/ Mayer Hoffman McCann P.C.
San Diego, California
April 2, 2021 (except for the effects of the reverse stock split described in Note 3 and the subsequent events described in Note 13, as to which the date is July 9, 2021)
| F-2 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Balance Sheets
|
| December 31, |
| |||||
|
| 2019 |
|
| 2020 |
| ||
|
|
|
|
|
|
| ||
Assets: |
|
|
|
|
|
| ||
Current assets: |
|
|
|
|
|
| ||
Cash |
| $ |
|
| $ | 530,400 |
| |
Prepaid expenses and other current assets |
|
|
|
|
| 75,053 |
| |
|
|
|
|
|
|
|
|
|
Total current assets |
|
|
|
|
| 605,453 |
| |
Fixed assets, net |
|
|
|
|
| - |
| |
|
|
|
|
|
|
|
|
|
Total assets |
| $ |
|
| $ | 605,453 |
| |
|
|
|
|
|
|
|
|
|
Liabilities and Members’ Equity: |
|
|
|
|
|
|
|
|
Liabilities: |
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
Accounts payable |
| $ |
|
| $ | 104,276 |
| |
Accrued and other current liabilities |
|
|
|
|
| 133,477 |
| |
License and settlement liability |
|
|
|
|
|
|
| |
Convertible subordinated promissory notes, net of discount |
|
|
|
|
|
| 1,848,495 |
|
Related party convertible subordinated promissory notes, net of discount |
|
|
|
|
|
| 1,140,984 |
|
Current portion of long-term debt, net of debt discount |
|
|
|
|
| 556,160 |
| |
|
|
|
|
|
|
|
|
|
Total current liabilities |
|
|
|
|
| 3,783,392 |
| |
Long-term debt, net of debt discount |
|
|
|
|
| - |
| |
|
|
|
|
|
|
|
|
|
Total liabilities |
| $ |
|
| $ | 3,783,392 |
| |
|
|
|
|
|
|
|
|
|
Commitments and Contingencies (see Note 11) |
|
|
|
|
|
|
|
|
Members’ Equity (Deficit): |
|
|
|
|
|
|
|
|
Series A Preferred Units, no units issued or outstanding at December 31, 2019 or 2020 |
|
|
|
|
| - |
| |
Series 1 Preferred Units, |
|
|
|
|
| 6,833,877 |
| |
Series 1a Preferred Units, |
|
|
|
|
| 4,380,081 |
| |
Series 1a Preferred Warrant Units, 1,437,978 and |
|
|
|
|
| 723,431 |
| |
Series 1b Preferred Units, |
|
|
|
|
| 4,119,595 |
| |
Series 1c Preferred Units, |
|
|
|
|
| 6,491,592 |
| |
Class A Common Units, |
|
|
|
|
| 10,430 |
| |
Class B Common Units, |
|
|
|
|
| 2,342,853 |
| |
Accumulated deficit |
|
| ( | ) |
|
| (28,079,798 | ) |
|
|
|
|
|
|
|
|
|
Total members’ equity (deficit) |
|
| ( | ) |
|
| (3,177,939 | ) |
|
|
|
|
|
|
|
|
|
Total liabilities and members’ equity (deficit) |
| $ |
|
| $ | 605,453 |
| |
The accompanying notes are an integral part of these financial statements.
| F-3 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Statements of Operations and Comprehensive Loss for the Years Ended
|
| For the years ended |
| |||||
|
| 2019 |
|
| 2020 |
| ||
Costs and expenses: |
|
|
|
|
|
| ||
Research and development expenses |
| $ |
|
| $ |
| ||
General and administrative expenses |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Total operating expenses |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Loss from operations |
|
| ( | ) |
|
| ( | ) |
|
|
|
|
|
|
|
|
|
Other income and expenses: |
|
|
|
|
|
|
|
|
Gain from forgiveness of Payroll Protection Plan loan |
|
|
|
|
| ( | ) | |
Interest expense, net |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Net loss and comprehensive loss |
| $ | ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
Basic and diluted net loss per common unit |
| $ | ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
Weighted-average basic and diluted common units |
|
|
|
|
|
| ||
The accompanying notes are an integral part of these financial statements.
| F-4 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Statements of Members’ Equity (Deficit) for the Years Ended
December 31, 2020 and 2019
|
| Class A Common |
|
| Class B Common |
|
| Series 1 Preferred |
|
| Series 1a Preferred |
|
| Series 1a Warrants |
|
| Series 1b Preferred |
|
| Series 1c Preferred |
|
| Accumulated |
|
|
| ||||||||||||||||||||||||||||||||||||||
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Units |
|
| Units |
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Deficit |
|
| Total |
| ||||||||||||||||
Balance at December 31, 2018 |
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
| - |
|
| $ |
|
| $ | ( | ) |
| $ | ( | ) | |||||||||||||
Series 1c Preferred Units issued, net of issuance costs |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
| |||
Class B Common Units issued, net of forfeitures |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
|
| ( | ) |
Balance at December 31, 2019 |
|
|
|
| $ |
|
|
| 1,761,908 |
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
| $ | (24,843,268 | ) |
| $ | (210,159 | ) | |||||||||||||
Exercise of Series 1a Preferred Warrant Units |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||
Series 1c Preferred Units issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
| |||
Class B Common Units issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| - |
| |
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
|
| ( | ) |
Balance at December 31, 2020 |
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
| $ | ) |
| $ | ) | ||||||||||||||||
The accompanying notes are an integral part of these financial statements.
| F-5 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Statements of Cash Flows
|
| For the years ended |
| |||||
|
| 2019 |
|
| 2020 |
| ||
Cash flows from operating activities: |
|
|
|
|
|
| ||
Net loss |
| $ | ( | ) |
| $ | ( | ) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
|
|
Depreciation and amortization of fixed assets |
|
|
|
|
|
| ||
Amortization of debt discount costs |
|
|
|
|
|
| ||
Series 1c Preferred Units issued in connection with license settlement |
|
|
|
|
|
|
| |
Gain from forgiveness of Payroll Protection Plan loan |
|
|
|
|
|
| ( | ) |
Increase/(decrease) in cash resulting from changes in: |
|
|
|
|
|
|
|
|
Prepaid expenses and other current assets |
|
|
|
|
| ( | ) | |
Other assets |
|
|
|
|
|
|
| |
Accounts payable |
|
|
|
|
| ( | ) | |
Accrued and other current liabilities |
|
|
|
|
| ( | ) | |
License and settlement liability |
|
| ( | ) |
|
| ( | ) |
|
|
|
|
|
|
|
|
|
Total adjustments to reconcile net loss to net cash used in operations |
|
|
|
|
| ( | ) | |
|
|
|
|
|
|
|
|
|
Net cash used in operating activities |
|
| ( | ) |
|
| ( | ) |
|
|
|
|
|
|
|
|
|
Cash flows from financing activities: |
|
|
|
|
|
|
|
|
Costs associated with debt amendment |
|
| ( | ) |
|
|
| |
Principal payments on debt |
|
| ( | ) |
|
| ( | ) |
Proceeds from Payroll Protection Plan loan |
|
|
|
|
|
|
| |
Proceeds from issuance of convertible note |
|
|
|
|
|
| ||
Net proceeds from issuance of convertible subordinated promissory notes |
|
|
|
|
|
|
| |
Net proceeds from issuance of Series 1c Preferred Units |
|
|
|
|
|
| ||
Net proceeds from exercise of Series 1a Preferred Warrant Units |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Net cash provided by financing activities |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Net increase (decrease) in cash |
|
|
|
|
| ( | ) | |
Cash at beginning of year |
|
|
|
|
| 1,991,802 |
| |
|
|
|
|
|
|
|
|
|
Cash at end of year |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Non-cash investing and financing activities: |
|
|
|
|
|
|
|
|
Final loan fee related to debt amendment |
| $ |
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Series 1c Preferred Units issued for convertible debt and interest |
| $ |
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Supplemental disclosures: |
|
|
|
|
|
|
|
|
Cash paid for interest |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Cash paid for taxes |
| $ |
|
| $ |
| ||
The accompanying notes are an integral part of these financial statements.
| F-6 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Notes to Financial Statements
| 1. | The Company and Business Activities |
Dermata Therapeutics, Inc., (the “Company”), was formed as a Delaware limited liability company (“LLC”) under the name Dermata Therapeutics, LLC in December 2014. On March 24, 2021 the Company converted from an LLC to a Delaware C-corporation and changed its name to Dermata Therapeutics, Inc. The Company is a clinical-stage biotechnology company focused on the treatment of medical and aesthetic skin conditions.
| 2. | Liquidity and Going Concern Uncertainty |
Since its inception, the Company has not generated any revenue or commercialized any products. As of December 31, 2020, cash totaled $
Historically, the Company’s principal sources of cash have included proceeds from the issuance of common and preferred equity units and proceeds from the issuance of debt. The Company’s principal uses of cash have included cash used in operations, payments for license rights and payments relating to purchases of property and equipment. The Company expects that the principal uses of cash in the future will be for continuing operations, funding of research and development and general working capital requirements. The Company expects that as research and development expenses continue to grow, it will need to raise additional capital to sustain operations and research and development. The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. The financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from the possible inability of the Company to continue as a going concern.
Management’s Plan to Continue as a Going Concern
In order to continue as a going concern, the Company will need, among other things, additional capital resources. Until the Company can generate significant cash from operations, management’s plans to obtain such resources for the Company include proceeds from offerings of the Company’s equity securities or debt, or transactions involving product development, technology licensing or collaboration. Management can provide no assurance that any sources of a sufficient amount of financing will be available to the Company on favorable terms, if at all. Management is currently in the process of seeking additional equity financing, however management’s current plans do not alleviate substantial doubt about the Company’s ability to continue as a going concern.
| 3. | Summary of Significant Accounting Policies |
Basis of Presentation
The accompanying financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (“GAAP”) as found in the Accounting Standards Codification (“ASC”), the Accounting Standards Update (“ASU”) of the Financial Accounting Standards Board (“FASB”) and the rules and regulations of the U.S. Securities and Exchange Commission (“SEC”).
| F-7 |
| Table of Contents |
Reverse Stock Split
On July 1, 2021, the Company effected a reverse split of shares of the Company’s common stock at a ratio of 1-for-20.5 pursuant to an amendment to the amended and restated certificate of incorporation approved by the Company’s board of directors and stockholders. The par value was not adjusted as a result of the reverse split. All issued and outstanding common stock share and per share amounts contained in the financial statements have been retroactively adjusted to reflect this reverse split for all periods presented, and the conversion ratio of the preferred stock was adjusted accordingly.
Use of Estimates
The preparation of financial statements requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of expenses during the reporting period. On an ongoing basis, management evaluates these estimates and judgments, including those related to useful lives of long-lived assets, accrued research and development expenses and estimated fair values of equity instruments. The Company bases its estimates on various assumptions that it believes are reasonable under the circumstances. Actual results may differ from these estimates under different assumptions or conditions.
Segment Information
Operating segments are defined as components of an enterprise about which separate discrete information is available for evaluation by the chief operating decision maker, or decision-making group, in deciding how to allocate resources and in assessing performance. The Company and the Company’s chief operating decision maker view the Company’s operations and manage its business in one operating segment, which is the business of developing and commercializing pharmaceuticals. The Company operates in only one segment.
Cash
The Company places its cash with reputable financial institutions that are insured by the Federal Deposit Insurance Corporation, or FDIC. At times, deposits held may exceed the amount of insurance provided by the FDIC. The Company has not experienced any losses in its cash and believes they are not exposed to any significant credit risk.
Fair Value Measurement
The Company uses a three-tier fair value hierarchy to prioritize the inputs used in the Company’s fair value measurements. These tiers include: Level 1, defined as observable inputs such as quoted prices in active markets for identical assets; Level 2, defined as inputs other than quoted prices in active markets that are either directly or indirectly observable; and Level 3, defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions. The Company believes the carrying amount of cash, accounts payable, accrued expenses and debt approximate their estimated fair values due to the short-term maturities of these financial instruments.
Fixed Assets
Fixed assets consist of furniture and fixtures and computer equipment. Fixed assets are stated at cost less accumulated depreciation and amortization. Additions, improvements, and major renewals are capitalized. Maintenance, repairs, and minor renewals are expensed as incurred. Depreciation is determined using the straight-line method over the estimated useful lives of the assets, which is primarily three years. Depreciation and amortization expense for the years ended December 31, 2019 and 2020 was $
Patent Costs
Patent costs related to obtaining and maintaining patent protection in both the United States and other countries are expensed as incurred. The amounts expensed in the years ended December 31, 2019 and 2020 were $
| F-8 |
| Table of Contents |
Research and Development
Research and development costs consist of expenses incurred in connection with the development of the Company’s product candidates. Such expenses include expenses incurred under agreements with contract research organizations, manufacturing and supply scale-up expenses and the cost of acquiring and manufacturing preclinical and clinical trial supply, outsourced laboratory services, including materials and supplies used to support the Company’s research and development activities, and payments made for license fees and milestones that have not been demonstrated to have commercial value. Such expenses are expensed as incurred. The amounts expensed in the years ended December 31, 2019 and 2020 were $
Income Taxes
Through March 24, 2021, the Company operated as a limited liability company taxed as a partnership. Therefore, any income tax liability or benefit through that date accrued to the members.
Net Loss Per Common Unit
Basic net loss per unit is calculated by dividing net loss attributable to common unitholders by the weighted-average units outstanding during the period, without consideration of common unit equivalents. Diluted net loss per unit is calculated by adjusting weighted-average shares outstanding for the dilutive effect of common unit equivalents outstanding for the period. For purposes of the diluted net loss per unit calculation, preferred units, profit interests, and warrants to purchase preferred units are considered to be common unit equivalents but are excluded from the calculation of diluted net loss per common unit if their effect would be anti-dilutive.
The common unit equivalents that are not included in the calculation of diluted net loss per common unit but could potentially dilute basic earnings per share in the future are as follows:
|
| December 31, |
| |||||
|
| 2019 |
|
| 2020 |
| ||
Numerator: |
|
|
|
|
|
| ||
Net loss |
| $ | ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
Denominator: |
|
|
|
|
|
|
|
|
Weighted-average basic and diluted common units |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Basic and diluted net loss per common unit |
| $ | ( | ) |
| $ | ( | ) |
|
| December 31, |
| |||||
|
| 2019 |
|
| 2020 |
| ||
Series 1 Preferred 1 Preferred Units |
|
|
|
|
|
| ||
Series 1a Preferred Units |
|
|
|
|
|
| ||
Series 1a Preferred Warrant Units |
|
|
|
|
|
| ||
Series 1b Preferred Units |
|
|
|
|
|
| ||
Series 1c Preferred Units |
|
|
|
|
|
| ||
Class B Common Units Profits Interests |
|
|
|
|
|
| ||
Total potentially dilutive securities |
|
|
|
|
|
| ||
Recently Adopted Accounting Pronouncements
In February 2016, the FASB issued ASU No. 2016-02, Leases (“ASC 842”), which supersedes all existing lease guidance. This guidance offers specific accounting guidance for a lessee, a lessor and sale and leaseback transactions. The new standard requires lessees to recognize an operating lease with a term greater than one year on their balance sheets as a right-of-use asset and corresponding lease liability, measured at the present value of the lease payments. Lessees are required to classify leases as either finance or operating leases. If the lease is effectively a financed-purchase by the lessee, it is classified as a financing lease, otherwise, it is classified as an operating lease. This classification will determine whether lease expense is recognized based on an effective interest method or on a straight-line basis over the term of the lease. ASC 842 provides accounting guidance for transactions that meet specific criteria for a leaseback transaction. If the criteria are not met, the transaction is considered a “failed sale” and the transaction must be accounted for as a financing arrangement. The new standard was effective for the Company as of January 1, 2019. Upon adoption, lessees must apply a modified retrospective transition approach for leases existing at, or entered after, the beginning of the earliest comparative period presented in the financial statements. Adoption of this new guidance did not have an impact on the Company’s financial position and results of operations.
| F-9 |
| Table of Contents |
In July 2017, the FASB issued authoritative guidance changing the classification analysis of certain equity-linked financial instruments (or embedded features) with down round features, whereby a down round feature no longer precludes equity classification when assessing whether the instrument is indexed to an entity’s own stock, and also clarifying existing disclosure requirements for equity-classified instruments. This guidance is effective for fiscal years beginning after December 15, 2019 and interim periods within fiscal years beginning after December 15, 2020. Early adoption is permitted. The Company adopted this guidance with no material impact on its financial statements or disclosures.
Recently Issued Accounting Pronouncements
In December 2019, the FASB issued ASU No. 2019-12, Income Taxes (Topic 740): Simplifying the Accounting for Income Taxes (“ASU 2019-12”). ASU 2019-12 simplifies the accounting for income taxes by removing certain exceptions to the general principles in Topic 740. ASU 2019-12 also improves the consistent application, and the simplification, of other areas of Topic 740 by clarifying and amending existing guidance. ASU 2019-12 is effective for fiscal years beginning after December 15, 2020, and interim periods within those fiscal years, with early adoption permitted. The Company is currently evaluating the potential impact that this standard may have on its financial statements and related disclosures.
In August 2020, the FASB issued ASU 2020-06, Debt - Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging - Contracts in an Entity’s Own Equity (Subtopic 815-40) (“ASU 2020-06”). ASU 2020-06 eliminates the beneficial conversion and cash conversion accounting models for convertible instruments. It also amends the accounting for certain contracts in an entity’s own equity that are currently accounted for as derivatives because of specific settlement provisions. In addition, ASU 2020-06 modifies how particular convertible instruments and certain contracts that may be settled in cash or shares impact the diluted earnings per share computation. The amendments in ASU 2020-06 are effective for smaller reporting companies as defined by the SEC for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years. Early adoption is permitted, but not earlier than fiscal years beginning after December 15, 2020. The Company is currently evaluating the impact of ASU 2020-06 on its financial statements
| 4. | Balance Sheet Details |
The following provides certain balance sheet details:
|
| December 31, |
| |||||
|
| 2019 |
|
| 2020 |
| ||
Prepaid Expenses: |
|
|
|
|
|
| ||
Prepaid insurance |
| $ |
|
| $ | 68,003 |
| |
Prepaid clinical trial expense |
|
|
|
|
| 7,050 |
| |
|
|
|
|
|
|
|
|
|
Total prepaid expenses |
| $ |
|
| $ | 75,053 |
| |
|
|
|
|
|
|
|
|
|
Fixed Assets: |
|
|
|
|
|
|
|
|
Furniture and office equipment |
| $ |
|
| $ | 59,382 |
| |
Computer equipment |
|
|
|
|
| 17,225 |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 76,607 |
| |
Less: accumulated depreciation and amortization |
|
| ( | ) |
|
| (76,607 | ) |
|
|
|
|
|
|
|
|
|
Total fixed assets, net |
| $ |
|
| $ | - |
| |
|
| December 31, |
| |||||
|
| 2019 |
|
| 2020 |
| ||
Accrued and Other Current Liabilities: |
|
|
|
|
|
| ||
Accrued payroll |
| $ |
|
| $ |
| ||
Accrued interest payable |
|
|
|
|
| 49,169 |
| |
Accrued employee benefits |
|
|
|
|
|
| ||
Accrued vacation |
|
|
|
|
| 84,308 |
| |
|
|
|
|
|
|
|
|
|
Total accrued and other current liabilities |
| $ |
|
| $ | 133,477 |
| |
| F-10 |
| Table of Contents |
| 5. | Subordinated Convertible Promissory Notes |
In July and October, 2020, the Company issued an aggregate of $3,000,000 of subordinated convertible promissory notes (“The Notes”). Notes in the amount of $
| 6. | Long-Term Debt |
In February 2017, the Company entered into a Loan and Security Agreement with Silicon Valley Bank (SVB) whereas SVB agreed to provide term loans to the Company in two tranches. The first tranche of $2,500,000 was drawn in February 2017 and bears interest at a rate of 1.5% above the prime rate, which was 3.25% as of December 31, 2020, payable monthly through February 9, 2021. The second tranche was not utilized.
In connection with the Loan and Security Agreement, SVB also received warrant units to purchase, at any time after February 9, 2017 and prior to February 9, 2027, 187,978 Series 1a Preferred Units or the equivalent Series A Preferred Units had they purchased Series 1a Preferred Units, if Series A Preferred Units are issued, at a price of $1.00 per unit. The estimated fair value of these warrant units of $104,630 (See Note 8), as well as costs associated with the term loan, including provision for a final payment of $225,000, was recorded as a discount to outstanding debt and is being amortized to interest expense utilizing the effective interest method over the underlying term of the loan.
In June 2019, the Company and SVB entered into a First Amendment to the Loan and Security Agreement whereby if the Company did not achieve certain capital milestones by December 1, 2019, Term Loan principal payments would be deferred from December 21, 2019 through May 1, 2020 with the deferred principal payments being payable in equal monthly installments, in addition to those principal payments already scheduled to be paid, starting on June 1, 2020 and extending through the February 9, 2021 maturity date of the Term Loan. In addition, if those principal payments were deferred for that six-month period, a non-refundable amendment fee of $100,000 would be due and payable on the earliest to occur of the Maturity Date, the prepayment of the Term Loan or the occurrence of an Event of Default. The capital milestones were not achieved by December 1, 2019 and, therefore, the defined principal repayments were deferred.
The non-refundable amendment fee of $100,000, as well as $12,280 of costs associated with the amendment, have been recorded as a discount to outstanding debt and are being amortized to interest expense utilizing the effective interest method over the remaining underlying term of the loan.
Pursuant to the Loan and Security Agreement, the Company is bound by certain affirmative and negative covenants setting forth actions that must and must not take place during the term thereof. Upon the occurrence of an event of default under the Loan and Security Agreement, subject to cure periods for certain events of default, all amounts owed by the Company thereunder shall begin to bear interest at a rate of 5.0% above the rate that is otherwise applicable thereto and may be declared immediately due and payable by SVB. As of December 31, 2020, the Company was in compliance with all covenants. In addition, in accordance with the terms of the First Amendment, SVB was granted a lien on all of the Company’s intellectual property rights.
Future principal payments of $
| F-11 |
| Table of Contents |
Payroll Protection Program
On April 22, 2020, the Company received proceeds of a $133,592 loan from SVB under provisions of the Small Business Administration Payroll Protection Program (PPP). This loan was forgiven in December 2020 under provisions of the PPP and the gain is included in other income on the statement of operations and comprehensive loss.
| 7. | Equity Securities |
Series 1 Preferred Units
From the Company’s formation on December 8, 2014 through 2016, the Company issued 6,906,244 Series 1 Preferred Units for net consideration of $6,833,877.
Series 1a Preferred Units
In 2016, the Company issued 5,000,000 Series 1a Preferred Units in exchange for cash of $5,000,000 and net of issuance costs of $19,868. Purchasers of the Series 1a Preferred Units also received 1,250,000 Warrant Units to purchase an additional amount of Series 1a Preferred Units (see Note 8 - Series 1a Warrants Outstanding). The estimated fair value of the warrant units has been recorded as a separate component of members’ equity (deficit) in the accompanying balance sheet with an offset to the Series 1a proceeds.
Series 1b Preferred Units
In 2018, the Company issued 6,500,000 Series 1b Preferred Units in exchange for cash of $6,500,000 and net of issuance costs of $40,405. Purchasers of the Series 1b Preferred Units also received 26,000,000 Class B Common Units, not representing a profits interest. The estimated fair value of the Class B Common units has been recorded as a component of members’ equity (deficit) in the accompanying balance sheet with an offset to the Series 1b proceeds.
Series 1c Preferred Units
On June 14, 2019, the Company closed participation in a $5,785,000 Series 1c financing from current and new investors. As of December 31, 2019, cash of $5,535,000, including $150,000 from the conversion of a convertible note issued to a Managing Member of the Company for a loan made to the Company, net of issuance costs of $25,857 had been received. The interest on the convertible note in the amount of $1,487 was also converted into Series 1c Preferred units. The remaining balance of $250,000 committed to the financing was paid in 2020. In addition, in June 2019 5,221,156 Series 1c Preferred units were issued in connection with the settlement and license agreement described in Note 11 - Commitments and Contingencies.
Class A Common Units
During 2014 and 2015, the Company issued, in exchange for consideration of $
| F-12 |
| Table of Contents |
Class B Common Units
The Company has
The remaining
During 2019 and 2020, the Company issued
Liquidation Preference
So long as there are no Series A Preferred Units outstanding at the time of a liquidity event, any liquidity event proceeds will be distributed as follows: first, proceeds to Series 1c Preferred Unit holders sufficient to cover two times their Series 1c investment; second, proceeds to Series 1, Series 1a, Series 1b and Series 1c Preferred Unit holders sufficient to cover interest at the rate of 8% per annum on the Series 1 Preferred Units, the Series 1a Preferred Units, the Series 1b Preferred Units and the Series 1c Preferred Units; third, proceeds to the Series 1, Series 1a, Series 1b and Series 1c Preferred Unit holders sufficient to cover the unit value of Series 1 Preferred Units, Series 1a Preferred Units, Series 1b Preferred Units and Series 1c Preferred Units; fourth, to Class A and Class B Common holders proceeds sufficient to cover their pro-rata portion of distributions made to Series 1, Series 1a, Series 1b and Series 1c Preferred Unit holders, provided that no Class B Common Units will share in any distribution until after the point at which the amount per Class A Common Unit exceeds the amount of such Class B Common Unit’s Participation Threshold; and fifth, a pro-rata distribution of the remaining proceeds to all equity holders. Upon the issuance of Series A Preferred Units, each Series 1 Preferred Unit, each Series 1a Preferred Unit, each Series 1b Preferred Unit and each Series 1c Preferred Unit will automatically convert into the number of Series A Preferred Units equal to the sum of the unit value of the Series 1, Series 1a, Series 1b or Series 1c Preferred Units plus all accumulated preferred return as of the conversion date that would be due with respect to such Series 1, Series 1a, Series 1b or Series 1c Preferred Units in the case of a liquidity event. As of December 31, 2020, no Series A Preferred Units have been issued.
Conversion Rights
Upon the first issuance by the Company of any Series A Preferred Units, each Series 1 Preferred Unit and each Series 1a Preferred Unit and each Series 1b Preferred Unit and each Series 1c Preferred Unit shall automatically be converted into the number of Series A Preferred Units equal to the sum of the Unit Value with respect to such Series 1 Preferred Unit or Series 1a Preferred Unit or Series 1b Preferred Unit or Series 1c Preferred Unit as of the conversion date divided by the product of 0.80 multiplied by the Unit Value of the Series 1 Preferred Units or Series 1a Preferred Units or Series 1b Preferred Units or Series 1c Preferred Units issued on the conversion date. The Series A Preferred Units issued to the Series 1 Preferred Members and Series 1a Preferred Members and Series 1b Preferred Members and Series 1c Preferred Members upon conversion of such Series 1 Preferred Units and Series 1a Preferred Units and such Series 1b Preferred Units and Series 1c Preferred Units shall have the same rights, privileges and preferences as the other Series A Preferred Units issued by the Company on the conversion date.
The Company considered the classification of the Preferred Units and concluded that they were appropriately included as a component of equity since each class of Preferred Units participates in the same form of consideration received upon a change in control.
| F-13 |
| Table of Contents |
| 8. | Series 1a Warrants Outstanding |
In connection with the issuance of
In connection with the Loan and Security Agreement, SVB also received Warrant Units to purchase, at any time after February 9, 2017 and prior to February 9, 2027,
| 9. | 401(k) Plan |
The Company sponsors a 401(k) savings plan for all eligible employees. The Company may make discretionary matching contributions to the plan to be allocated to employee accounts based upon employee deferrals and compensation. To date, the Company has not made any matching contributions into the savings plan.
| 10. | License Agreements |
On March 31, 2017, the Company entered into a license agreement, as amended (“The License Agreement”) with Villani, Inc. (“Villani”) whereby Villani has granted the Company an exclusive, sub-licensable, royalty-bearing license (“The License”) under the Licensed Patents (as defined in the License Agreement), to formulate, develop, seek regulatory approval for, make or sell products that contain Spongilla lacustris (alone or in combination with other active or inactive ingredients) for the treatment of diseases, disorders and conditions of the skin, including but not limited to acne, rosacea, psoriasis, atopic dermatitis, seborrheic dermatitis, actinic keratosis and eczema that were developed using certain licensed know-how (“The Licensed Products”). The Company is responsible for the development (including manufacturing, packaging, non-clinical studies, clinical trials and obtaining regulatory approval and commercialization (including marketing, promotion, distribution, etc.)) for all Licensed Products.
In partial consideration of the License, the Company forgave a previous outstanding loan to Villani in the amount of $400,000. The License was amended in 2019 and, in consideration of the receipt of certain know-how and patents, the Company issued to Villani 5,221,156 Series 1c Preferred Units equal to 5% of the Company’s fully diluted capitalization, valued at $730,962. Pursuant to the License Agreement, the Company is required to make future milestone payments to Villani in an aggregate amount of up to $20.25 million upon the achievement of specified development and sales milestones, payable in cash or in equity, at the option of Villani.
Pursuant to the License Agreement, the Company is required to make royalty payments to Villani in amounts equal to a single-digit percentage of net sales of Licensed Products and HMW Combination Products (as defined in the License Agreement), subject to certain adjustments as set forth in the License Agreement. Royalties shall be payable, on a country-by-country and Licensed Product-by Licensed Product basis, for the period of time from the effective date of the License Agreement until the later of (i) the expiration of the last to expire valid claim in such country (which is set to expire in 2023), (ii) the expiration of regulatory exclusivity for such Licensed Product in such country, and (iii) 15 years from the date of the first commercial sale of the Licensed Product in such country.
The License Agreement may be terminated
The License agreement includes customary terms relating to, among others, indemnification, intellectual property protection, confidentiality, remedies and warranties.
| F-14 |
| Table of Contents |
| 11. | Commitments and Contingencies |
Employee Bonuses
In December 2016, the Company approved employee bonuses in the amount of $
Coronavirus Pandemic
On March 11, 2020, the World health Organization declared the outbreak of a coronavirus (COVID-19) pandemic. Significant uncertainties may arise with respect to potential shutdowns of operations or government orders to cease activities due to emergency declarations, inability to operate, or employee shortages, claims for business interruption insurance, etc. Each of these matters may have a significant impact on the future results of the Company.
Legal Proceedings
In 2018, the Company and Villani were involved in a dispute with respect to the payment of certain milestones, as defined in The License Agreement. On June 4, 2019, the Company entered into a License Amendment and Settlement Agreement with Villani, also a related party, regarding Villani’s claim for a license agreement milestone payment of $
The License Amendment and Settlement Agreement also modified The License Agreement entered into in 2017 as described above in Note 10.
In the normal course of business, the Company may be involved in legal proceedings or threatened legal proceedings. The Company is not a party to any legal proceedings or aware of any threatened legal proceedings which are expected to have a material adverse effect on its financial condition, results of operations or liquidity.
| 12. | Related Parties |
During 2019, a Managing Member of the Company loaned the Company through a convertible note $
The Chief Executive Officer of the licensor with whom the Company entered into the settlement and license agreement on June 4, 2019 is a holder of Class B Common units.
During 2020, the Managing Members and other related parties to the Company loaned the Company $
| F-15 |
| Table of Contents |
| 13. | Subsequent Events |
In January and February, 2021, the Company paid the final principal payments of $
On January 27, 2021, the Company amended the terms of The Notes to increase the maximum amount of convertible promissory notes to be issued from $
On March 15, 2021, the Company amended its LLC Agreement to provide for Series 1d Preferred Units at a cost of $
On March 24, 2021, the Company entered into a Plan of Conversion (“Conversion”) whereby the Company converted from a limited liability company (LLC) under the laws of the State of Delaware to a Delaware corporation with the name Dermata Therapeutics, Inc. In connection with the Conversion, each fully-paid Preferred and Common Unit in the LLC was converted into a like number of shares of Preferred and Common Stock of the Company with a par value $
In addition, each warrant to purchase Series 1a Preferred Units in the LLC were automatically converted into a warrant to purchase, upon the same terms and conditions, shares of Series 1a Preferred Stock of the Company and any subordinated convertible promissory notes outstanding at the time of Conversion were automatically converted into a subordinated convertible promissory note of the Company with the same terms and conditions, provided that any right to convert the subordinated convertible promissory note into membership interests of the LLC shall, instead, be a right to convert into equivalent shares of capital stock of the Company. Class B Common Units that were considered a profits interest were converted into Common Stock Options or warrants.
On March 24, 2021, in connection with our conversion from a limited liability company to a Delaware corporation, we issued common stock options exercisable for an aggregate of
In April 2021, the Board of Directors cancelled the employee bonuses (See Note 11).
On June 29, 2021, the Company’s board of directors amended its Certificate of Incorporation to adjust the conversion price and certain conversion mechanics of the Company’s issued and outstanding Series 1d Preferred Stock, whereby each share of Series 1d Preferred stock will convert into such number of Common Stock as determined by dividing (i) the product of (a) the Original Issue Price for the Series 1d Preferred Stock, multiplied by (b) 1.2, rounded to the nearest whole cent, by (ii) the 80% of the initial public per share offering price in the IPO. The Series 1d conversion shall not be subject to further adjustment for any stock split.
On June 29, 2021, the Company’s board of directors approved an amendment to the 2021 Plan to increase the number of shares of Common Stock available for issuances from
| F-16 |
| Table of Contents |
On June 29, 2021, the Company’s board of directors approved a
On July 8, 2021, the Company’s board of directors amended its Certificate of Incorporation to adjust the conversion price and certain conversion mechanics of the Company’s issued and outstanding Series 1d Preferred Stock, whereby each share of Series 1d Preferred Stock is convertible into such number of fully paid and nonassessable shares of Common Stock as is determined by multiplying the Adjusted As Converted Number (as defined below) by the Series 1d IPO Conversion Ratio (as defined below) (the “Series 1d IPO Conversion Number”). For the avoidance of doubt, and notwithstanding the terms set forth in Section 3.4, the Series 1d IPO Conversion Number shall not be subject to further adjustment for any subdivision or combination of the outstanding Common Stock effected in anticipation of the IPO.
(a) | The “Adjusted Conversion Price” means the product of (i) the Original Issue Price for the Series 1d Preferred Stock multiplied by (ii) 1.2, rounded to the nearest whole cent. |
|
|
(b) | The “Adjusted Conversion Ratio” means the quotient of (i) the Original Issue Price for the Series 1d Preferred Stock divided by (ii) the Adjusted Conversion Price. |
|
|
(c) | The “Adjusted As Converted Number” means the product of (i) one share of Series 1d Preferred Stock multiplied by (ii) the Adjusted Conversion Ratio. |
|
|
(d) | The “IPO Discount Ratio” means 80% of the initial public per share offering price in the IPO.” |
|
|
(e) | The “Series 1d IPO Conversion Ratio” means the quotient of (i) the Adjusted Conversion Price divided by (ii) the IPO Discount Price.” |
The Company has completed an evaluation of all subsequent events through July 9, 2021, which is the date the financial statements were available to be issued.
| F-17 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Condensed Balance Sheets
|
| September 30, 2021 |
|
| December 31, 2020 |
| ||
|
| (unaudited) |
|
|
|
| ||
Assets: |
|
|
|
|
|
| ||
Current assets: |
|
|
|
|
|
| ||
Cash |
| $ |
|
| $ |
| ||
Prepaid expenses and other current assets |
|
|
|
|
|
| ||
Total current assets |
|
|
|
|
|
| ||
Fixed assets, net |
|
|
|
|
|
| ||
Total assets |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Liabilities and Stockholders’ and Members’ Equity (Deficit): |
|
|
|
|
|
|
|
|
Liabilities: |
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
Accounts payable |
| $ |
|
| $ |
| ||
Accrued and other current liabilities |
|
|
|
|
|
| ||
Convertible subordinated promissory notes, net of discount |
|
|
|
|
|
| ||
Related party convertible subordinated promissory notes, net of discount |
|
|
|
|
|
| ||
Current portion of long-term debt, net of debt discount |
|
|
|
|
|
| ||
Total current liabilities |
|
|
|
|
|
| ||
Total liabilities |
|
|
|
|
|
| ||
Commitments and Contingencies (see Note 10) |
|
|
|
|
|
|
|
|
Stockholders’ and Members’ Equity (Deficit): |
|
|
|
|
|
|
|
|
Series 1 Preferred Units, |
|
|
|
|
|
| ||
Series 1a Preferred Units, 5,018,750 units authorized, issued and outstanding at December 31, 2020 |
|
|
|
|
|
| ||
Series 1a Preferred Warrant Units, 1,419,228 units issued and outstanding at December 31, 2020 |
|
|
|
|
|
| ||
Series 1b Preferred Units, |
|
|
|
|
|
| ||
Series 1c Preferred Units, |
|
|
|
|
|
| ||
Class A Common Units, |
|
|
|
|
|
| ||
Class B Common Units, 1,767,477 units authorized, issued and outstanding at December 31, 2020 |
|
|
|
|
|
| ||
Common Stock, par value $ |
|
|
|
|
|
| ||
Additional paid-in capital |
|
|
|
|
|
| ||
Accumulated deficit |
|
| ( | ) |
|
| ( | ) |
Total stockholders’ and members’ equity (deficit) |
|
|
|
|
| ( | ) | |
Total liabilities and stockholders’ and members’ equity (deficit) |
| $ |
|
| $ |
| ||
The accompanying notes are an integral part of these financial statements.
| F-18 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Condensed Statements of Operations
Unaudited
|
| For the three months ended September 30, |
|
| For the nine months ended September 30, |
| ||||||||||
|
| 2021 |
|
| 2020 |
|
| 2021 |
|
| 2020 |
| ||||
|
| (unaudited) |
|
| (unaudited) |
|
| (unaudited) |
|
| (unaudited) |
| ||||
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
| ||||
Research and development |
| $ |
|
| $ |
|
| $ |
|
| $ |
| ||||
General and administrative |
|
|
|
|
|
|
|
|
|
|
|
| ||||
Total operating expenses |
|
|
|
|
|
|
|
|
|
|
|
| ||||
Loss from operations |
|
| ( | ) |
|
| ( | ) |
|
| ( | ) |
|
| ( | ) |
Other income and expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense, net |
|
|
|
|
|
|
|
|
|
|
|
| ||||
Net loss |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
Deemed dividend upon the redemption of 5,221,156 shares of Series 1c preferred stock (see Note 6) |
| $ |
|
| $ |
|
| $ |
|
| $ |
| ||||
Deemed dividend upon the amendment of terms of the Series 1d convertible preferred stock (see Note 6) |
| $ |
|
| $ |
|
| $ |
|
| $ |
| ||||
Net loss attributable to common stockholders |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per share of common stock, basic and diluted |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
Weighted-average basic and diluted common units/shares |
|
|
|
|
|
|
|
|
|
|
|
| ||||
The accompanying notes are an integral part of these financial statements.
| F-19 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Statements of Stockholder’s and Member’s Equity (Deficit)
Unaudited
|
| Class A Common |
|
| Class B Common |
|
| Series 1 Preferred |
|
| Series 1a Preferred |
|
| Series 1a Warrants |
|
| Series 1b Preferred |
| ||||||||||||||||||||||||||||||
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
| ||||||||||||
Balance at December 31, 2019 |
|
| 508,777 |
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
| |||||||||||
Class B Common Units issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at March 31, 2020 |
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
| ||||||||||||
Series 1c Preferred Units issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at June 30, 2020 |
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
| ||||||||||||
Exercise of Series 1a Preferred Warrant Units |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 18,750 |
|
| $ |
|
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
| |
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at September 30, 2020 |
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
| 1,419,228 |
|
| $ |
|
|
|
|
| $ |
| |||||||||||
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2020 |
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
| 1,419,228 |
|
| $ |
|
|
|
|
| $ |
| |||||||||||
Series 1d Preferred Units issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Class B Common Units forfeited |
|
|
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Conversion of Common Units to Common Stock |
|
| ( | ) |
| $ | ( | ) |
|
| ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Conversion of Preferred Units to Preferred Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
| $ | ( | ) |
|
| ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
| ( | ) |
| $ | ( | ) |
Conversion of Warrant Units to Preferred Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
Stock-based compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at March 31, 2021 |
|
| - |
|
| $ | - |
|
|
| - |
|
| $ |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
| |
Stock-based compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at June 30, 2021 |
|
| - |
|
| $ |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
| |
Redemption of Series 1c preferred shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Conversion of Preferred Stock to Common Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Conversion of Preferred Stock Warrants to Common Stock Warrants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Conversion of Convertible Debt to Common Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of Common Stock and warrants, net issuance costs |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at September 30, 2021 |
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
The accompanying notes are an integral part of these financial statements.
| F-20 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Statements of Stockholder’s and Member’s Equity (Deficit) Continued
Unaudited
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Preferred Stock |
|
|
|
|
|
|
|
|
|
|
| ||||||||||||||||||||||||||
|
| Series 1c Preferred |
|
| Series 1d Preferred |
|
| Preferred Stock |
|
| Warrants |
|
| Common Stock |
|
| Additional |
|
|
|
|
| ||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| Par |
|
|
|
| Par |
|
|
|
| Par |
|
| Paid-in |
|
| Accumulated |
|
|
| |||||||||||||||||||||
|
| Units |
|
| Amount |
|
| Units |
|
| Amount |
|
| Shares |
|
| Value |
|
| Shares |
|
| Value |
|
| Shares |
|
| Value |
|
| Capital |
|
| Deficit |
|
| Total |
| |||||||||||||
Balance at December 31, 2019 |
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) | |||||||||||
Class B Common Units issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) |
Balance at March 31, 2020 |
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) | ||
Series 1c Preferred Units issued |
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| |||
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) |
Balance at June 30, 2020 |
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) | ||
Exercise of Series 1a Preferred Warrant Units |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| |
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) |
Balance at September 30, 2020 |
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) | ||
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) |
Balance at December 31, 2020 |
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) | ||
Series 1d Preferred Units issued |
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| |||
Class B Common Units forfeited |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| |
Conversion of Common Units to Common Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
|
| $ |
| ||||
Conversion of Preferred Units to Preferred Stock |
|
| ( | ) |
| $ | (6,491,592 | ) |
|
| ( | ) |
| $ | ( | ) |
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| ||||
Conversion of Warrant Units to Preferred Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | - |
| |||
Stock-based compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| ||
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) |
Balance at March 31, 2021 |
|
|
|
| $ |
|
|
| - |
|
| $ | - |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ | ( | ) |
| $ |
| ||||||||||
Stock-based compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| ||
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) |
Balance at June 30, 2021 |
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ |
|
|
|
|
| $ | ( | ) |
| $ | ( | ) | |||||||
Redemption of Series 1c preferred shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
| $ | ( | ) |
Conversion of Preferred Stock to Common Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
|
| $ | - |
| |||
Conversion of Preferred Stock Warrants to Common Stock Warrants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | - |
| ||
Conversion of Convertible Debt to Common Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
|
| $ |
| ||||
Issuance of Common Stock and warrants, net issuance costs |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
|
|
|
|
|
|
|
|
| $ |
| ||||
Stock-based compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ |
| ||
Net loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $ | ( | ) |
| $ | ( | ) |
Balance at September 30, 2021 |
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
| - |
|
| $ | - |
|
|
|
|
| $ |
|
|
|
|
| $ | ( | ) |
| $ |
| ||||
The accompanying notes are an integral part of these financial statements.
| F-21 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Condensed Statements of Cash Flows
|
| For the nine months ended September 30, |
| |||||
|
| 2021 |
|
| 2020 |
| ||
|
| (unaudited) |
|
| (unaudited) |
| ||
Cash flows from operating activities: |
|
|
|
|
|
| ||
Net loss |
| $ | ( | ) |
| $ | ( | ) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
|
|
Stock-based compensation |
|
|
|
|
|
| ||
Amortization of debt discount costs |
|
|
|
|
|
| ||
Depreciation of fixed assets |
|
|
|
|
|
| ||
Increase/(decrease) in cash resulting from changes in: |
|
|
|
|
|
|
|
|
Prepaid expenses and other current assets |
|
| ( | ) |
|
| ( | ) |
Accounts payable |
|
|
|
|
| ( | ) | |
Accrued and other current liabilities |
|
|
|
|
|
| ||
License and settlement agreement |
|
|
|
|
| ( | ) | |
Total adjustments to reconcile net loss to net cash used in operations |
|
|
|
|
| ( | ) | |
Net cash used in operating activities |
|
| ( | ) |
|
| ( | ) |
Cash flows from financing activities: |
|
|
|
|
|
|
|
|
Proceeds from issuance of common stock and warrants, net of issuance costs |
|
|
|
|
|
| ||
Redemption of Series 1c preferred stock |
|
| ( | ) |
|
|
| |
Payments on debt |
|
| ( | ) |
|
| ( | ) |
Proceeds from Paycheck Protection Plan loan |
|
|
|
|
|
| ||
Net proceeds from issuance of convertible subordinated promissory notes |
|
|
|
|
|
| ||
Proceeds from issuance of Series 1d preferred units |
|
|
|
|
|
| ||
Proceeds from issuance of Series 1c preferred units |
|
|
|
|
|
| ||
Proceeds from exercise of Series 1a preferred warrant units |
|
|
|
|
|
| ||
Net cash provided by financing activities |
|
|
|
|
|
| ||
Net increase (decrease) in cash |
|
|
|
|
| ( | ) | |
Cash at beginning of period |
|
|
|
|
|
| ||
Cash at end of period |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Supplemental disclosures: |
|
|
|
|
|
|
|
|
Cash paid for interest |
| $ |
|
| $ |
| ||
Cash paid for taxes |
| $ |
|
| $ |
| ||
Non-cash financing activities: |
|
|
|
|
|
|
|
|
Conversion of common and preferred units and warrants to common and preferred stock and warrants |
| $ |
|
| $ |
| ||
Conversion of convertible subordinated promissory notes to Series 1d preferred units |
| $ |
|
| $ |
| ||
Deemed dividend upon amendment to the terms to the Series 1d convertible preferred stock |
| $ |
|
| $ |
| ||
Conversion of convertible subordinated promissory notes and accrued interest to common shares at IPO |
| $ |
|
| $ |
| ||
Conversion of preferred stock to common stock at IPO |
| $ |
|
| $ |
| ||
The accompanying notes are an integral part of these financial statements.
| F-22 |
| Table of Contents |
DERMATA THERAPEUTICS, INC.
(FORMERLY DERMATA THERAPEUTICS, LLC)
Notes to Financial Statements
1. Organization and Basis of Presentation
Dermata Therapeutics, Inc., (the “Company”), was formed in December 2014 as a Delaware limited liability company (“LLC”) under the name Dermata Therapeutics, LLC. On March 24, 2021, the Company converted from an LLC to a Delaware C-corporation and changed its name to Dermata Therapeutics, Inc. Any references in these Notes to Financial Statements to equity securities as “units” refer to pre-conversion equity securities and any references to “shares” or “stock” in these Notes to Financial Statements refer to post-conversion equity securities. The Company is a clinical-stage biotechnology company focused on the treatment of medical and aesthetic skin conditions.
Initial Public Offering
On August 17, 2021, the Company completed its initial public offering (“IPO”), in which it sold
Each of the following occurred in connection with the completion of the IPO in August 2021:
| - | The sale of |
|
|
|
| - | The conversion of |
|
|
|
| - | The conversion of $ |
|
|
|
| - | The conversion of |
Each purchaser in the IPO received one share of common stock and one warrant to purchase one share of common stock at a combined offering price of $7.00. Each warrant to purchase common stock entitles the holder to purchase one share of common stock at an exercise price of $7.00 per share, are immediately exercisable, and expire five years from the date of issuance. The Company evaluated the terms of the warrants issued and determined that they should be classified as equity instruments.
The Company’s shares of common stock and warrants are listed on the Nasdaq Stock Market LLC under the symbols “DRMA,” and “DRMAW,” respectively, and both began trading in August 2021.
After the IPO, there were no shares of preferred stock or preferred stock warrants outstanding. Prior to the IPO, the Company had
Reverse Stock Split
On July 1, 2021, the Company effected a reverse split of shares of the Company’s common stock at a ratio of 1-for-20.5 pursuant to an amendment to the Company’s certificate of incorporation approved by the Company’s board of directors and stockholders. The par value was not adjusted as a result of the reverse split. All issued and outstanding common stock shares and per share amounts contained in the financial statements have been retroactively adjusted to reflect this reverse stock split for all periods presented, and the conversion ratios for the Company’s outstanding preferred stock was adjusted accordingly. See Note 6 - Equity Securities for additional information.
| F-23 |
| Table of Contents |
Liquidity and Going Concern Uncertainty
Since its inception, the Company has devoted substantially all of its resources to research and development activities and has not generated any revenue or commercialized any product candidates. As of September 30, 2021, cash totaled $12.6 million and the Company had an accumulated deficit of $
Historically, the Company’s principal sources of cash have included proceeds from the issuance of common and preferred equity units and proceeds from the issuance of debt. The Company’s principal uses of cash have included cash used in operations and payments for license rights. The Company expects that the principal uses of cash in the future will be for continuing operations, funding of research and development, conducting preclinical studies and clinical trials, and general working capital requirements. The Company expects that as research and development expenses continue to grow, it will need to raise additional capital to sustain operations and research and development. The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. The financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from the possible inability of the Company to continue as a going concern.
Management’s Plan to Continue as a Going Concern
To continue as a going concern, the Company will need, among other things, to raise additional capital resources. Until the Company can generate significant cash from operations, management’s plans to obtain such resources for the Company include proceeds from offerings of the Company’s equity securities or debt, or transactions involving product development, technology licensing or collaboration. Management can provide no assurance that any sources of a sufficient amount of financing or collaboration agreements will be available to the Company on favorable terms, if at all. Additionally, the COVID-19 pandemic continues to evolve and has already disrupted global financial markets. The Company’s ability to raise additional capital may be adversely impacted by potential worsening of global economic conditions and the recent disruptions to, and volatility in, the credit and financial markets in the United States and worldwide resulting from the pandemic. If the disruption persists or deepens, the Company could experience an inability to access additional capital.
The Company has raised additional capital through the initial public offering of its common stock and warrants; however, management’s current plans do not alleviate substantial doubt about the Company’s ability to continue as a going concern.
Basis of Presentation
The accompanying unaudited financial statements have been prepared in accordance with accounting principles generally accepted in the United States (“GAAP”) for interim financial information and in accordance with the instructions to Form 10-Q and Article 10 of Regulation S-X. Accordingly, since they are interim statements, the accompanying financial statements do not include all of the information and notes required by GAAP for complete financial statements. In the opinion of management, the accompanying financial statements reflect all adjustments (consisting of normal recurring adjustments) that are necessary for a fair statement of the financial position, results of operations, cash flows, and stockholders’ equity for the interim periods presented. Interim results are not necessarily indicative of results for a full year. The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the amounts reported in the financial statements and the accompanying notes. Actual results could differ materially from those estimates.
| F-24 |
| Table of Contents |
2. Summary of Significant Accounting Policies
Use of Estimates
The Company’s financial statements are prepared in accordance with GAAP. The preparation of the Company’s financial statements requires management to make estimates and assumptions that impact the reported amounts of assets, liabilities, and expenses and disclosure of contingent assets and liabilities in the financial statements and accompanying notes. On an ongoing basis, management evaluates these estimates and judgments, including those related to accrued research and development expenses, stock-based compensation, and the estimated fair values of equity instruments. Management evaluates its estimates on an ongoing basis. The Company bases its estimates on various assumptions that it believes are reasonable under the circumstances. Actual results may differ from these estimates under different assumptions or conditions.
Segment Information
Operating segments are defined as components of an enterprise about which separate discrete information is available for evaluation by the chief operating decision maker, or decision-making group, in deciding how to allocate resources and in assessing performance. The Company and the Company’s chief operating decision maker view the Company’s operations and manage its business in one operating segment, which is the business of developing and commercializing pharmaceuticals. The Company operates in only one segment.
Deferred Financing Costs
The Company capitalizes certain legal, accounting, and other fees and costs that are directly attributable to in-process equity financings as deferred offering costs until such financings are completed. Upon the completion of an equity financing, these costs are recorded as a reduction of additional paid-in capital of the related offering. Upon the completion of the IPO in August 2021, approximately $
Cash
The Company deposits its cash with reputable financial institutions that are insured by the Federal Deposit Insurance Corporation (“FDIC”). This cash is held in checking and savings accounts. At times, deposits held may exceed the amount of insurance provided by the FDIC. The Company has not experienced any losses in its cash and believes they are not exposed to any significant credit risk.
Fair Value Measurement
The Company uses a three-tier fair value hierarchy to prioritize the inputs used in the Company’s fair value measurements. These tiers include Level 1, defined as observable inputs such as quoted prices in active markets for identical assets; Level 2, defined as inputs other than quoted prices in active markets that are either directly or indirectly observable; and Level 3, defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions. The Company believes the carrying amount of cash, accounts payable, accrued expenses and debt approximate their estimated fair values due to the short-term maturities of these financial instruments.
Fixed Assets
Fixed assets consist of furniture and fixtures and computer equipment. Fixed assets are stated at cost less accumulated depreciation and amortization. Additions, improvements, and major renewals are capitalized. Maintenance, repairs, and minor renewals are expensed as incurred. Depreciation is determined using the straight-line method over the estimated useful lives of the assets, which is primarily three years. Depreciation and amortization expense for the nine months ended September 30, 2021 and 2020 were $
| F-25 |
| Table of Contents |
Patent Costs
Patent costs related to obtaining and maintaining patent protection in both the United States and other countries are expensed as incurred. Patents costs are classified as general and administrative expenses.
Research and Development
Research and development costs consist of expenses incurred in connection with the development of the Company’s product candidates. Such expenses include expenses incurred under agreements with contract research organizations, manufacturing and supply scale-up expenses and the cost of acquiring and manufacturing preclinical and clinical trial supply, outsourced laboratory services, including materials and supplies used to support the Company’s research and development activities, and payments made for license fees and milestones that have not been demonstrated to have commercial value. Such costs are expensed in the periods in which they are incurred. Upfront payments and milestone payments for licensed technology are expensed as research and development as incurred or when the milestone is achieved or is determined to be probable of being achieved. Advanced payments for goods or services to be received in the future for research and development activities are recorded as prepaid expenses and expensed as the related goods are received or services are performed.
Income Taxes
From inception until March 24, 2021, the Company operated as a limited liability company taxed as a partnership. Therefore, any income tax liability or benefit through that date accrued to the Company’s members. Since March 24, 2021, the Company has operated as a C-Corporation and accounts for income taxes under the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements. Under this method, deferred tax assets and liabilities are determined based on the differences between the financial statements and tax basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. The Company recognizes net deferred tax assets to the extent that the Company believes these assets are more likely than not to be realized. In making such a determination, management considers all available positive and negative evidence. The Company records the difference between the benefit recognized and measured pursuant to the accounting guidance on accounting for uncertain tax positions taken or expected to be taken on the Company’s tax return. The Company records uncertain tax positions on the basis of a two-step process whereby (1) management determines whether it is more likely than not that the tax positions will be sustained on the basis of the technical merits of the position and (2) for those tax positions that meet the more-likely-than-not recognition threshold, management recognizes the largest amount of tax benefit that is more than 50% likely to be realized upon ultimate settlement with the related tax authority. The liabilities are adjusted in light of changing facts and circumstances, such as the outcome of tax audits. To the extent that the assessment of such tax positions changes, the change in estimate is recorded in the period in which the determination is made. There are no uncertain tax positions.
Stock-Based Compensation
In March 2021, the Company’s board of directors and shareholders approved the 2021 Omnibus Equity Incentive Plan (“the 2021 Plan”). For stock options granted under the 2021 Plan, the Company measures and recognizes compensation expense for all stock-based awards made to employees, directors, and non-employees, based on estimated fair values recognized using the straight-line method over the requisite service period. The fair value of options to purchase common stock granted to employees is estimated on the grant date using the Black-Scholes valuation model. The calculation of stock-based compensation expense requires that the Company make certain assumptions and judgments about variables used in the Black-Scholes model, including the expected term of the stock-based award, expected volatility of the underlying common stock, dividend yield, and the risk-free interest rate. Forfeitures are accounted for in the period they occur. Refer to Note 7- Equity Incentive Plan for further discussion.
Net Loss Per Common Unit/Share
On March 24, 2021, the Company converted from an LLC to a C-corporation. Upon the conversion, each outstanding common unit and preferred unit was converted into one share of common stock and preferred stock, respectively. Common units had similar rights and characteristics of common stock issued upon the conversion. In calculating net loss per share, the Company retrospectively applied the effects of the conversion to the number of common units outstanding prior to the conversion. Net loss per share for periods prior to the conversion to a C-corporation refers to net loss per common unit.
| F-26 |
| Table of Contents |
Basic net loss per unit/share is calculated by dividing net loss attributable to common unitholders or shareholders by the weighted-average number of units or shares outstanding during the period, without consideration of common unit or share equivalents. Diluted net loss per unit or share is calculated by adjusting weighted-average shares outstanding for the dilutive effect of common unit or share equivalents outstanding for the period. For purposes of the diluted net loss per unit or share calculation, preferred units or shares, profit interests, and warrants to purchase preferred units or shares are considered to be common unit or share equivalents but are excluded from the calculation of diluted net loss per common unit or share if their effect would be anti-dilutive.
As the Company has reported a net loss for the periods presented, diluted net loss per common unit or share is the same as the basic net loss per common unit or share for the periods presented.
|
| Three Months Ended September 30, |
|
| Nine Months Ended September 30, |
| ||||||||||
|
| 2021 |
|
| 2020 |
|
| 2021 |
|
| 2020 |
| ||||
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
Net loss |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
Deemed dividend upon redemption of 5,221,156 shares of Series 1c convertible preferred stock |
| $ |
|
| $ |
|
| $ |
|
| $ |
| ||||
Deemed dividend upon amendment of the terms to the Series 1d convertible preferred stock |
| $ |
|
| $ |
|
| $ |
|
| $ |
| ||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss attributable to common stockholders |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted net loss per common unit/share |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
| $ | ( | ) |
Weighted-average basic and diluted common units/shares |
|
|
|
|
|
|
|
|
|
|
|
| ||||
The common unit or share equivalents that are not included in the calculation of diluted net loss per common unit or share but could potentially dilute basic earnings per share in the future are as follows:
|
| As of |
| |||||
|
| September 30, 2021 |
|
| September 30, 2020 |
| ||
|
|
|
|
|
|
| ||
Series 1 Preferred Units/Shares |
|
| - |
|
|
|
| |
Series 1a Preferred Units/Shares |
|
| - |
|
|
|
| |
Series 1a Preferred Warrant |
|
| - |
|
|
|
| |
Series 1b Preferred Units/Shares |
|
| - |
|
|
|
| |
Series 1c Preferred Units/Shares |
|
| - |
|
|
|
| |
Series 1d Preferred Units/Shares |
|
| - |
|
|
| - |
|
Class B Common Units Profits Interests |
|
| - |
|
|
|
| |
Common Stock Options |
|
|
|
|
| - |
| |
Common Stock Warrants |
|
|
|
|
| - |
| |
Total potentially dilutive securities |
|
|
|
|
|
| ||
| F-27 |
| Table of Contents |
Recently Adopted Accounting Pronouncements
In December 2019, the Financial Accounting Standards Board (“FASB”) issued Account Standards Update (“ASU”) No. 2019-12, Income Taxes (Topic 740): Simplifying the Accounting for Income Taxes (“ASU 2019-12”). ASU 2019-12 simplifies the accounting for income taxes by removing certain exceptions to the general principles in Topic 740. ASU 2019-12 also improves the consistent application, and the simplification, of other areas of Topic 740 by clarifying and amending existing guidance. ASU 2019-12 is effective for fiscal years beginning after December 15, 2020, and interim periods within those fiscal years, with early adoption permitted. Adoption of this new guidance on January 1, 2021 did not have an impact on the Company’s financial position and results of operations.
In August 2020, the FASB issued ASU 2020-06, Debt - Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging - Contracts in an Entity’s Own Equity (Subtopic 815-40) (“ASU 2020-06”). ASU 2020-06 eliminates the beneficial conversion and cash conversion accounting models for convertible instruments. It also amends the accounting for certain contracts in an entity’s own equity that are currently accounted for as derivatives because of specific settlement provisions. In addition, ASU 2020-06 modifies how particular convertible instruments and certain contracts that may be settled in cash or shares impact the diluted earnings per share computation. The amendments in ASU 2020-06 are effective for smaller reporting companies as defined by the U.S. Securities and Exchange Commission (“SEC”) for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years. Early adoption is permitted, but not earlier than fiscal years beginning after December 15, 2020. Adoption of this new guidance on January 1, 2021 did not have an impact on the Company’s financial position and results of operations.
3. Balance Sheet Details
The following provides certain balance sheet details:
|
| September 30, |
|
| December 31, |
| ||
|
| 2021 |
|
| 2020 |
| ||
|
|
|
|
|
|
| ||
Prepaid expenses and other current assets |
|
|
|
|
|
| ||
Prepaid insurance |
| $ |
|
| $ |
| ||
Prepaid research and development costs |
|
|
|
|
|
| ||
Total prepaid expenses and other current assets |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Fixed assets |
|
|
|
|
|
|
|
|
Furniture and office equipment |
| $ |
|
| $ |
| ||
Computer equipment |
|
|
|
|
|
| ||
|
|
|
|
|
|
| ||
Less: accumulated depreciation and amortization |
|
| ( | ) |
|
| ( | ) |
Total fixed assets, net |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Accrued and other current liabilities |
|
|
|
|
|
|
|
|
Accrued interest payable |
| $ |
|
| $ |
| ||
Accrued compensation and benefits |
|
|
|
|
|
| ||
Accrued research and development costs |
|
|
|
|
|
| ||
Total accrued and other current liabilities |
| $ |
|
| $ |
| ||
| F-28 |
| Table of Contents |
4. Subordinated Convertible Promissory Notes
In July and October 2020, the Company issued an aggregate of $
On January 27, 2021, the Company amended the terms of the Notes to increase the maximum amount of convertible promissory notes to be issued from $
In March 2021, the Company further amended the terms of the Notes to allow for the conversion of the Notes into Series 1d Preferred Units at the same price as purchasers of Series 1d Preferred Units. As of March 15, 2021, $
The Company considers the above modification of the Notes in March 2021 to be a substantial modification requiring extinguishment accounting under Accounting Standards Codification (“ASC”) 470-50-40-10. Based upon an independent valuation of the reacquisition price of the Notes, the difference between the reacquisition price and the net carrying amount of the Notes immediately prior to the modification is not material to the financial statements.
In connection with the Company’s IPO in August 2021, the outstanding principal of the Notes and accrued interest totaling $
As of September 30, 2021, the Company had no promissory notes outstanding.
5. Long-Term Debt
In February 2017, the Company entered into a Loan and Security Agreement with Silicon Valley Bank (“SVB”) whereas SVB agreed to provide term loans to the Company in two tranches. The first tranche of $
In connection with the Loan and Security Agreement, SVB also received warrant units to purchase, at any time after February 9, 2017 and prior to February 9, 2027,
In June 2019, the Company and SVB entered into a First Amendment to the Loan and Security Agreement whereby if the Company did not achieve certain capital milestones by December 1, 2019, term loan principal payments would be deferred from December 21, 2019 through May 1, 2020 with the deferred principal payments being payable in equal monthly installments, in addition to those principal payments already scheduled to be paid, starting on June 1, 2020 and extending through the February 9, 2021 maturity date of the term loan. In addition, if those principal payments were deferred for that six-month period, a non-refundable amendment fee of $
| F-29 |
| Table of Contents |
The non-refundable amendment fee of $
In January and February 2021, the company paid the final principal payments of $
As of September 30, 2021, the Company had no long-term debt outstanding.
Paycheck Protection Program
On April 22, 2020, the Company received proceeds of a $
6. Equity Securities
Common Stock and Preferred Stock
On March 24, 2021, the Company entered into a Plan of Conversion (“Conversion”) whereby the Company converted from an LLC under the laws of the State of Delaware to a Delaware C-corporation with the name Dermata Therapeutics, Inc. In connection with the Conversion, each fully paid Preferred and Common Unit in the LLC was converted into a like number of shares of Preferred and Common Stock of the Company with a par value of $
On July 1, 2021, the Company effected a reverse split of shares of the Company’s common stock at a ratio of 1-for-20.5 pursuant to an amendment to the Company’s certificate of incorporation approved by the Company’s board of directors and stockholders. The par value was not adjusted as a result of the reverse split. All issued and outstanding common stock share and per share amounts contained in the financial statements have been retroactively adjusted to reflect this reverse split for all periods presented.
On August 17, 2021, the Company completed its IPO, in which it sold
Each of the following occurred in connection with the completion of the IPO in August 2021:
| - | The sale of |
|
|
|
| - | The conversion of |
|
|
|
| - | The conversion of $ |
|
|
|
| - | The conversion of |
| F-30 |
| Table of Contents |
After the IPO, there were no shares of preferred stock or preferred stock warrants outstanding. Prior to the IPO, the Company had
The Company’s total common stock issued and outstanding was
Series 1 Preferred Units
From the Company’s formation on December 8, 2014 through 2016, the Company issued
Series 1a Preferred Units
In 2016, the Company issued
Series 1b Preferred Units
In 2018, the Company issued
Series 1c Preferred Units
On June 14, 2019, the Company closed participation in a $
In June 2019,
The Company’s Series 1c Preferred Units were converted to preferred stock during the first quarter of 2021. Additionally, in August 2021, the Company converted all preferred stock into common stock. No Series 1c Preferred Units were outstanding as of September 30, 2021.
| F-31 |
| Table of Contents |
Series 1d Preferred Units
In March 2021, the Company issued
Class A Common Units
During 2014 and 2015, the Company issued
Class B Common Units
The Company had
Liquidation Preference
Prior to the Company’s IPO in August 2021, the Company’s preferred units were subject to liquidation preferences contained herein. So long as there were no Series A Preferred Units outstanding at the time of a liquidity event, any liquidity event proceeds would have been distributed as follows: First, the Series 1d Preferred Units had a two times preference in liquidation over the Series 1c Preferred Units and then participated with the Series 1c, 1b and 1a Preferred Units once the Series 1c Preferred Unit preferences had been satisfied. Second, proceeds to Series 1c Preferred Unit holders sufficient to cover two times their Series 1c investment; third, proceeds to Series 1, Series 1a, Series 1b, Series 1c, and Series 1d Preferred Unit holders sufficient to cover interest at the rate of
| F-32 |
| Table of Contents |
Conversion Rights
Prior to the Company’s IPO in August 2021, the Company’s preferred units were subject to conversion rights contained herein. Upon the first issuance by the Company of any Series A Preferred Units, each Series 1 Preferred Unit and each Series 1a Preferred Unit and each Series 1b Preferred Unit and each Series 1c Preferred Unit and each Series 1d Preferred Unit would have automatically been converted into the number of Series A Preferred Units equal to the sum of the Unit Value with respect to such Series 1 Preferred Unit or Series 1a Preferred Unit or Series 1b Preferred Unit or Series 1c Preferred Unit or Series 1d Preferred Unit as of the conversion date divided by the product of
Stockholders’ Agreements
On March 24, 2021, in connection with the conversion of Dermata Therapeutics, LLC into a Delaware corporation, the Company entered into a Stockholders’ Agreement (as amended, the Stockholders’ Agreement) with all of its then-existing stockholders, including Proehl Investment Ventures, LLC and Hale Biopharma Ventures, LLC. The Stockholders’ Agreement among other things, provided for certain restrictions on transfer of the Company’s shares of capital stock, set forth agreements and understandings with respect to how shares of its capital stock held by the stockholders party thereto will have been voted on, or tendered in connection with, an acquisition of the Company and provided for certain voting rights with respect to the election of directors. In addition, pursuant to the Stockholders’ Agreement, holders of the Company’s Series 1a Preferred Stock were entitled to purchase, at any time prior to March 14, 2026, such number of shares of the Company’s Series 1a Preferred Stock as such Series 1a Stockholder shall request, up to an aggregate number of shares of Series 1a Preferred Stock not to exceed the product of
On June 29, 2021, with effectiveness on July 1, 2021, the Company’s board of directors amended its Certificate of Incorporation to adjust the conversion price and certain conversion mechanics of the Company’s issued and outstanding Series 1d Preferred Stock, whereby each share of Series 1d Preferred stock would convert into such number of Common Stock as determined by dividing (i) the product of (a) the Original Issue Price for the Series 1d Preferred Stock, multiplied by (b) 1.2, rounded to the nearest whole cent, by (ii) the 80% of the initial public per share offering price in the IPO. The Series 1d conversion shall not be subject to further adjustment for any stock split.
On June 29, 2021, with effectiveness on July 1, 2021, the Company’s board of directors approved an amendment to the 2021 Plan to increase the number of shares of Common Stock available for issuances from
| F-33 |
| Table of Contents |
On June 29, 2021, the Company’s board of directors approved a
On July 12, 2021, the Company’s board of directors amended its Certificate of Incorporation to further adjust the conversion price and certain conversion mechanics of the Company’s issued and outstanding Series 1d Preferred Stock. The two amendments to the Series 1d Preferred Stock conversion terms were combined for purposes of accounting for the amendments. In order to determine if these amendments resulted in a modification or extinguishment of the Series 1d Preferred Stock, pursuant to the related authoritative guidance, the Company engaged an independent third-party valuation firm to assist with determining the fair value of the Series 1d Preferred Stock immediately before the change in conversion terms, as well as immediately after the change in conversion terms. This resulted in a substantive increase in fair value, and as such, the Company determined the amendments resulted in extinguishment accounting. Accordingly, the Company applied ASC 260, Earnings per Share, and ASC 470, Debt, by analogy to determine the appropriate measurement and presentation. The Company compared the fair value of the Series 1d Preferred Stock, as amended, to its carrying value and recorded the resulting difference of approximately $2.3 million as a deemed dividend for the Series 1d preferred shareholders. The Company recorded the deemed dividend to additional paid-in capital because the Company is in an accumulated deficit position, thereby increasing the net loss attributable to the common shareholders for the three and nine months ended September 30, 2021.
On July 30, 2021, the Company entered into a Second Amendment to the License and Settlement Agreement (or, the Second License Amendment), whereby, for the settlement of certain disputes arising under the License Agreement, the Company agreed to exchange the shares of Series 1c Preferred Stock owned by Villani, Inc. (“Villani”) for an increase of milestone payments and royalty rates due to Villani under the License Agreement. On July 30, 2021, Villani surrendered
On August 14, 2021, the Company’s board of directors approved an amendment to the Company’s Certificate of Incorporation to increase the number of shares of Common Stock authorized to
Warrants
Warrants issued at IPO
On August 17, 2021, the Company completed its IPO, in which it sold
The Company evaluated the terms of the warrants issued at the IPO and determined that they should be classified as equity instruments based upon accounting guidance provided in ASC 480, Distinguishing Liabilities from Equity, and ASC 815, Derivatives and Hedging. Since the Company determined that the warrants were equity classified, the Company recorded the proceeds from the IPO, net of issuance costs, within common stock at par value and the balance of proceeds to additional paid in capital. The fair value of each warrant on August 17, 2021 was $
As of September 30, 2021, the Company had
| F-34 |
| Table of Contents |
Warrants issued with Class B Common Units
In March 2021, the Company granted Class B Common Units Profits interests to certain former employees and consultants. In connection with the conversion from an LLC to a C-Corporation, the Company converted
As of September 30, 2021, the Company had
Warrants issued with Series 1a Preferred Units
In connection with the issuance of
In connection with the Loan and Security Agreement, SVB also received Warrant Units to purchase, at any time after February 9, 2017 and prior to February 9, 2027,
In July 2021, the Company effected a reverse split of shares of the Company’s common stock at a ratio of 1-for-20.5, and the conversion ratio of the preferred stock was adjusted accordingly. In August 2021, in connection with the Company’s IPO, the outstanding Series 1a preferred warrants were converted into
As of September 30, 2021, the Company had
7. Equity Incentive Plan
Under the Company’s 2021 Omnibus Equity Incentive Plan (the “2021 Plan”), the Company may grant options to purchase common stock, restricted stock awards, performance stock awards, incentive bonus awards, other cash-based awards or directly issue shares of common stock to employees, directors, and consultants of the Company. The 2021 Plan provides for the issuance of up to
As of September 30, 2021, there remain an additional
| F-35 |
| Table of Contents |
Stock Award Activity
A summary of the Company’s Equity Plans stock option activity is as follows:
|
| Number of Options Outstanding |
|
| Weighted-Average Exercise Price |
|
| Weighted-Average Remaining Contractual Term (in Years) |
| |||
Balance at December 31, 2020 |
|
|
|
| $ |
|
|
|
| |||
Options granted |
|
|
|
|
|
|
|
|
| |||
Options exercised |
|
| - |
|
|
| - |
|
|
| - |
|
Options cancelled |
|
| - |
|
|
| - |
|
|
| - |
|
Balance at September 30, 2021 |
|
|
|
| $ |
|
|
|
| |||
|
|
|
|
|
|
|
|
|
|
|
|
|
Options exercisable at September 30, 2021 |
|
|
|
| $ |
|
|
|
| |||
The aggregate intrinsic value of options exercisable as of September 30, 2021 is calculated as the difference between the exercise price of the underlying options and the closing market price of the Company’s common stock on that date, which was $
Fair Value Measurement
The Company uses the Black-Scholes option valuation model, which requires the use of highly subjective assumptions, to determine the fair value of stock-based awards. The fair value of each employee stock option is estimated on the grant date under the fair value method using the Black-Scholes model. The estimated fair value of each stock option is then expensed over the requisite service period, which is generally the vesting period. The assumptions and estimates that the Company uses in the Black-Scholes model are as follows:
| · | Fair Value of Common Stock. The estimated fair value of the common stock underlying the Company’s stock option plan was determined by management by considering various factors as discussed below. All options to purchase shares of the Company’s common stock are intended to be exercisable at a price per share not less than the per-share fair value of the Company’s common stock underlying those options on the date of grant. In the absence of a public trading market for the Company’s common stock, before the initial public offering, on each grant date, the Company developed an estimate of the fair value of its common stock based on the information known to the Company on the date of grant, upon a review of any recent events and their potential impact on the estimated fair value per share of the common stock and in part on input from an independent third-party valuation firm. After the Company’s initial public offering, the fair value of common stock is measured as the Company’s closing price of common stock on the date of grant. |
|
|
|
| · | Risk-Free Interest Rate. The Company bases the risk-free interest rate used in the Black-Scholes valuation model on the implied yield available on U.S. Treasury zero-coupon issues with a term equivalent to that of the expected term of the options. |
|
|
|
| · | Expected Term. The expected term represents the period that the Company’s stock-based awards are expected to be outstanding. Because of the limitations on the sale or transfer of the Company’s common stock as a privately held company, the Company does not believe its historical exercise pattern is indicative of the pattern it will experience as a publicly traded company. The Company plans to continue to use the SAB 110 simplified method until it has sufficient trading history as a publicly traded company. |
|
|
|
| · | Volatility. The Company determines the price volatility based on the historical volatilities of industry peers as it has limited trading history for its common stock price. Industry peers consist of several public companies in the biotechnology industry with comparable characteristics, including clinical trials progress and therapeutic indications. |
|
|
|
| · | Dividend Yield. The expected dividend assumption is based on the Company’s current expectations about its anticipated dividend policy. To date, the Company has not declared any dividends to common shareholders, and therefore the Company has used an expected dividend yield of zero. |
| F-36 |
| Table of Contents |
The following table presents the weighted-average assumptions used for the stock option grants for the three and nine months ended September 30, 2021:
|
| Three Months Ended 2021 |
|
| Nine Months Ended September 30, 2021 |
| ||
Grant date fair value |
| $ |
|
| $ |
| ||
Risk-free interest rate |
|
| % |
|
| % | ||
Dividend yield |
|
| % |
|
| % | ||
Expected life in years |
|
|
|
|
|
| ||
Expected volatility |
|
| % |
|
| % | ||
Stock-based Compensation Expense
In general, stock-based compensation is allocated to research and development expense or general and administrative expense according to the classification of cash compensation paid to the employee, director, or consultant to whom the stock award was granted.
On March 24, 2021, in connection with the conversion from an LLC to a C-Corporation, the Company converted
The following table summarizes the total stock-based compensation expense included in the Company’s statements of operations for the periods presented:
|
| Three Months Ended September 30, |
|
| Nine Months Ended September 30, |
| ||||||||||
|
| 2021 |
|
| 2020 |
|
| 2021 |
|
| 2020 |
| ||||
Research and development |
| $ |
|
| $ |
|
| $ |
|
| $ |
| ||||
General and administrative |
|
|
|
|
|
|
|
|
|
|
|
| ||||
|
| $ |
|
| $ |
|
| $ |
|
| $ |
| ||||
As of September 30, 2021, total unrecognized compensation cost related to stock options was approximately $
| F-37 |
| Table of Contents |
8. 401(k) Plan
The Company sponsors a 401(k) savings plan for all eligible employees. The Company may make discretionary matching contributions to the plan to be allocated to employee accounts based upon employee deferrals and compensation. To date, the Company has not made any matching contributions into the savings plan.
9. License Agreements
On March 31, 2017, the Company entered into a license agreement, as amended (“The License Agreement”) with Villani, Inc. whereby Villani has granted the Company an exclusive, sub-licensable, royalty-bearing license (“The License”) under the Licensed Patents (as defined in the License Agreement), to formulate, develop, seek regulatory approval for, make or sell products that contain Spongilla lacustris (alone or in combination with other active or inactive ingredients) for the treatment of diseases, disorders and conditions of the skin, including but not limited to acne, rosacea, psoriasis, atopic dermatitis, seborrheic dermatitis, actinic keratosis and eczema that were developed using certain licensed know-how (“Licensed Products”). The Company is responsible for the development (including manufacturing, packaging, non-clinical studies, clinical trials and obtaining regulatory approval and commercialization (including marketing, promotion, distribution, etc.)) for all Licensed Products. In partial consideration of the License, the Company forgave a previous outstanding loan to Villani in the amount of $
The original License Agreement was amended in 2019 and, in consideration of the receipt of certain know-how and patents, the Company issued to Villani
On July 30, 2021, the Company further amended the license agreement with Villani in the Second Amendment to the License and Settlement Agreement (“Second Amendment”). In consideration of the Second Amendment, Villani exchanged the 5,221,156 Series 1c Preferred Shares issued to Villani in 2019 for an increase in milestones and royalty rates and the Company paid Villani $
10. Commitments and Contingencies
Coronavirus Pandemic
On March 11, 2020, the World Health Organization declared the outbreak of a coronavirus (COVID-19) pandemic. Significant uncertainties may arise with respect to potential shutdowns of operations or government orders to cease activities due to emergency declarations, inability to operate, employee shortages, or claims for business interruption insurance, etc. Each of these matters may have a significant impact on the future results of the Company.
Legal Proceedings
In the normal course of business, the Company may be involved in legal proceedings or threatened legal proceedings. The Company is not a party to any legal proceedings or aware of any threatened legal proceedings which are expected to have a material adverse effect on its financial condition, results of operations or liquidity.
| F-38 |
| Table of Contents |
11. Related Parties
Prior to the Company converting from an LLC to a C corporation in March 2021, the Company had two Managing Members. One of the Managing Members remained the Company’s majority stockholder upon the close of the Company’s IPO and serves as the Company’s President, Chief Executive Officer, and Chairman of the Board of Directors. The other Managing Member remained a beneficial owner upon the close of the Company’s IPO and serves as the Company’s Lead Director of the Board of Directors. Hereinafter these two Managing Members, and their affiliates, are referred to collectively as the Principal Stockholders after the completion of the IPO.
During 2020, the Managing Members and other related parties loaned the Company $
During the third quarter of 2021, the Company amended the conversion terms of its Series 1d preferred stock, as described in Note 6 - Equity Securities. As a result of the Series 1d preferred stock amendments, the Company presented a deemed dividend of approximately $
| F-39 |
Units consisting of
4,444,444 Shares of Common Stock or Pre-Funded Warrant to Purchase 4,444,444 Shares of Common Stock
and
Warrants to purchase up to 4,444,444 Shares of Common Stock

Dermata Therapeutics, Inc.
PROSPECTUS
Sole Book Running Manager
Maxim Group LLC
, 2022
146 |
| Table of Contents |
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution.
The following table indicates the expenses to be incurred in connection with the offering described in this registration statement, other than underwriting discounts and commissions, all of which will be paid by us. All amounts are estimated except the Securities and Exchange Commission registration fee.
|
| Amount |
| |
Securities and Exchange Commission registration fee |
| $ | 2,235.60 |
|
FINRA filing fee |
|
| 3,950 |
|
Underwriter Legal Fees and Expenses |
|
| 75,000 |
|
Accountants’ fees and expenses |
|
| 90,000 |
|
Legal fees and expenses |
|
| 125,000 |
|
Transfer Agent’s fees and expenses |
|
| 9,000 |
|
Printing and engraving expenses |
|
| 3,400 |
|
Miscellaneous |
|
| 11,414 |
|
Total expenses |
| $ | 320,000 |
|
Item 14. Indemnification of Directors and Officers.
As permitted by Section 102 of the Delaware General Corporation Law, we have adopted provisions in our amended and restated certificate of incorporation and bylaws that limit or eliminate the personal liability of our directors for a breach of their fiduciary duty of care as a director. The duty of care generally requires that, when acting on behalf of the corporation, directors exercise an informed business judgment based on all material information reasonably available to them. Consequently, a director will not be personally liable to us or our stockholders for monetary damages for breach of fiduciary duty as a director, except for liability for:
• | any breach of the director’s duty of loyalty to us or our stockholders; |
• | any act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; |
• | any act related to unlawful stock repurchases, redemptions or other distributions or payment of dividends; or |
• | any transaction from which the director derived an improper personal benefit. |
These limitations of liability do not affect the availability of equitable remedies such as injunctive relief or rescission. Our amended and restated certificate of incorporation also authorizes us to indemnify our officers, directors and other agents to the fullest extent permitted under Delaware law.
As permitted by Section 145 of the Delaware General Corporation Law, our bylaws provide that:
• | we may indemnify our directors, officers, and employees to the fullest extent permitted by the Delaware General Corporation Law, subject to limited exceptions; |
• | we may advance expenses to our directors, officers and employees in connection with a legal proceeding to the fullest extent permitted by the Delaware General Corporation Law, subject to limited exceptions; and |
• | the rights provided in our bylaws are not exclusive. |
Our amended and restated certificate of incorporation, to be attached as Exhibit hereto, and our amended and restated bylaws, to be attached as Exhibit hereto, provide for the indemnification provisions described above and elsewhere herein. We have entered into and intend to continue to enter into separate indemnification agreements with our directors and officers which may be broader than the specific indemnification provisions contained in the Delaware General Corporation Law. These indemnification agreements generally require us, among other things, to indemnify our officers and directors against liabilities that may arise by reason of their status or service as directors or officers, other than liabilities arising from willful misconduct. These indemnification agreements also generally require us to advance any expenses incurred by the directors or officers as a result of any proceeding against them as to which they could be indemnified. In addition, we have purchased a policy of directors’ and officers’ liability insurance that insures our directors and officers against the cost of defense, settlement or payment of a judgment in some circumstances. These indemnification provisions and the indemnification agreements may be sufficiently broad to permit indemnification of our officers and directors for liabilities, including reimbursement of expenses incurred, arising under the Securities Act of 1933, as amended, or the Securities Act.
| II-1 |
| Table of Contents |
The form of Underwriting Agreement, to be attached as Exhibit 1.1 hereto, under some circumstances provides for indemnification by the underwriters of us and our officers who sign this Registration Statement and directors for specified liabilities, including matters arising under the Securities Act.
Item 15. Recent Sales of Unregistered Securities.
Original Issuances of Stock
On March 24, 2021, we converted from a limited liability company to a Delaware corporation and we changed our name to Dermata Therapeutics, Inc., resulting in a new capital structure consisting of common stock and preferred stock, each having a par value of $0.0001. This conversion resulted in conversion of the prior Dermata members’ interests into an aggregate of 65,823,015 shares of our preferred stock (which converted into 3,701,586 shares of common stock upon the closing of our initial public offering), and 1,911,009 shares of our common stock.
In connection with our conversion to a Delaware corporation, we also issued warrants exercisable for 1,419,228 shares of our preferred stock (or the Preferred Stock Warrants), and warrants exercisable for 65,303 shares of our common stock (or the Common Stock Warrants). The Preferred Stock Warrants have an exercise price of $1.00 per share and the Common Stock Warrants have a weighted-average exercise price of $5.82 per share.
Convertible Promissory Note Offering
In July 2020, we held the first closing of the Notes for an aggregate principal amount of $2,330,000, including $500,000 from Proehl Investment Ventures, LLC. In October 2020, we held the second closing of the Notes for an aggregate principal amount of $670,000, including $420,000 from Proehl Investment Ventures, LLC. In February 2021, we held the third closing of the Notes for an aggregate principle amount of $1,556,000, including $825,000 from Proehl Investment Ventures, LLC, $100,000 from the Proehl Family Trust and $250,000 Hale Biopharma Ventures, LLC.
On March 15, 2021, we completed the conversion of$4,391,000 of Notes into 5,379,247 Series 1d Preferred Units. At that time Proehl Investment Ventures, LLC, Sean Michael Proehl Irrevocable Trusts Dated December 18, 2020 and Hale Biopharma Ventures, LLC held an aggregate principal amount of $1,745,000, $100,000 and $250,000 of Notes, respectively. Mr. Proehl, our President and Chief Executive Officer and a member of the board of directors, is the managing member of Proehl Investment Ventures, LLC and the trustee of Sean Michael Proehl Irrevocable Trusts Dated December 18, 2020. Mr. Hale, a member of the board of directors, is the managing member of Hale Biopharma Ventures, LLC. In addition, Wendell Wierenga, a member of our board of directors, held $45,000 principal amount of the Notes. Each of Messrs. Proehl, Hale and Wierenga converted their aggregate principal amounts of Notes into Series 1d Preferred Units on March 15, 2021, which units were subsequently converted into shares of our Series 1d Preferred Stock in connection with our conversion to a Delaware corporation. The shares of Series 1d Preferred Stock held by Mr. Proehl, Hale and Wierenga automatically converted into 291,831, 39,180 and 7,142 shares of common stock, respectively, upon the completion of our initial public offering, at a conversion price equal to $6.40 (80% of the initial offering price).
The Notes had an interest rate of 4.0% per annum, were unsecured, had a maturity date of December 31, 2021 and provided for conversion into our common stock upon the earlier of (i) qualified Series A Financing (as defined in the Notes) which resulted in aggregate gross proceeds to the Company of at least Ten Million Dollars ($10,000,000), or (ii) the closing of our initial public offering.
At the closing of our initial public offering the aggregate principal amount and all accrued but unpaid interest on the Notes automatically converted into an aggregate of 32,219 shares of our common stock at a conversion price of $5.60 per share (which was 80% of the initial offering price).
| II-2 |
| Table of Contents |
Stock Options
On March 24, 2021, in connection with our conversion from a limited liability company to a Delaware corporation, we issued common stock options exercisable for an aggregate of 398,199 shares of our common stock. These options have an exercise price of $5.74 per share.
Underwriter Warrants
Upon the closing of our initial public offering, we issued to the underwriters warrants exercisable for a period of five years from the closing of the initial public offering which entitle the underwriters to purchase 128,571 of shares of common stock, at an exercise price equal to 115% of the public offering price, or $8.05 per share. The warrants will not be exercisable until February 12, 2022.
Securities Act Exemptions
We deemed the offers, sales and issuances of the securities described above under “Original Issuances of Stock,” “Convertible Promissory Note Offering” and “Underwriter Warrants” to be exempt from registration under the Securities Act in reliance on Section 4(2) of the Securities Act, including Regulation D and Rule 506 promulgated thereunder, relative to transactions by an issuer not involving a public offering. All purchasers of securities in transactions exempt from registration pursuant to Regulation D represented to us that they were accredited investors and were acquiring the shares for investment purposes only and not with a view to, or for sale in connection with, any distribution thereof and that they could bear the risks of the investment and could hold the securities for an indefinite period of time. The purchasers received written disclosures that the securities had not been registered under the Securities Act and that any resale must be made pursuant to a registration statement or an available exemption from such registration.
We deemed the grants of stock options and issuances of common stock upon exercise of such options described above under “Stock Options” to be exempt from registration under the Securities Act in reliance on Rule 701 of the Securities Act as offers and sales of securities under compensatory benefit plans and contracts relating to compensation in compliance with Rule 701. Each of the recipients of securities in any transaction exempt from registration either received or had adequate access, through employment, business or other relationships, to information about us.
| II-3 |
| Table of Contents |
Item 16. Exhibits and Financial Statement Schedules.
| II-4 |
| Table of Contents |
| ||||
|
|
| ||
| ||||
| ||||
|
|
| ||
|
| |||
|
|
| ||
| ||||
| ||||
| ||||
|
|
| ||
| ||||
|
|
| ||
| ||||
| ||||
| ||||
| ||||
23.2* | Consent of Lowenstein Sandler LLP (included in Exhibit 5.1). | |||
24.1* | Power of Attorney (included on the signature page of this Registration Statement). | |||
| ||||
_________
* | Filed herewith |
† | Denotes management compensation plan or contract. |
# | Portions of this exhibit (indicated by asterisks) are omitted in accordance with the rules of the SEC. |
| II-5 |
| Table of Contents |
Item 17. Undertakings.
The undersigned registrant hereby undertakes:
(1) To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
(i) To include any prospectus required by Section 10(a)(3) of the Securities Act of 1933;
(ii) To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Securities and Exchange Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and
(iii) To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement;
(2) That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
(3) To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
(4) That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A (§230.430A of this chapter), shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
(5) That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities:
The undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser:
(i) Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424;
(ii) Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant;
(iii) The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and
(iv) Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
(6) Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act of 1933 and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act of 1933 and will be governed by the final adjudication of such issue.
| II-6 |
| Table of Contents |
SIGNATURES
Pursuant to the requirements of the Securities Act, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of San Diego, California, on the 4th day of February, 2022.
Dermata Therapeutics, Inc. |
| ||
| |||
By: | /s/ Gerald T. Proehl |
| |
| Name: | Gerald T. Proehl |
| |
| Title: | Chief Executive Officer |
| |
POWER OF ATTORNEY
We, the undersigned officers and directors of Dermata Therapeutics, Inc., hereby severally constitute and appoint Gerald T. Proehl and Kyri K. Van Hoose, and each of them singly (with full power to each of them to act alone), to sign any and all amendments (including post-effective amendments) to this registration statement (or any other registration statement for the same offering that is to be effective upon filing pursuant to Rule 462(b) under the Securities Act of 1933), and to file the same, with all exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite or necessary to be done in and about the premises, as full to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or any of them, or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act of 1933, this Registration Statement has been signed by the following persons in the capacities held on the dates indicated:
Signature | Title | Date | |||||||||
/s/ Gerald T. Proehl | Chief Executive Officer, Chairman (Principal Executive Officer) | February 4, 2022 | |||||||||
Gerald T. Proehl |
|
|
|
| |||||||
/s/ Kyri K. Van Hoose | Chief Financial Officer (Principal Financial and Accounting Officer) | February 4, 2022 | |||||||||
Kyri K. Van Hoose |
|
|
|
| |||||||
/s/ David Hale | Lead Director | February 4, 2022 | |||||||||
David Hale |
|
|
|
| |||||||
/s/ Wendell Wierenga, Ph.D. | Director | February 4, 2022 | |||||||||
Wendell Wierenga, Ph.D. |
| ||||||||||
|
|
|
|
| |||||||
/s/ Mary Fisher | Director | February 4, 2022 | |||||||||
Mary Fisher |
|
| |||||||||
|
|
| |||||||||
/s/ Andrew Sandler, M.D. | Director | February 4, 2022 | |||||||||
Andrew Sandler, M.D. |
|
| |||||||||
|
|
| |||||||||
/s/ Steven J. Mento, Ph.D. | Director | February 4, 2022 | |||||||||
Steven J. Mento, Ph.D. |
|
| |||||||||
|
|
| |||||||||
/s/ Kathleen Scott |
|
| |||||||||
Kathleen Scott | Director | February 4, 2022 | |||||||||
|
| ||||||||||
/s/ Brittany Bradrick |
|
| |||||||||
Brittany Bradrick | Director | February 4, 2022 | |||||||||
| II-7 |